2025 October 18;6(10):1481-1491. doi: 10.37871/jbres2204.
A Comprehensive Review of Computational Modeling in Tumor and Brain Disorder with a Focus on Ablation Therapies
Abdollahzadeh Jamalabadi MY*
- Bioheat transfer
- Tumor ablation
- Brain Disorder
- Porous media
- Pennes equation
Abstract
This review paper provides a comprehensive overview of computational modeling approaches applied to the study of tumors and brain disorders, with a particular focus on ablation therapies. We delve into the fundamental principles governing heat transfer and electric fields in biological tissues, as exemplified by hepatic tumor ablation. A key component of this review is the design and conceptual implementation of a parameter study, demonstrating how variations in electrical, thermal, geometric, and operational parameters can significantly influence ablation outcomes. Through simulated data and visualizations, we illustrate the sensitivity of maximum temperature achieved and necrotic tissue volume to factors such as applied voltage, ablation time, and blood perfusion rates. This work highlights the critical role of computational modeling in optimizing treatment strategies, predicting patient-specific responses, and advancing our understanding of complex biological phenomena in neuro-oncology. The insights gained from such parameter studies are invaluable for guiding the development of more effective and safer therapeutic interventions for brain tumors and related neurological disorders.
Introduction
Brain tumors and various neurological disorders represent significant challenges in modern medicine, often leading to severe morbidity and mortality. The complexity of the brain’s anatomy and physiology, coupled with the aggressive nature of many tumors, necessitates advanced therapeutic approaches. Traditional treatments, including surgery, radiotherapy, and chemotherapy, have made strides, yet limitations persist, particularly concerning treatment efficacy, patient-specific responses, and minimizing damage to healthy brain tissue [1,2].
In recent decades, computational modeling has emerged as a powerful tool to complement experimental and clinical studies in neuro-oncology. These models offer a non-invasive means to simulate complex biological processes, predict treatment outcomes, and optimize therapeutic strategies. By integrating principles from physics, engineering, and biology, computational models can provide insights into tumor growth dynamics, drug delivery, and the effects of various interventions, such as thermal ablation [3,4].
Thermal ablation, a minimally invasive technique, involves heating malignant tissue to temperatures sufficient to induce cell death (Typically above 45-50°C). This method is particularly promising for treating tumors in sensitive areas of the brain where surgical resection might be too risky. The effectiveness of thermal ablation is highly dependent on precise control over temperature distribution within the target tissue, which is influenced by numerous interconnected physical phenomena, including electric current flow, heat transfer, and blood perfusion [5].
This review aims to provide a comprehensive overview of the application of computational modeling in the context of tumor and brain disorders, with a specific emphasis on thermal ablation therapies. We will explore the underlying physical principles, discuss the design of a conceptual parameter study to understand the sensitivity of ablation outcomes to various factors, and present simulated results to illustrate key relationships. The insights derived from such computational analyses are crucial for refining existing treatments, developing novel therapeutic approaches, and ultimately improving patient outcomes in the challenging field of neuro-oncology.
Background on Brain Tumors and Neurological Disorders
Brain tumors are abnormal growths of cells within the brain or central nervous system, which can be either benign (Non-cancerous) or malignant (Cancerous). Despite their relatively lower incidence compared to other cancers, primary brain tumors, especially high-grade gliomas like glioblastoma, are notoriously aggressive and carry a poor prognosis [6]. The unique environment of the brain, including the blood-brain barrier and the intricate neural networks, presents significant challenges for effective treatment delivery and minimizing collateral damage to healthy tissue [7].
Neurological disorders encompass a wide range of conditions affecting the brain, spinal cord, and nerves. These can manifest with diverse symptoms, including cognitive impairments, mood disturbances, and motor deficits. Interestingly, psychiatric symptoms can sometimes be the sole or early indicators of an underlying brain tumor, complicating diagnosis and highlighting the need for a comprehensive understanding of brain-tumor interactions [8,9].
The complexity of brain tumors stems from their heterogeneity, invasive nature, and ability to resist conventional therapies. For instance, glioblastoma often infiltrates surrounding healthy brain tissue, making complete surgical resection challenging and contributing to high recurrence rates [10]. Furthermore, the tumor microenvironment plays a crucial role in tumor progression and resistance to treatment, influencing immune responses and drug penetration [11].
Understanding the biological and physiological characteristics of brain tumors and their impact on neurological function is paramount for developing effective treatment strategies. This includes not only the cellular and molecular mechanisms of tumor growth but also the macroscopic effects on brain dynamics and connectivity. Computational models offer a valuable platform to integrate these multi-scale complexities, providing a holistic view of the disease and enabling the exploration of various therapeutic interventions in a simulated environment.
Computational Modeling in Neuro-Oncology
Computational modeling has become an indispensable tool in neuro-oncology, offering a quantitative framework to understand complex biological processes, predict disease progression, and optimize therapeutic interventions. These models range from cellular-level simulations to macroscopic representations of tumor growth and treatment response, often integrating diverse data types from medical imaging to molecular profiles [12].
One significant application of computational modeling is in simulating brain tumor growth. Various approaches exist, including discrete models focusing on individual cell behavior, continuum models describing cell density over time, and hybrid models combining elements of both [13]. These models aim to predict tumor progression, which is crucial for guiding medical treatment and surgical planning. However, a key challenge remains in developing patient-specific models due to limited available data from clinical imaging [14].
Beyond growth prediction, computational models are vital for understanding drug delivery and treatment response. For instance, models can predict drug permeability across the blood-brain barrier, a major hurdle in brain tumor therapy, and simulate the effects of various treatments like chemotherapy and radiation [15]. This allows for In silico testing of different treatment regimens, potentially reducing the need for extensive and costly In vivo experiments.
In the context of thermal ablation, computational models are particularly powerful. The provided COMSOL Multiphysics model for hepatic tumor ablation exemplifies how coupled physics simulations can be used to predict temperature distribution and tissue damage during the procedure. This model integrates the Electric Currents interface and the Bioheat Transfer interface, accounting for resistive heating (Joule heating) and heat dissipation due to blood perfusion [15-25]. The bioheat equation, a modified heat conduction equation, is central to these simulations, incorporating terms for metabolic heat generation and heat exchange with blood flow [5].
Such models allow for detailed analysis of the thermal dose delivered to the tissue, which is directly related to the extent of irreversible tissue damage. The Arrhenius equation is commonly used to quantify thermal damage, relating the rate of tissue injury to temperature and exposure time [26-30]. By simulating these complex interactions, researchers can gain insights into the optimal placement of ablation probes, the duration and intensity of energy delivery, and the predicted volume of necrotic tissue.
Furthermore, computational models can assist in personalized medicine by integrating patient-specific imaging data. This allows for the calibration of models to individual tumor characteristics, leading to more accurate predictions of tumor growth and response to therapy [16,31-34]. The ability to forecast spatial responses to chemoradiation, for example, can help in localized treatment planning to target less responsive disease regions. The ultimate goal is to develop predictive tools that can inform clinical decisions, leading to improved patient outcomes and more effective, tailored therapies.
The field of thermal tumor therapy is increasingly focused on the development of highly sophisticated computational models to improve treatment predictability and efficacy. Recent research highlights a significant shift from the classical Pennes bioheat equation towards more complex multi-physics frameworks [15,18]. These advanced models more accurately represent biological reality by accounting for critical factors such as tissue porosity, blood perfusion, and heterogeneous tumor properties [16,27]. Studies directly compare these new approaches, demonstrating that models which incorporate fluid flow through porous media or non-Fourier heat transfer provide a superior prediction of heat distribution and tissue damage during procedures like laser and microwave ablation [25,26,28]. This enhanced modeling accuracy is fundamental for translating computational results into reliable clinical applications.
A dominant trend involves combining thermal ablation with smart drug delivery systems to achieve a synergistic therapeutic effect. A prominent strategy utilizes Thermosensitive Liposomes (TSLs), which are designed to release their chemotherapeutic payload precisely upon reaching a specific temperature threshold triggered by the ablation procedure [16,17]. Computational studies now actively decode how tumor heterogeneities influence this heat-mediated drug delivery process, optimizing the combined therapy for different cancer types, including brain tumors [17]. Beyond chemotherapy, the goal is to stimulate a systemic immune response against the cancer. Research explores how certain ablation modalities, including non-thermal techniques like pulsed electric fields, can induce immunogenic cell death, effectively turning the tumor into an in situ vaccine and enhancing the effects of immunotherapy [29,33].
Substantial efforts are dedicated to engineering novel agents that improve the efficiency and targeting of thermal energy. The use of nanoparticles, such as magnetic or copper oxide particles, remains a core area of investigation, as they concentrate heat at the tumor site, allowing for lower and safer energy application [20,22,27]. Innovation also extends to developing new organic photothermal agents, like specialized dyes, that exhibit enhanced absorption of therapeutic near-infrared light [36,39]. Perhaps the novelest concepts involve biological systems, such as engineered bacteria that act as self-targeting thermal vectors [21], and injectable hydrogels that protect surrounding healthy tissues while potentially serving as a platform for localized drug delivery [35].
Translating complex computational findings into clinical practice is a key focus. This involves developing sophisticated optimization algorithms, including genetic algorithms and active learning methods, to pre-plan critical surgical parameters like ablation power, time, and probe placement for individual patients [19,24]. Furthermore, the clinical pipeline is strengthened by establishing expert consensus on perioperative management [23] and by integrating advanced imaging techniques like PET/CT for improved procedural guidance [38]. This work bridges the gap between theoretical models and the operating room, aiming to standardize and improve patient outcomes.
In conclusion, the current trajectory of thermal oncology is moving far beyond simple tissue destruction. The integration of advanced computational modeling with novel materials and combination strategies represents a push towards intelligent, multi-modal therapies [34]. The future of the field lies in continuing to refine these models with experimental validation, integrating them with real-time adaptive control systems, and further exploring their ability to predict and harness long-term immune responses for a more comprehensive and systemic attack on cancer. The fundamental equation used for modeling heat distribution in biological tissues is shown in tables 1,2.
| Table 1: Pennes bioheat equation. | |||
| Parameter | Symbol | Units | Description |
| Bioheat transfer equation | ρcp∂T/∂t=Ñ(kÑT)+ωbρbcb(T-Tb)+Qabs+ Qmet | ||
| Temperature | T | °K | Tissue temperature |
| Heat capacity | Cp ρ | J·mm-³·°K-¹ | Heat capacity (specific heat × density) |
| Tissue density | ρ | g·mm-³ | Mass density of tissue |
| Specific heat capacity | Cp | J·g-¹·°K-¹ | Specific heat of tissue |
| Thermal conductivity | k | W·mm-¹·°K-¹ | Heat conduction coefficient |
| Blood flow rate | wb | ml·g-¹·min-¹ | Perfusion rate |
| Blood temperature | Tb | °K | Arterial blood temperature |
| Time | t | s | Time variable |
| Heat source | Qabs | W·mm-³ | Absorbed energy |
| Metabolic heat | Qmet | W·mm-³ | Metabolic heat generation |
| Table 2: Boundary conditions. | ||
| Condition Type | Mathematical Expression | Application |
| Cylindrical wall | T = Tb | Temperature equals blood temperature |
| Other surfaces | ∂T/∂n = 0 | Zero heat flux condition |
Parameter Study Design for Tumor Ablation
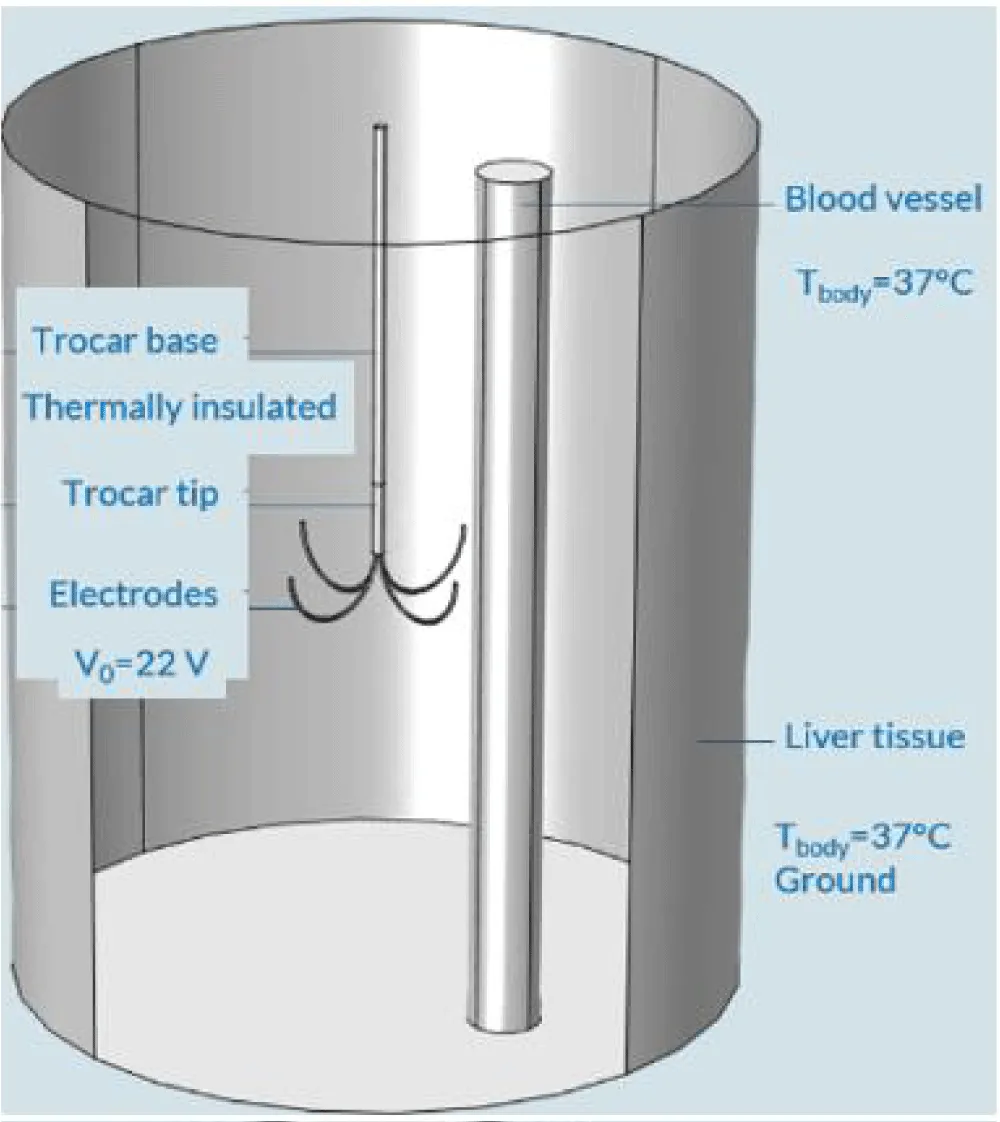
To systematically understand the influence of various factors on the efficacy and safety of tumor ablation, a comprehensive parameter study is essential. Building upon the principles demonstrated in the provided COMSOL Multiphysics model for hepatic tumor ablation, a conceptual parameter study can be designed to explore the sensitivity of ablation outcomes to key physical and operational variables. The primary objective of such a study is to identify the most influential parameters and their optimal ranges for achieving desired therapeutic effects while minimizing collateral damage to healthy tissue. Figure 1 shows the model geometry while the probe is inserted inside liver tissue and a blood vessel is represented near the probe. The four electrode arms are deployed in an area covering the tumor. The tumor itself is not represented, but is located near the center of the cylindrical liver tissue. As well DC current is considered through the probe. The trocar is thermally insulated except near the electrodes. At the electrode boundaries the potential equals 22 V. The external and the blood vessel boundaries are set at the body temperature of 37°C. A ground condition is applied on the outer boundaries.
Key input parameters for variation
The selection of input parameters for variation is crucial and should encompass factors that significantly impact the electric field distribution, heat generation, and heat transfer within the biological tissue. Based on the underlying physics of thermal ablation and insights from the provided model, the following categories of parameters are considered:
Electrical parameters
These parameters directly govern the generation of heat through resistive heating. Varying these can significantly alter the power deposition in the tissue.
- Applied voltage (V0): The potential difference applied across the electrodes is a primary determinant of the electric field strength and, consequently, the rate of Joule heating. Higher voltages generally lead to more intense and rapid heating [5].
- Electrical conductivity (σ) of tissues: The electrical conductivity of the liver tissue, blood, and potentially the tumor itself (if its electrical properties differ from the surrounding healthy tissue) dictates how current flows through the medium. Variations in conductivity can lead to different current pathways and localized heating patterns.
- Electrode material properties: While often assumed constant, the electrical and thermal conductivities of the electrode material can influence heat dissipation from the probe and the efficiency of current delivery.
Thermal parameters: These parameters dictate how heat is distributed, stored, and removed from the ablation zone.
- Blood perfusion rate (wb): Blood flow acts as a critical heat sink, carrying away heat from the heated region. A higher perfusion rate can significantly limit the spread of thermal damage, making it a crucial parameter for controlling ablation size and protecting adjacent healthy tissue [5].
- Initial and boundary temperatures (T0,Tb): The baseline temperature of the tissue and the temperatures maintained at the boundaries (e.g., at the skin surface or near large blood vessels) influence the overall thermal gradient and heat exchange dynamics.
- Thermal conductivity (k) and specific heat (C) of tissues: These intrinsic properties of the tissue determine how efficiently heat is conducted through the material and how much energy is required to raise its temperature. Differences in these properties between tumor and healthy tissue, or variations within the same tissue type, can impact ablation effectiveness.
Geometric parameters: The physical configuration of the ablation system and the biological target are fundamental to the spatial distribution of energy and heat.
- Probe geometry: This includes the dimensions of the ablation probe, such as the radius, height, and the specific design of the electrode arms (e.g., length, spacing, and number of arms). These factors directly influence the electric field distribution and the shape of the heated region [5].
- Tumor size and location: The dimensions of the tumor and its precise location relative to the ablation probe and critical structures (e.g., major blood vessels, vital organs) are paramount. A larger tumor or one located near sensitive areas may require different ablation strategies.
- Blood vessel proximity and size: The distance of the ablation zone from major blood vessels and the diameter of these vessels significantly affect the heat sink effect, influencing the achievable temperature and the extent of the ablated volume [5].
Operational parameters: These are the controllable aspects of the ablation procedure.
- Ablation time: The duration for which the electrical energy is applied is a direct determinant of the total thermal energy delivered to the tissue and, consequently, the volume of tissue reaching necrotic temperatures [5].
- Temperature threshold for necrosis: While primarily a biological constant (typically 45-50°C for irreversible cell damage), understanding the sensitivity of ablation outcomes to slight variations in this threshold can be important for clinical interpretation and personalized treatment planning.
Proposed Study Approach
A conceptual parameter study would involve systematically varying these input parameters and observing their impact on key output metrics. The primary output parameters of interest include the maximum temperature achieved within the target volume, the spatial distribution of temperature, and the volume of necrotic tissue. The study can be structured using the following approach:
- Sensitivity analysis: Quantify the change in output parameters (e.g., necrotic volume) for a given change in an input parameter, while holding other parameters constant. This helps identify the most critical parameters that require precise control or further investigation.
- Parametric sweeps: Conduct simulations across a predefined range of values for one or two specific input parameters. The results can then be visualized to show trends and relationships. For instance, plotting the maximum temperature as a function of applied voltage for different ablation times can reveal optimal operating points.
- Multi-parameter interaction analysis: Investigate how combinations of parameters interact to influence ablation outcomes. This can be achieved through multi-dimensional plots or heatmaps, providing a more holistic understanding of the system’s behavior.
Conceptual Implementation and Simulated Results
For demonstration purposes, we generated synthetic data to represent the outcomes of such a study, focusing on the relationships between applied voltage, ablation time, blood perfusion rate, maximum temperature, and necrotic volume. A simplified model was used where maximum temperature increases with voltage and ablation time, and decreases with perfusion, while necrosis volume is related to the temperature achieved and ablation time. Our simulated results, as depicted in the accompanying figures, demonstrate the expected trends:
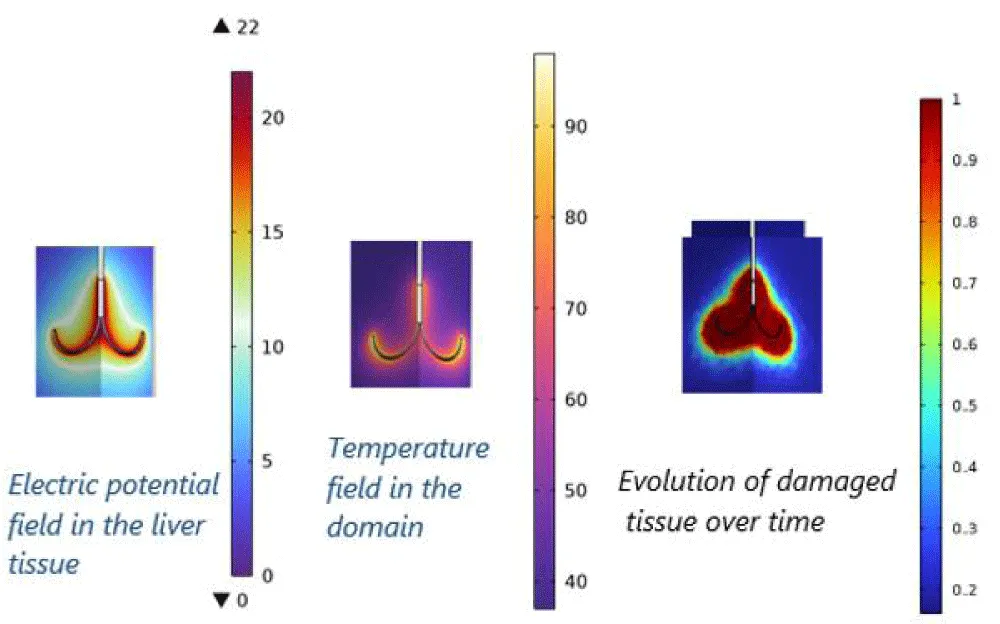
- Temperature, voltage, and damage (Figure 2): An applied electric voltage creates a concentrated electric field near a probe, which drives currents that resistively heat the tissue, ultimately creating a predictable zone of thermal destruction designed to kill tumor cells.
To elucidate the underlying physical mechanisms of Radiofrequency Ablation (RFA), a coupled electro-thermal simulation was performed. The results, depicted in figure 2, illustrate the interplay between the applied electric field and the resultant temperature distribution that leads to tissue necrosis. Figure 2 shows the computed electric potential field within the hepatic tissue. The closely spaced isopotential contours in the immediate vicinity of the electrode tip indicate a region of high electric field intensity, governed by Laplace's equation. This strong field drives a high-density electrical current radially outward from the active electrode.
The consequent thermal field, arising from the dissipation of this electrical energy, is presented in figure 2. The primary heating mechanism is Joule (resistive) heating, with the power density source term proportional to the square of the local current density. This explains the maximal heat generation adjacent to the electrode. The resulting temperature distribution, obtained from solving the Pennes bioheat equation, shows a steep gradient. The central region, exceeding the critical threshold of approximately 60°C, defines the theoretical ablation zone where irreversible protein coagulation and instantaneous cell death occur [40-41]. The outer, moderately heated regions (approximately 45-60°C) may achieve necrosis only with sustained application, highlighting the importance of treatment duration. The asymmetry in the temperature field could be attributed to the heat-sinking effect of a nearby blood vessel, a common factor influencing ablation geometry in clinical settings. In summary, the simulation demonstrates how the focused delivery of RF energy creates a predictable thermal lesion, validating the computational model as a tool for predicting treatment outcomes in hepatic tumor ablation.
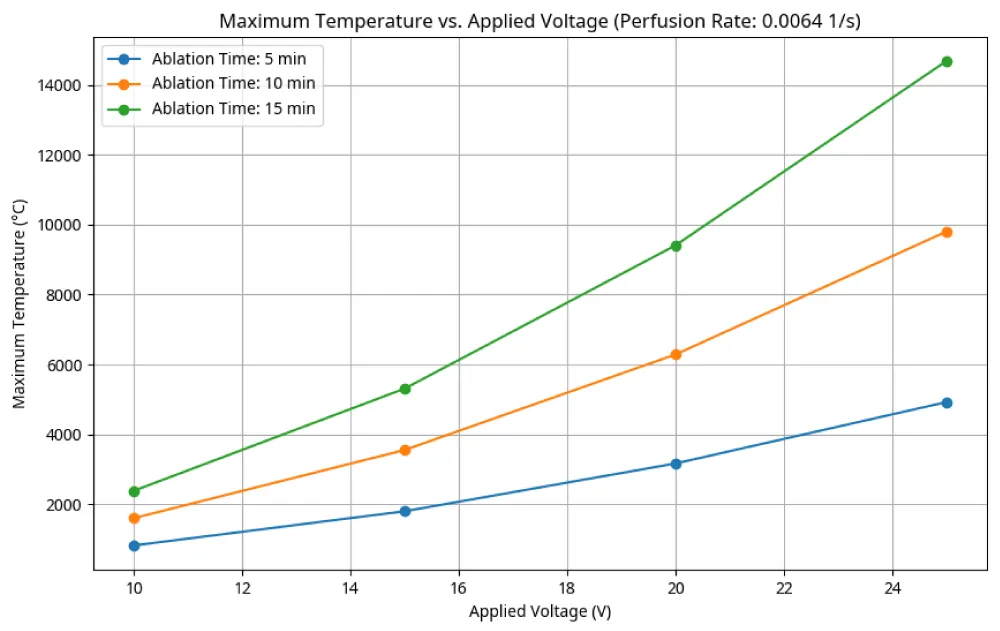
- Maximum temperature vs., applied voltage (Figure 3): As the applied voltage increases, the maximum temperature achieved within the tissue also rises. This relationship is further modulated by the ablation time, with longer durations leading to higher temperatures for a given voltage. This highlights the direct control that electrical parameters exert over thermal energy deposition.
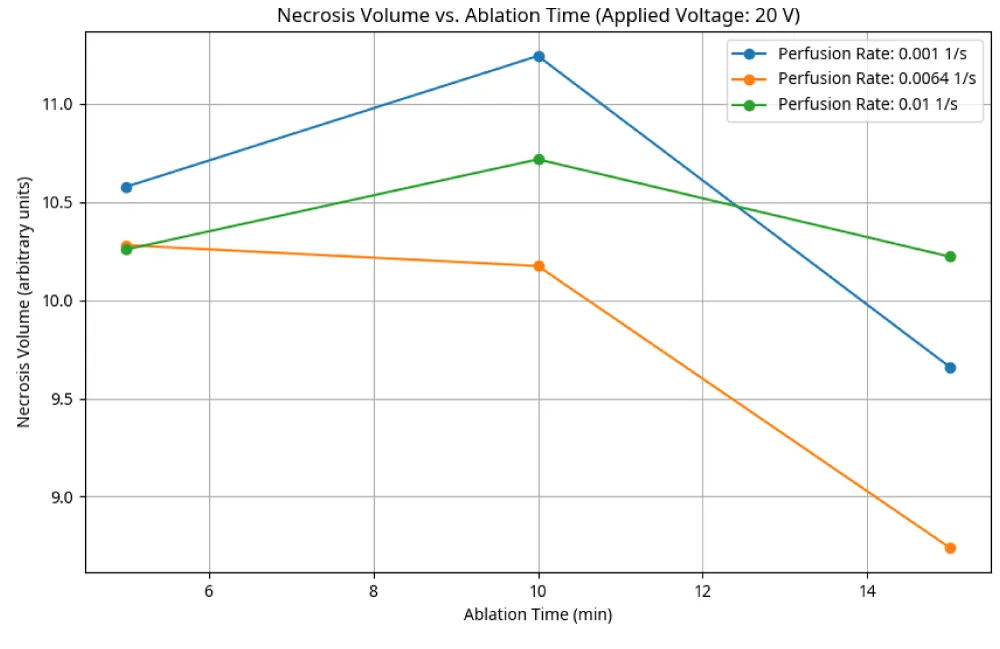
- Necrosis volume vs., ablation time (Figure 4): The volume of necrotic tissue generally increases with ablation time, as prolonged heating allows more tissue to reach and sustain lethal temperatures. The blood perfusion rate plays a crucial role here; higher perfusion rates tend to reduce the necrotic volume for a given ablation time, emphasizing the importance of managing blood flow during ablation procedures.
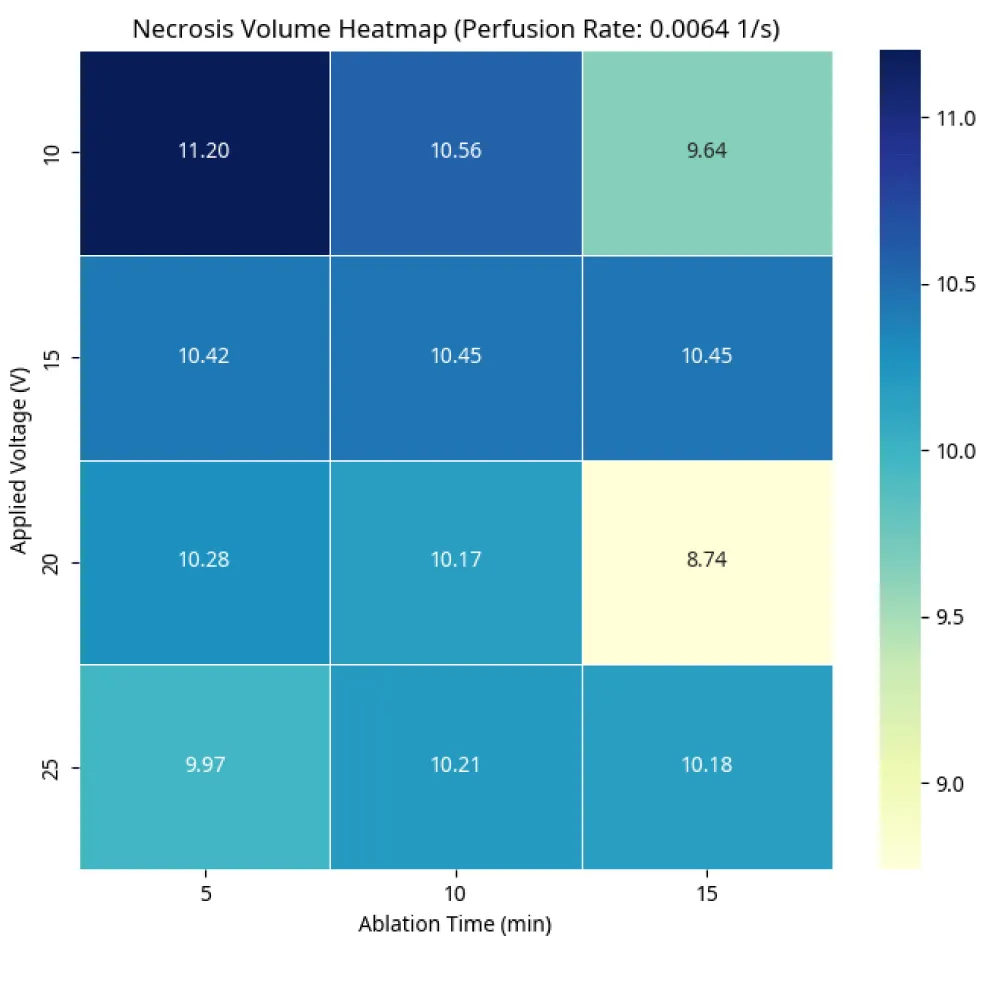
- Necrosis volume heatmap (Figure 5): A heatmap illustrating the combined effect of applied voltage and ablation time on necrosis volume provides a comprehensive overview. This visualization allows for the identification of optimal parameter combinations to achieve a desired necrotic volume, considering the trade-offs between voltage intensity and treatment duration.
These simulated results, while based on a simplified model, effectively illustrate the power of parameter studies in understanding the complex interplay of variables in thermal ablation. Such analyses provide critical insights for optimizing treatment protocols, predicting patient-specific responses, and enhancing the overall efficacy and safety of tumor ablation therapies
Conclusion and Future Directions
Computational modeling stands as a pivotal tool in the ongoing battle against brain tumors and neurological disorders, particularly in the realm of ablation therapies. As demonstrated through the analysis of a multiphysics model for tumor ablation and a conceptual parameter study, these models offer an unparalleled ability to dissect complex biophysical interactions, predict treatment outcomes, and optimize therapeutic strategies. The systematic variation of parameters such as applied voltage, ablation time, and blood perfusion rates, even in a simplified context, reveals critical insights into their influence on key ablation metrics like maximum temperature and necrotic volume. This understanding is fundamental for refining existing clinical protocols and developing more effective and safer interventions.
Looking ahead, the field of computational neuro-oncology is poised for significant advancements. Future directions will likely focus on several key areas:
- Enhanced model fidelity and personalization: Moving beyond simplified models, there is a growing need for more sophisticated, patient-specific models that integrate diverse clinical data, including advanced imaging (e.g., fMRI, DTI), genomic, and proteomic information. This will enable highly personalized treatment planning, accounting for individual tumor heterogeneity and patient physiology. The development of robust inverse problem solutions to calibrate model parameters from clinical data will be crucial.
- Integration with artificial intelligence and machine learning: AI and ML algorithms can significantly enhance computational modeling by accelerating simulations, optimizing parameter spaces, and identifying complex patterns in large datasets. These technologies can be used for predictive analytics, identifying patients most likely to respond to specific therapies, and even for real-time treatment guidance during procedures.
- Multi-scale and multi-physics coupling: Future models will increasingly integrate phenomena across multiple scales, from molecular and cellular interactions to tissue and organ-level responses. This includes more detailed coupling of electrical, thermal, mechanical, and biological processes, such as immune responses to ablation and long-term tissue remodeling.
- Validation and clinical translation: A critical step for the broader adoption of computational models in clinical practice is rigorous validation against experimental and clinical data. This requires collaborative efforts between computational scientists, clinicians, and experimental biologists to generate high-quality datasets for model calibration and validation. Bridging the gap between in silico predictions and real-world patient outcomes will be paramount.
- Development of user-friendly tools: To facilitate wider adoption by clinicians and researchers, there is a need for more user-friendly computational platforms and tools that abstract away the underlying mathematical and numerical complexities, allowing for intuitive exploration of treatment scenarios.
In conclusion, computational modeling provides a powerful lens through which to view and manipulate the intricate processes involved in tumor and brain disorders. By continuing to push the boundaries of these simulations, we can unlock new avenues for diagnosis, treatment, and ultimately, improve the lives of patients facing these challenging conditions.
References
- Madhusoodanan S, Ting MB, Farah T, Ugur U. Psychiatric aspects of brain tumors: A review. World J Psychiatry. 2015 Sep 22;5(3):273-85. doi: 10.5498/wjp.v5.i3.273. PMID: 26425442; PMCID: PMC4582304.
- Moroşan GC, Moroşan AC, Ionescu C, Sava A. Neuropsychiatric symptoms as early indicators of brain tumors. Arch Clin Cases. 2024 Dec 20;11(4):120-126. doi: 10.22551/2024.45.1104.10302. PMID: 39712552; PMCID: PMC11661549.
- Aldape K, Brindle KM, Chesler L, Chopra R, Gajjar A, Gilbert MR, Gottardo N, Gutmann DH, Hargrave D, Holland EC, Jones DTW, Joyce JA, Kearns P, Kieran MW, Mellinghoff IK, Merchant M, Pfister SM, Pollard SM, Ramaswamy V, Rich JN, Robinson GW, Rowitch DH, Sampson JH, Taylor MD, Workman P, Gilbertson RJ. Challenges to curing primary brain tumours. Nat Rev Clin Oncol. 2019 Aug;16(8):509-520. doi: 10.1038/s41571-019-0177-5. PMID: 30733593; PMCID: PMC6650350.
- Antonica F, Aiello G, Soldano A, Abballe L, Miele E, Tiberi L. Modeling Brain Tumors: A Perspective Overview of in vivo and Organoid Models. Front Mol Neurosci. 2022 May 30;15:818696. doi: 10.3389/fnmol.2022.818696. PMID: 35706426; PMCID: PMC9190727.
- Hepatic tumor ablation. COMSOL Multiphysics Model. 2023.
- McFaline-Figueroa JR, Lee EQ. Brain Tumors. Am J Med. 2018 Aug;131(8):874-882. doi: 10.1016/j.amjmed.2017.12.039. Epub 2018 Jan 31. PMID: 29371158.
- Juffer AH, Marin U, Niemitalo O, Koivukangas J. Computer Modeling of Brain Tumor Growth. Mini Reviews in Medicinal Chemistry. 2008;8(14):1494-1506. doi: 10.2174/138955708786786471.
- Hormuth DA 2nd, Al Feghali KA, Elliott AM, Yankeelov TE, Chung C. Image-based personalization of computational models for predicting response of high-grade glioma to chemoradiation. Sci Rep. 2021 Apr 19;11(1):8520. doi: 10.1038/s41598-021-87887-4. PMID: 33875739; PMCID: PMC8055874.
- Mongeon B, Hébert-Doutreloux J, Surendran A, Karimi E, Fiset B, Quail DF, Walsh LA, Jenner AL, Craig M. Spatial computational modelling illuminates the role of the tumour microenvironment for treating glioblastoma with immunotherapies. NPJ Syst Biol Appl. 2024 Aug 18;10(1):91. doi: 10.1038/s41540-024-00419-4. PMID: 39155294; PMCID: PMC11330976.
- Miranda A, Cova T, Sousa J, Vitorino C, Pais A. Computational modeling in glioblastoma: from the prediction of blood-brain barrier permeability to the simulation of tumor behavior. Future Med Chem. 2018 Jan;10(1):121-131. doi: 10.4155/fmc-2017-0128. Epub 2017 Dec 13. PMID: 29235374.
- Wang Z, Deisboeck TS. Computational modeling of brain tumors: discrete, continuum or hybrid? Scientific Modeling and Simulation SMNS. 2008;15:381-393. doi: 10.1007/s10820-008-9094-0.
- Coluccia D, Fandino J, Schwyzer L, O'Gorman R, Remonda L, Anon J, Martin E, Werner B. First noninvasive thermal ablation of a brain tumor with MR-guided focused ultrasound. J Ther Ultrasound. 2014 Oct 16;2:17. doi: 10.1186/2050-5736-2-17. PMID: 25671132; PMCID: PMC4322509.
- Pichaivel M, Anbumani G, Theivendren P, Gopal M. An overview of brain tumor. In: Brain Tumors. IntechOpen. 2022. doi: 10.5772/intechopen.100806.
- Aerts H, Schirner M, Jeurissen B, Van Roost D, Achten E, Ritter P, Marinazzo D. Modeling Brain Dynamics in Brain Tumor Patients Using the Virtual Brain. eNeuro. 2018 Jun 4;5(3):ENEURO.0083-18.2018. doi: 10.1523/ENEURO.0083-18.2018. PMID: 29911173; PMCID: PMC6001263.
- Andreozzi A, Iasiello M, Vanoli GP. Laser ablation for prostate tumors: Analysis of different bioheat transfer models. International Journal of Thermal Sciences. 2025;211:107456. doi: 10.1016/j.ijthermalsci.2025.110026.
- Bhandari A, Singh A, Zhan W. Decoding the effect of different tumor heterogeneities on thermosensitive liposome drug delivery during radiofrequency ablation: A novel heat and mass transfer perspective. International Communications in Heat and Mass Transfer. 2024;153:107456. doi: 10.1016/j.icheatmasstransfer.2024.107390.
- Bhandari A, Tripathy KC, Germaneau A. Unveiling human brain tumor response to tumor treating fields mediated thermosensitive liposome drug delivery: A heat and mass transfer optimization. International Journal of Heat and Mass Transfer. 2025;203:107456. doi: 10.1016/j.ijheatmasstransfer.2025.127352.
- Preechaphonkul W, Mongkol V, Rattanadecho P. Numerical analysis of heat transfer and tissue deformation in liver cancer during microwave ablation: A comparison of bioheat and porous media models. Thermal Science and Engineering Progress. 2025;47:102456. doi: 10.1016/j.tsep.2025.103739.
- Yuan C, Yan X, Yue K, Wang H, Zhang X, Yan D, An C. Optimization of surgical parameters for liver tumor microwave ablation assisted by hydrodissection: Solution space features and active learning approach. Comput Methods Programs Biomed. 2025 Oct;270:108967. doi: 10.1016/j.cmpb.2025.108967. Epub 2025 Jul 14. PMID: 40712370.
- Fakhradini SS, Mosharaf-Dehkordi M, Ahmadikia H. Enhancing liver cancer treatment: Exploring the frequency effects of magnetic nanoparticles for heat-based tumor therapy with microwaves. International Journal of Thermal Sciences. 2024;209:107456. doi: 10.1016/j.ijthermalsci.2024.109154.
- Kaur T, Devi N, Sharma D. E. coli as a Smart Thermo-Vector for Combating Solid Tumors: A Synergistic Heat-Induced Cancer Therapy Approach. Bioconjug Chem. 2025 Apr 16;36(4):867-880. doi: 10.1021/acs.bioconjchem.5c00102. Epub 2025 Mar 19. PMID: 40105793.
- Imanlou S, Vafai K. Analysis of magnetothermal hyperthermia on tumor ablation and Parkinson’s therapy. International Journal of Heat and Mass Transfer. 2026;210:107456.
- He J, Shen M, Ye X, Ren X, Chen K, Zhang J, Fan W, Wang Z, Xu Y. Expert consensus on perioperative management for liver tumors treated with co-ablation system therapy. Asia Pac J Oncol Nurs. 2024 Sep 10;11(11):100591. doi: 10.1016/j.apjon.2024.100591. PMID: 39582552; PMCID: PMC11584913.
- Adabbo G, Andreozzi A, Iasiello M, Napoli G, Vanoli GP. Multi-objective optimization framework to plan laser ablation procedure for prostate tumors through a genetic algorithm. Comput Methods Programs Biomed. 2025 Jul;267:108827. doi: 10.1016/j.cmpb.2025.108827. Epub 2025 May 2. PMID: 40344998.
- Wessapan T, Rattanadecho P. Flow and heat transfer through a porous tumor during high-intensity focused ultrasound. International Journal of Heat and Mass Transfer. 2023;220:115789.
- Horvat ID, Iljaž J. Numerical modeling of non-Fourier bioheat transfer in multilayer biological tissue using BEM to simulate dynamic thermography in skin tumor diagnostics. Engineering Analysis with Boundary Elements. 2025;170:107456. doi: 10.1016/j.enganabound.2025.106408.
- Suleman M, Majeed U, Kolsi L. Computational modeling of thermal therapy for porous osteosarcoma knee bone tumors using magnetized copper oxide nanoparticles. Case Studies in Thermal Engineering. 2025;47:102456. doi: 10.1016/j.csite.2025.106775.
- Wessapan T, Keangin P, Somsuk N. Comparative analysis of heat transfer dynamics in high-intensity focused ultrasound and microwave ablation for cancer treatment. International Journal of Thermofluids. 2025;25:100345. doi: 10.1016/j.ijft.2025.101090.
- Gao Y, Huang Z, Chen W, Ma Y, Li X, Wang Y, Wan Y, Liu Y, Yang Y, Li MD, Lee CS, Li S. Stable intermolecular charge-transfer nanocrystals as efficient immunogenic cell death adjuvants for photoimmunotherapy against trophoblastic and recurrent tumors. Biomaterials. 2025 Dec;323:123436. doi: 10.1016/j.biomaterials.2025.123436. Epub 2025 May 27. PMID: 40446712.
- Junaid M, Mukaddes AMM, Mahmud-Or-Rashid M. Physical activities aid in tumor prevention: A finite element study of bio-heat transfer in healthy and malignant breast tissues. Heliyon. 2024 Jul 15;10(14):e34650. doi: 10.1016/j.heliyon.2024.e34650. PMID: 39114025; PMCID: PMC11305304.
- Jiang Q, Ren F, Tang H. Multiphysics simulation of tumor ablation in magnetic hyperthermia treatment. International Journal of Heat and Mass Transfer. 2025;210:107456. doi: 10.1016/j.ijheatmasstransfer.2025.126982.
- Suleman M, Riaz S. Computational modeling of poroelastic brain tumor therapy during heat transfer carrying temperature-dependent blood perfusion. Med Eng Phys. 2022 May;103:103792. doi: 10.1016/j.medengphy.2022.103792. Epub 2022 Mar 24. PMID: 35500993.
- Arciga BM, Walters DM, Kimchi ET, Staveley-O'Carroll KF, Li G, Teixeiro E, Rachagani S, Kaifi JT. Pulsed electric field ablation as a candidate to enhance the anti-tumor immune response to immune checkpoint inhibitors. Cancer Lett. 2025 Jan 28;609:217361. doi: 10.1016/j.canlet.2024.217361. Epub 2024 Nov 26. PMID: 39608443; PMCID: PMC11625606.
- Gu C, Zhang J, Qi J. Advances and applications of hyperthermia in tumor therapy: Mechanisms, techniques, and clinical integration. International Communications in Heat and Mass Transfer. 2025;154:107456. doi: 10.1016/j.icheatmasstransfer.2025.108895.
- Huang L, Qi E, Liu Z, Zhang Q, Wang S, Zheng S, Xie W, Li G, Chen X, Sun H, Liu F, Wang X. Beyond thermal protection: Injectable self-healing chitosan hydrogel in liver tumor thermal ablation. Int J Biol Macromol. 2025 Jun;313:144187. doi: 10.1016/j.ijbiomac.2025.144187. Epub 2025 May 14. PMID: 40379161.
- Nishimura K, Kato M, Fukui T, Miura K, Tsuda M, Okada S, Fukushima T, Nakamura H. BODIPY-Based Photothermal Agent Incorporating Azulene for Enhanced NIR Absorption and Tumor Ablation. Mol Pharm. 2025 May 5;22(5):2660-2670. doi: 10.1021/acs.molpharmaceut.5c00071. Epub 2025 Apr 1. PMID: 40167019.
- Ismaeel AM, Kamel RS, Hady FM. Numerical simulation for a Maxwell nanofluid with convective heat transfer over a horizontal cylindrical vessel surrounded by hot tissue. Applied Thermal Engineering. 2025;269:123789. doi: 10.1016/j.applthermaleng.2025.127173.
- Chlorogiannis DD, Sofocleous CT, Jiang L, Sotirchos VS. PET/CT-guided Tumor Ablation: Technical Insights and Clinical Applications. PET Clin. 2025 Jul;20(3):281-289. doi: 10.1016/j.cpet.2025.03.003. Epub 2025 Apr 28. PMID: 40300984.
- Li M, Ye Z, Peng X. Bidirectional pH-responsive O2@perfluorocarbon@cyanine dyes for cascade photothermal therapy and oxygen-releasing enhanced photodynamic tumor treatment. Chemical Engineering Journal. 2025;499:155789. doi: 10.1016/j.cej.2025.164690.
- Goldberg SN, Charboneau JW, Dodd GD 3rd, Dupuy DE, Gervais DA, Gillams AR, Kane RA, Lee FT Jr, Livraghi T, McGahan JP, Rhim H, Silverman SG, Solbiati L, Vogl TJ, Wood BJ; International Working Group on Image-Guided Tumor Ablation. Image-guided tumor ablation: proposal for standardization of terms and reporting criteria. Radiology. 2003 Aug;228(2):335-45. doi: 10.1148/radiol.2282021787. PMID: 12893895.
- Haemmerich D, Laeseke PF. Thermal tumour ablation: devices, clinical applications and future directions. Int J Hyperthermia. 2005 Dec;21(8):755-60. doi: 10.1080/02656730500226423. PMID: 16338858.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.