2025 October 13;6(10):1432-1439. doi: 10.37871/jbres2199.
Safety Profile of COVID-19 Vaccines: Retrospective Analysis of Short, Medium, and Long-Term Side Effects: The Military Hospital Experience
Antonio Sabba1*, Giancarlo Pontoni2, Maria Santangelo1, Nadir Rachedi1, Maurizio D Ercole1, Davide Lardo1, Angelo Raffaele Chiarelli1 and Vincenzo Campagna1
2Foligno Army selection centre, Italy
- COVID-19
- Smallpox
- Vaccination
- Novavax
- Military polyclinic
Abstract
Background: Vaccination is a key strategy to reduce infectious disease incidence and COVID-19 severity. Monitoring vaccine safety is essential to identify Adverse Reactions (ARs) and ensure public confidence.
Objective: To evaluate adverse reactions following COVID-19 vaccination at the Military Polyclinic of Rome “Celio” between 2020 and 2023, analyzing variables such as sex, age, vaccine type, and dose number, and comparing results with national (AIFA) and European (EMA) data.
Methods: A retrospective observational study was conducted on 41,276 doses administered across five vaccine types (Pfizer, Moderna, AstraZeneca, Johnson & Johnson, Novavax). ARs were classified by severity and system involved. Statistical analyses, including Chi-square and Fisher’s Exact Test, assessed correlations by age, sex, dose, and vaccine type.
Results: Total ARs: 41 (0.1% of doses); 92.7% non-severe, 7.3% severe.
- Most ARs involved musculoskeletal (29.2%) and immune (26.8%) systems.
- Female subjects experienced more ARs (66%) than males (34%).
- ARs were most frequent after the first dose, decreasing with subsequent doses.
- By vaccine type, AstraZeneca showed the highest relative rate of ARs (0.41%), followed by Moderna (0.09%) and Pfizer (0.05%).
- Severe ARs were rare, predominantly in the 30–49 age group.
- Local reporting rates (0.1%) aligned closely with national data (0.097%).
Conclusions: COVID-19 vaccines administered at the Military Polyclinic demonstrated a high safety profile. Severe adverse events were rare, nonsevere reactions predominated, and findings were consistent with national and European surveillance data. The benefits of vaccination in preventing severe disease, hospitalization, and death significantly outweigh the risks.
Introduction
Vaccinations are among the most effective strategies to reduce infectious disease incidence and prevent millions of deaths. Advances in science and technology have shifted the medical focus from treatment to prevention, enabling the management and control of global epidemics and pandemics, including smallpox and influenza. The COVID-19 pandemic posed an unprecedented global challenge, requiring rapid interventions. Vaccination emerged as a key tool to reduce transmission, severity, hospitalizations, and deaths, aiding the return to normalcy.
Purpose of the Study
This study aimed to analyse adverse reactions reported in Italy between 2020 and 2023 following the administration of authorized COVID-19 vaccines. Variables examined included sex, age, vaccine type, and dose number. Results were compared with national (AIFA) and European (EMA) pharmacovigilance data to identify possible correlations and assess consistency with broader trends.
Materials and Methods
A retrospective observational study was conducted on data collected from January 1, 2021, to December 31, 2023, at the Rome Military Hospital.
The study presented in this paper was approved by the Direction of the Military Polyclinic of Rome "Celio" and the Italian Army Health Command. The study was a retrospective analysis of data related to COVID-19 vaccine administrations considering the time period between 2020 and 2023, in which the doses administered were distributed among five different types of vaccines: Pfizer (PF); AstraZeneca (AZ); Moderna (MO); Johnson&Johnson (JJ); Novavax (NVX).
The data presented here come from the National Pharmacovigilance Network (RNF), the Italian system dedicated to reporting and monitoring adverse reactions to drugs and vaccines, considering:
- Total number of doses administered for each vaccine
- Reports of adverse reactions, classified by severity and type
- Demographic distribution of vaccinated subjects (sex, age groups)
- Type of vaccine (PF; AZ; MO; JJ; NVX)
- Number of doses administered for primary cycle and booster doses Study Sample: 41,280 vaccine doses administered, divided among:
- Pfizer-BioNTech (Comirnaty): 57.5% of administrations
- Moderna (Spikevax): 33.4%
- AstraZeneca (Vaxzevria): 8.2%
- Johnson & Johnson (Jcovden): 0.8%Novavax (Nuvaxovid): 0.1%
Demographic Distribution
The sample analyzed in this study was heterogeneous based on sex and age, which was divided into 8 different age groups (18-29; 30-39; 40-49; 5059;60-69; 70-79; 80-89; >90).
Statistical Analysis
The statistical analysis was performed using STATA Software version 14.2, and evaluating all available data, the following analyses were conducted:
Descriptive analysis
Classification of Adverse Reactions (ARs) by type (non-severe vs. severe) and by involved organ system (musculoskeletal, immune, gastrointestinal, etc.) Calculation of relative and absolute frequencies by sex, age, vaccine type, and administered dose Analysis of ARs distribution among vaccines (Pfizer vs. Moderna, etc.) and among administered doses (first dose, second dose, etc.)
Inferential statistics
The association between the main variables collected was measured. To compare frequencies, the Chi-square test or Fisher's Exact Test was used, depending on applicability, and the level of statistical significance was set at a p-value < 0.05.
Statistical Evaluations: The results obtained from the analysis of this local sample were then compared with COVID-19 vaccine surveillance data published by AIFA in its Surveillance Report on COVID-19 vaccines (period 27/12/2020 - 26/12/2022, Report number 14) and with vaccine safety information monitored by EMA through the EudraVigilance system.
The comparison focused on the overall frequency of reports, severity, distribution by sex, dose, and type of adverse reaction, in order to highlight the alignment of local data with large-scale observations.
Results
During the time span to which the collected data refer (2020-2023), a total of 41.276 doses of COVID-19 vaccine were administered at the Military Polyclinic of Rome "Celio".
The distribution of administered doses based on established age groups is visible in Table 1, where it can be noted that the highest number of doses, 11,750, of which 5,741 were male patients and 6,009 female patients, was administered in the 50-59 age group; followed by the 40-49 age group with a total of 10,825 doses, of which 5,238 were administered to male patients and 5,587 to female patients (Table 1).
| Table 1: Distribution of COVID-19 vaccine doses administered by age group and sex at the military polyclinic of Rome “Celio” (2020-2023). | ||||||||
| Type \ Age | 18-29 | 30-39 | 40-49 | 50-59 | 60-69 | 70-79 | 80-89 | >90 |
| Total by age | 1235 | 4178 | 10825 | 11750 | 8852 | 2850 | 1570 | 20 |
| Doses to male patients | 538 | 1751 | 5238 | 5741 | 4573 | 1221 | 326 | 4 |
| Doses to female patients | 697 | 2427 | 5587 | 6009 | 4279 | 1629 | 1244 | 16 |
There was a certain uniformity in the distribution of doses based on sex, which was 47% in male patients and 53% in female patients.
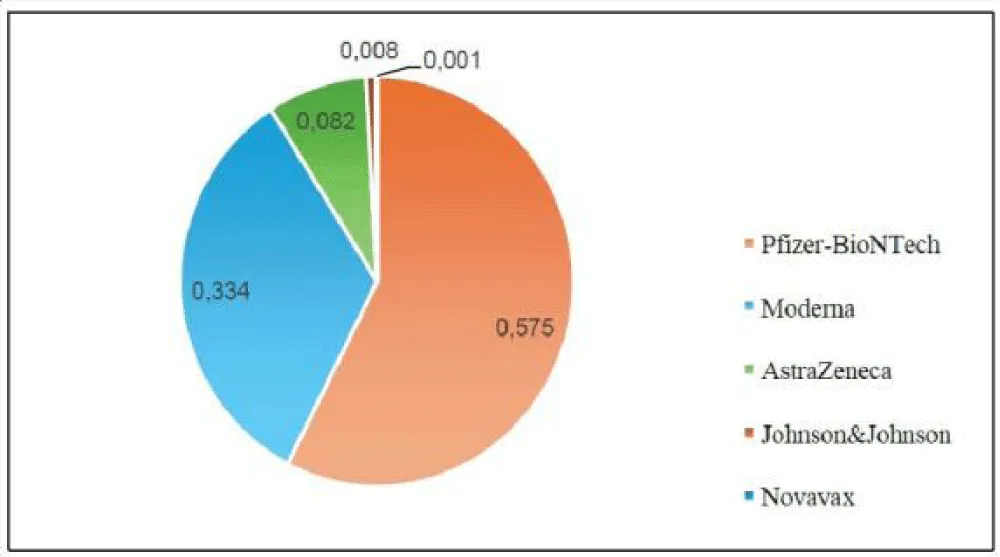
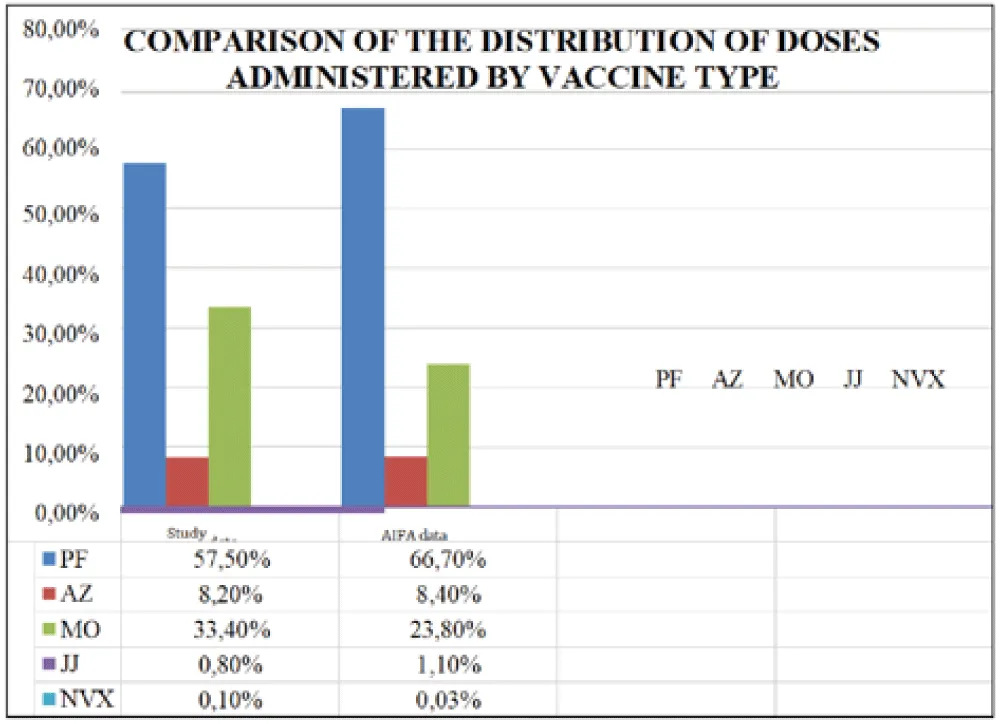
The distribution of doses by vaccine type, in numerical terms, was as follows (Figure 1):
- Pfizer (PF): 23,739, equal to 57.5% of administered doses
- Moderna (MO): 13,780, equal to 33.4% of administered doses
- AstraZeneca (AZ): 3,385, equal to 8.2% of administered doses
- Johnson & Johnson (JJ): 330, equal to 0.8% of administered doses
- Novavax (NVX): 41, equal to 0.10% of administered doses
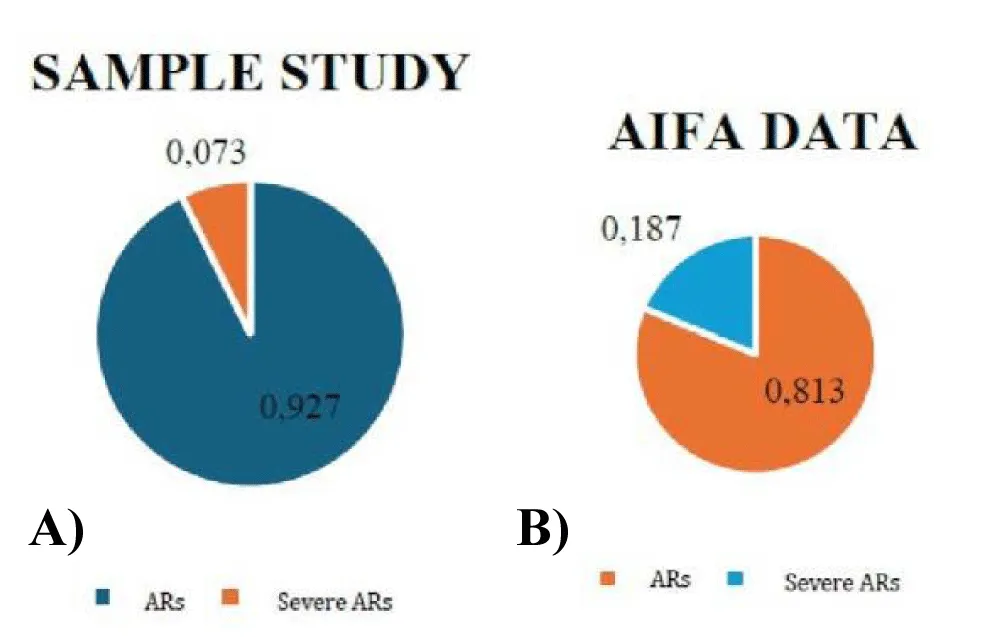
- Regarding ARs, out of 41,276 administered doses, a total of 41 adverse reactions were recorded, corresponding to a reporting rate of 0.1% of the examined sample. Of these 41 adverse reactions, 3 (7.3%) were severe and 38 (92.7%) nonsevere.
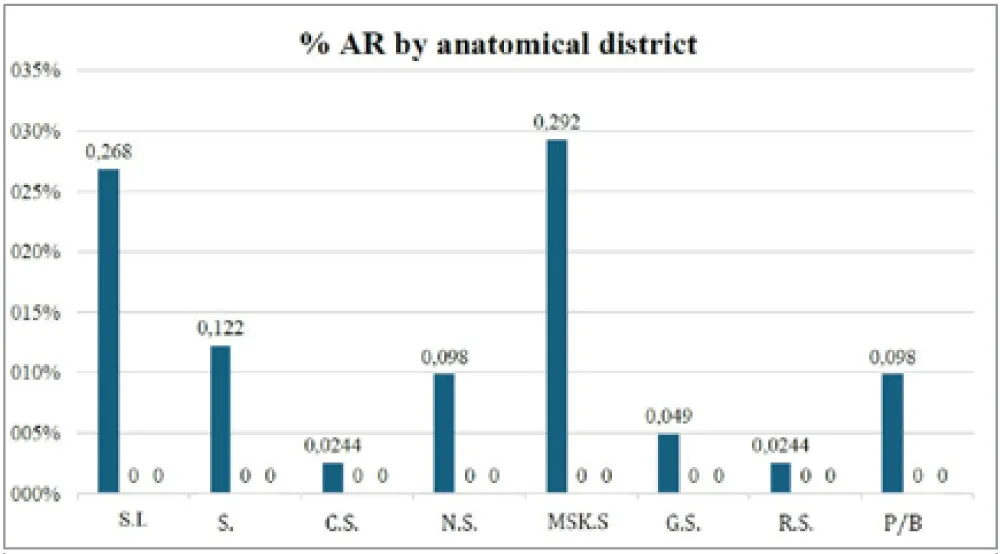
- The ARs recorded in the sample examined mainly affected the districts and systems reported (Table 2), where it was possible to highlight a higher number affecting the musculoskeletal system (12 ARs, 29.2%) followed by reactions affecting the immune system (11 ARs, 26.8%) (Figure 2).
| Table 2: Main demographic and clinical variables of subjects who experienced an adverse reaction. | |||||
| Demographic Variables | n (number) | % (percentage) | Clinical Variables | n (number) | % (percentage) |
| Age Group (years) | Adverse Reaction Type | ||||
| 18-29 | 6 | 14.6 | Severe | 3 | 7.3 |
| 30-39 | 9 | 22 | Non-severe | 38 | 92.7 |
| 40-49 | 7 | 17.1 | |||
| 50-59 | 8 | 19.5 | Affected System | ||
| 60-69 | 5 | 12.2 | Immune System | 11 | 26.8 |
| 70-79 | 2 | 4.9 | Skin and Appendages | 5 | 12.2 |
| 80-89 | 2 | 4.9 | Cardiovascular System | 1 | 2.44 |
| >90 | 2 | 4.9 | Nervous System | 4 | 9.8 |
| Musculoskeletal System | 12 | 29.2 | |||
| Age | Gastrointestinal System | 2 | 4.9 | ||
| Under 50 | 22 | 53.7 | Respiratory System | 1 | 2.44 |
| Over 50 | 19 | 46.3 | Psychological/Behavioral 4 | 9.8 | |
| Sex | Dose Number | ||||
| Male | 14 | 34.1 | I | 24 | 58.5 |
| Female | 27 | 65.9 | II | 12 | 29.3 |
| III | 4 | 9.8 | |||
| IV | 1 | 2.4 | |||
| Vaccine Name | |||||
| Pfizer | |||||
| 12 | 29.3 | ||||
| AstraZeneca | 14 | 34.2 | |||
| Moderna | 13 | 31.7 | |||
| J&J | 1 | 2.4 | |||
| Novavax | 1 | 2.4 | |||
| *Percentages may not total 100 due to rounding. | |||||
Regarding the frequency distribution of ARs based on sex and age, distinguishing severe from non-severe Ars (Tab.3), the highest number of ARs occurred in the female population included in the study with a total of 27 ARs (66%), of which 2 were severe, compared to 14 ARs (34%) that affected male patients, of which 1 was severe. Furthermore, severe reactions were recorded in the 30-39 age group, of which 1 male and 1 female, and another severe reaction occurred in a female patient belonging to the 40-49 age group.
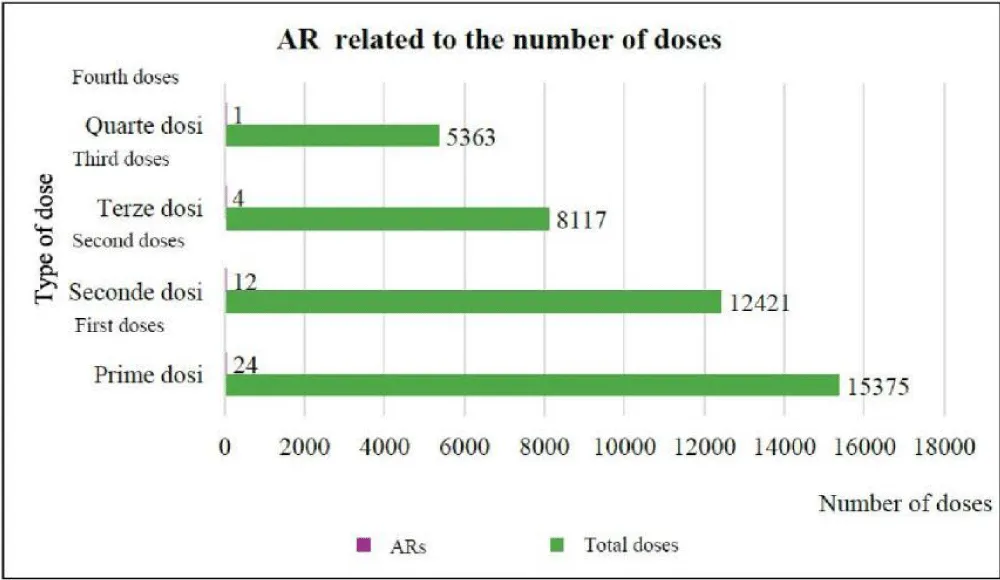
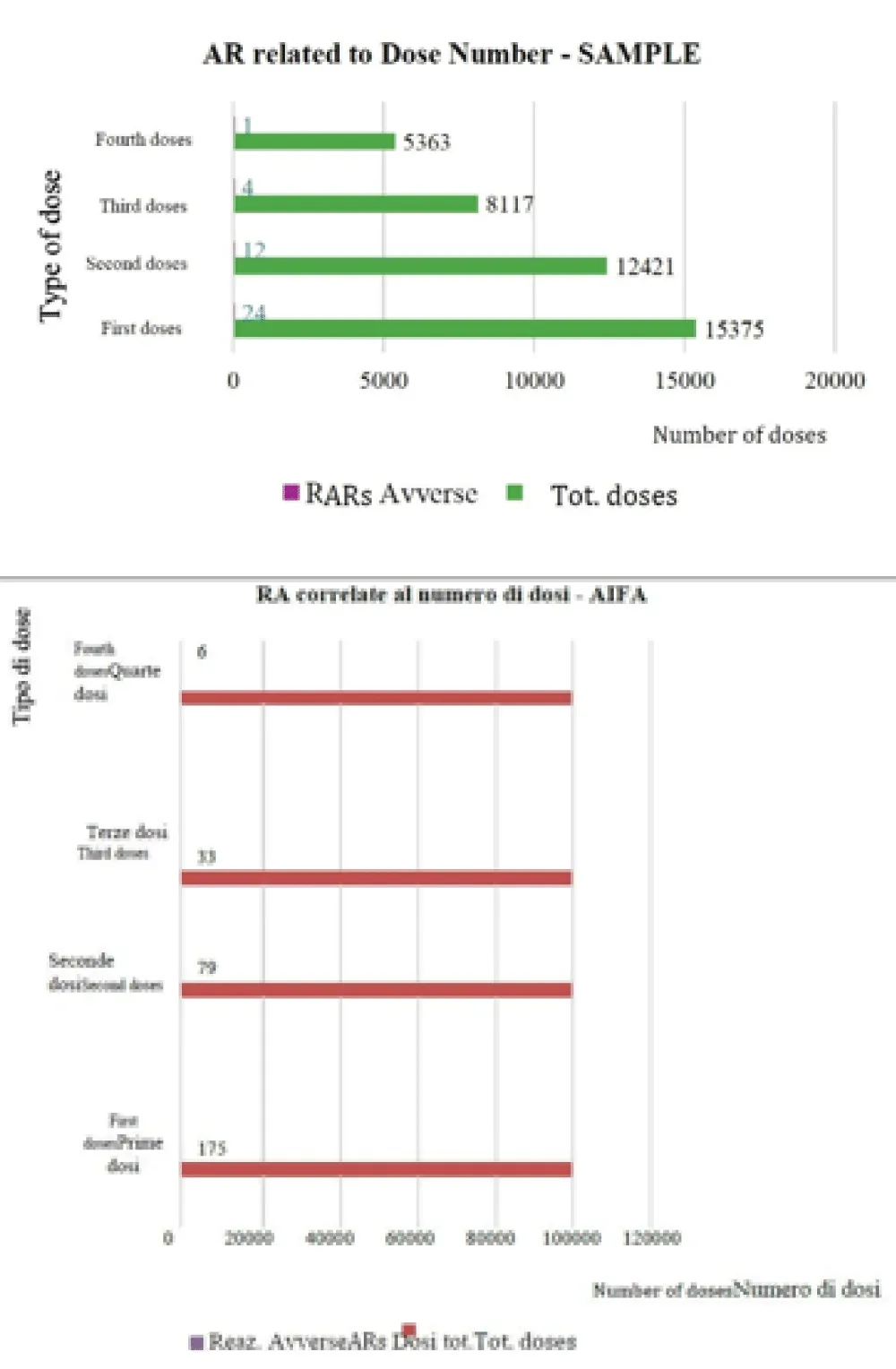
Analyzing the number of vaccinations divided by the number of doses (Figure 3) and correlating them to the reported ARs, we see that a total of 15,375 first doses were recorded, and of these, only in 24 cases (0.16%) did an AR occur; 12,421 were second doses with 12 ARs (0.10%); 8,117 were third doses with 4 ARs (0.05%); and 5,363 were fourth doses with 1 AR (0.02%).
Regarding the correlation of ARs to the type of vaccine administered (Table 3), the highest number occurred with the AstraZeneca vaccine (14, 34.2%) followed by Moderna (13, 31.7%) and Pfizer (12, 29.3%) vaccines. The reported percentages refer to the total number of ARs; if instead we analyze these data by comparing the number of ARs to the number of doses administered by vaccine type, we obtain:
| Table 3: Adverse Reactions divided by age group and sex. | ||||||||
| Type \ Age | 18-29 | 30-39 | 40-49 | 50-59 | 60-69 | 70-79 | 80-89 | >90 |
| Male ARs | 1 | 1 | 2 | 4 | 2 | 1 | 1 | 1 |
| Severe Male ARs | 1 | |||||||
| Female ARs | 5 | 6 | 4 | 4 | 3 | 1 | 1 | 1 |
| Severe Female ARs | 1 | |||||||
- Pfizer: Out of 23,739 doses administered, ARs occurred in 12 cases (0.05%)
- AstraZeneca: Out of 3,385 doses administered, ARs occurred in 14 cases (0.41%)
- Moderna: Out of 13,780 doses administered, ARs occurred in 13 cases (0.09%)
Therefore, the highest number of adverse reactions by vaccine type, correlating them to the number of doses administered, was with AstraZeneca, though still with very low values.
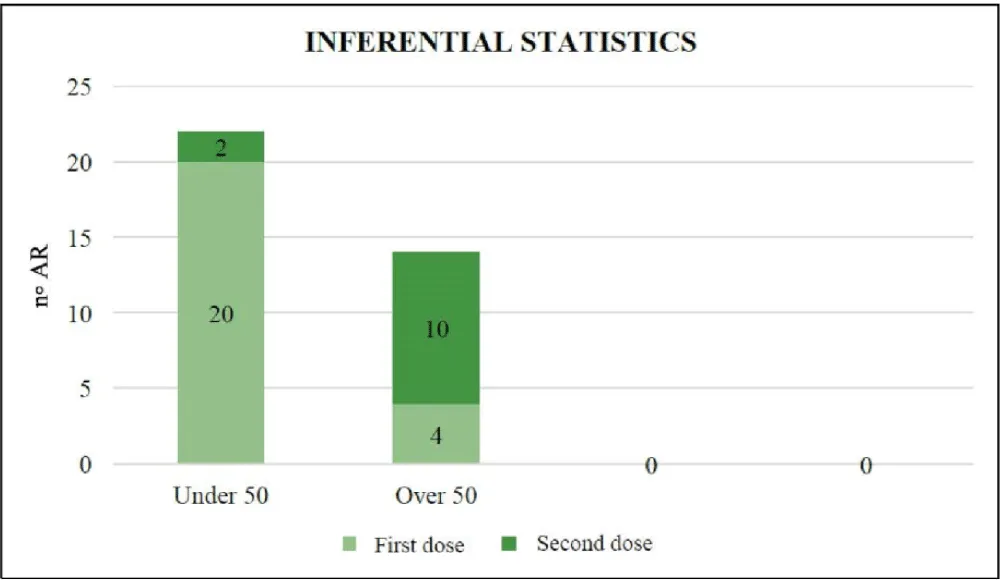
An inferential statistical evaluation was made by dividing the examined sample into two groups based on age, under and over 50 years, correlating ARs to the administration of the first and second doses, which are those for which a higher number of reactions were recorded (Figure 4).
From the graph, it is possible to evaluate that an adverse reaction to the first dose affected 91% (n = 20) of subjects under 50 years of age and 21% (n = 4) of subjects over 50 years; this difference in percentage evaluated with Fisher's exact test with p < 0.001 was statistically significant.
Regarding ARs to the second dose, 9% (n = 2) were recorded for subjects under 50 years of age and 53% (n = 10) for subjects over 50 years of age; in this case too, the difference was statistically significant with Fisher's exact test with p = 0.005.
Statistical evaluations and direct comparisons
Finally, a comparison was made between the data obtained in this study with AIFA (Italian Medicines Agency) data, which manages the National Pharmacovigilance Network (RNF), using as reference the Surveillance Report on COVID-19 vaccines (period 27/12/2020 - 26/12/2022, Report number 14) to see if the results obtained were in line with those at the national level. Regarding EMA (European Medicines Agency), this is a European agency responsible for approving vaccines that can be used in Europe and, together with national regulatory authorities (AIFA in Italy), monitors their safety through data collected by national agencies.
Dose administration
The distribution of doses administered in the local context differs slightly from the national distribution (Figure 5) particularly in the sample data analyzed, the distribution of Pfizer vaccine doses was slightly lower than the national one, while for Moderna vaccine doses it was slightly higher. For other types of vaccines, the values are quite similar.
Reporting rates
Below is a direct comparison between the data from the analyzed sample and AIFA data regarding reporting rate and Confidence Interval: Aifa data
- 140,595 reports on 144,354,770 doses administered
- National overall reporting rate of 97 per 100,000 doses administered, or about 0.097%
- 95% Confidence Interval for the national rate is 96-98
Data from the studied sample
- Regarding the data presented in this study in the time period from 2020 to 2023, a total of 41,276 vaccine doses were administered
- In the same period, 41 Adverse Reactions (AR) were recorded, corresponding to 0.1% of administered doses
- Converting this rate per 100,000 doses: (41 AR / 41,276 doses) * 100,000 ≈ 99.3 AR per 100,000 doses
The overall reporting rate observed in this study was 0.1% compared to 0.097%, with a minimal difference between the two, probably influenced in the first case by the reduced sample size compared to that of AIFA. Consequently, the Confidence Level of the study sample (about 99.3 per 100,000 doses) was very similar to the overall national one reported by AIFA (97 per 100,000 doses), thus falling exactly within the national confidence interval.
An additional comparison was also made in reference to ARs as visible from the comparison of the distributions below between the data reported in this study and national data (Figure 6).
In particular, at the national level, 26,305 severe ARs were reported, equal to 18.7%, corresponding to 18 severe events per 100,000 doses administered. The percentage of ARs classified as severe in the study of the sample analyzed in this study (7.3%) was significantly lower than the national data, but this may have depended on the specific characteristics of the population vaccinated at the Military Polyclinic, local reporting thresholds, or severity classification.
What emerges is that in the local sample, as at the national level, severe adverse reactions were significantly less frequent than non-severe ones.
ARs by dose number
Also regarding the distribution of Adverse Reactions based on dose number, the data analyzed in this study indicate that most ARs occurred after the first dose followed in order by subsequent ones, reflecting the national trend as shown in graph (Figure 6).
Conclusions
The retrospective study conducted at the Military Polyclinic of Rome "Celio" confirms the high safety profile of COVID-19 vaccines administered between 2020 and 2023.
Among 41,276 doses, 41 adverse reactions (0.1%) were recorded, aligning closely with the national rate reported by AIFA (0.097%). Most reactions (92.7%) were non-severe; severe events (7.3%) were rare and occurred mainly in individuals aged 30-49.
Adverse reactions primarily affected the musculoskeletal (29.2%) and immune systems (26.8%), with a greater incidence in female subjects (66%). The majority occurred after the first dose, consistent with national trends. Vaccine distribution showed minor local deviations from national averages, notably a slightly higher use of Moderna and lower of Pfizer.
Ongoing pharmacovigilance by AIFA and EMA continues to ensure safety, with no evidence to date of medium- or longterm risks. Current data supports the effectiveness and safety of vaccination in preventing severe disease, hospitalization, and death, especially in vulnerable populations. The benefits of COVID-19 vaccination clearly outweigh its risks [1-33].
References
- Hosseini R, Askari N. A review of neurological side effects of COVID-19 vaccination. Eur J Med Res. 2023 Feb 25;28(1):102. doi: 10.1186/s40001-023-00992-0. PMID: 36841774; PMCID: PMC9959958.
- Ogar CK, Quick J, Gilbert HN, Vreman RA, Mantel-Teeuwisse AK, Mugunga JC. Adverse Events to SARS-CoV-2 (COVID-19) Vaccines and Policy Considerations that Inform the Funding of Safety Surveillance in Low- and Middle-Income Countries: A Mixed Methods Study. Drug Saf. 2023 Apr;46(4):357-370. doi: 10.1007/s40264-023-01279-3. Epub 2023 Feb 22. PMID: 36811813; PMCID: PMC9945828.
- Salmon DA, Lambert PH, Nohynek HM, Gee J, Parashar UD, Tate JE, Wilder-Smith A, Hartigan-Go KY, Smith PG, Zuber PLF. Novel vaccine safety issues and areas that would benefit from further research. BMJ Glob Health. 2021 May;6(Suppl 2):e003814. doi: 10.1136/bmjgh-2020-003814. PMID: 34011502; PMCID: PMC8137224.
- Barda N, Dagan N, Ben-Shlomo Y, Kepten E, Waxman J, Ohana R, Hernán MA, Lipsitch M, Kohane I, Netzer D, Reis BY, Balicer RD. Safety of the BNT162b2 mRNA Covid-19 Vaccine in a Nationwide Setting. N Engl J Med. 2021 Sep 16;385(12):1078-1090. doi: 10.1056/NEJMoa2110475. Epub 2021 Aug 25. PMID: 34432976; PMCID: PMC8427535.
- Patone M, Handunnetthi L, Saatci D, Pan J, Katikireddi SV, Razvi S, Hunt D, Mei XW, Dixon S, Zaccardi F, Khunti K, Watkinson P, Coupland CAC, Doidge J, Harrison DA, Ravanan R, Sheikh A, Robertson C, Hippisley-Cox J. Neurological complications after first dose of COVID-19 vaccines and SARS-CoV-2 infection. Nat Med. 2021 Dec;27(12):2144-2153. doi: 10.1038/s41591-021-01556-7. Epub 2021 Oct 25. Erratum in: Nat Med. 2021 Dec;27(12):2249. doi: 10.1038/s41591-021-01644-8. PMID: 34697502; PMCID: PMC8629105.
- Klein NP, Lewis N, Goddard K, Fireman B, Zerbo O, Hanson KE, Donahue JG, Kharbanda EO, Naleway A, Nelson JC, Xu S, Yih WK, Glanz JM, Williams JTB, Hambidge SJ, Lewin BJ, Shimabukuro TT, DeStefano F, Weintraub ES. Surveillance for Adverse Events After COVID-19 mRNA Vaccination. JAMA. 2021 Oct 12;326(14):1390-1399. doi: 10.1001/jama.2021.15072. PMID: 34477808; PMCID: PMC8511971.
- Beatty AL. Analysis of myocarditis among 2.5 million vaccinated individuals. Annals of Internal Medicine. 2021;174:1686-1688.
- Mevorach D, Anis E, Cedar N, Bromberg M, Haas EJ, Nadir E, Olsha-Castell S, Arad D, Hasin T, Levi N, Asleh R, Amir O, Meir K, Cohen D, Dichtiar R, Novick D, Hershkovitz Y, Dagan R, Leitersdorf I, Ben-Ami R, Miskin I, Saliba W, Muhsen K, Levi Y, Green MS, Keinan-Boker L, Alroy-Preis S. Myocarditis after BNT162b2 mRNA Vaccine against Covid-19 in Israel. N Engl J Med. 2021 Dec 2;385(23):2140-2149. doi: 10.1056/NEJMoa2109730. Epub 2021 Oct 6. PMID: 34614328; PMCID: PMC8531987.
- Hause AM, Baggs J, Gee J, Marquez P, Myers TR, Shimabukuro TT, Shay DK. Safety Monitoring of an Additional Dose of COVID-19 Vaccine - United States, August 12-September 19, 2021. MMWR Morb Mortal Wkly Rep. 2021 Oct 1;70(39):1379-1384. doi: 10.15585/mmwr.mm7039e4. PMID: 34591835; PMCID: PMC8486391.
- Shimabukuro TT, Kim SY, Myers TR, Moro PL, Oduyebo T, Panagiotakopoulos L, Marquez PL, Olson CK, Liu R, Chang KT, Ellington SR, Burkel VK, Smoots AN, Green CJ, Licata C, Zhang BC, Alimchandani M, Mba-Jonas A, Martin SW, Gee JM, Meaney-Delman DM; CDC v-safe COVID-19 Pregnancy Registry Team. Preliminary Findings of mRNA Covid-19 Vaccine Safety in Pregnant Persons. N Engl J Med. 2021 Jun 17;384(24):2273-2282. doi: 10.1056/NEJMoa2104983. Epub 2021 Apr 21. Erratum in: N Engl J Med. 2021 Oct 14;385(16):1536. doi: 10.1056/NEJMx210016. PMID: 33882218; PMCID: PMC8117969.
- Ledford H. Why COVID vaccines are so difficult to compare. Nature. 2021 Mar;591(7848):16-17. doi: 10.1038/d41586-021-00409-0. PMID: 33623151.
- Kaur RJ, Dutta S, Bhardwaj P, Charan J, Dhingra S, Mitra P, Singh K, Yadav D, Sharma P, Misra S. Adverse Events Reported From COVID-19 Vaccine Trials: A Systematic Review. Indian J Clin Biochem. 2021 Oct;36(4):427-439. doi: 10.1007/s12291-021-00968-z. Epub 2021 Mar 27. PMID: 33814753; PMCID: PMC7997788.
- Gee J, Marquez P, Su J, Calvert GM, Liu R, Myers T, Nair N, Martin S, Clark T, Markowitz L, Lindsey N, Zhang B, Licata C, Jazwa A, Sotir M, Shimabukuro T. First Month of COVID-19 Vaccine Safety Monitoring - United States, December 14, 2020-January 13, 2021. MMWR Morb Mortal Wkly Rep. 2021 Feb 26;70(8):283-288. doi: 10.15585/mmwr.mm7008e3. PMID: 33630816; PMCID: PMC8344985.
- Lee EJ, Cines DB, Gernsheimer T, Kessler C, Michel M, Tarantino MD, Semple JW, Arnold DM, Godeau B, Lambert MP, Bussel JB. Thrombocytopenia following Pfizer and Moderna SARS-CoV-2 vaccination. Am J Hematol. 2021 May 1;96(5):534-537. doi: 10.1002/ajh.26132. Epub 2021 Mar 9. PMID: 33606296; PMCID: PMC8014568.
- Klein NP, Lewis N, Goddard K, Fireman B, Zerbo O, Hanson KE, Donahue JG, Kharbanda EO, Naleway A, Nelson JC, Xu S, Yih WK, Glanz JM, Williams JTB, Hambidge SJ, Lewin BJ, Shimabukuro TT, DeStefano F, Weintraub ES. Surveillance for Adverse Events After COVID-19 mRNA Vaccination. JAMA. 2021 Oct 12;326(14):1390-1399. doi: 10.1001/jama.2021.15072. PMID: 34477808; PMCID: PMC8511971.
- Oster ME, Shay DK, Su JR, Gee J, Creech CB, Broder KR, Edwards K, Soslow JH, Dendy JM, Schlaudecker E, Lang SM, Barnett ED, Ruberg FL, Smith MJ, Campbell MJ, Lopes RD, Sperling LS, Baumblatt JA, Thompson DL, Marquez PL, Strid P, Woo J, Pugsley R, Reagan-Steiner S, DeStefano F, Shimabukuro TT. Myocarditis Cases Reported After mRNA-Based COVID-19 Vaccination in the US From December 2020 to August 2021. JAMA. 2022 Jan 25;327(4):331-340. doi: 10.1001/jama.2021.24110. PMID: 35076665; PMCID: PMC8790664.
- Krug A. Incidence of severe adverse events after vaccination with mRNA-based COVID-19 vaccines. Vaccine. 2022;40:4487-4494.
- Chouchana L. Incidence of myocarditis and pericarditis after COVID-19 mRNA vaccines. Clinical Pharmacology & Therapeutics. 2022;112:1003-1012.
- Høeg TB, Kruget A, Mandrola J. SARS-CoV-2 mRNA vaccinationassociated myocarditis in children aged. 2021:12-17. doi: 10.1101/2021.08.30.21262866.
- Kaur U. Systematic review of adverse events associated with COVID-19 vaccines. Frontiers in Public Health. 2022;10:912671.
- Wise J. Covid-19: European countries suspend use of Oxford-AstraZeneca vaccine after reports of blood clots. BMJ. 2021 Mar 11;372:n699. doi: 10.1136/bmj.n699. PMID: 33707182.
- Greinacher A, Thiele T, Warkentin TE, Weisser K, Kyrle PA, Eichinger S. Thrombotic Thrombocytopenia after ChAdOx1 nCov-19 Vaccination. N Engl J Med. 2021 Jun 3;384(22):2092-2101. doi: 10.1056/NEJMoa2104840. Epub 2021 Apr 9. PMID: 33835769; PMCID: PMC8095372.
- Schulz JB. COVID-19 vaccine-associated neurological autoimmune diseases. Brain. 2021;144:3576-3588.
- Wang W. Adverse events of COVID-19 vaccines in patients with autoimmune diseases. Autoimmunity Reviews 2022;21:103160. doi: 10.1016/S2665-9913(21)00181-8.
- Al Khames Aga Q. Safety of COVID-19 vaccines in pregnancy: A systematic review.Pharmaceuticals. 2021;14:849. doi: 10.1016/j.vaccine.2023.03.038.
- Kaminski M. Immunological responses to COVID-19 vaccines: A review. Cells. 2022;11:439.
- Hatmal MM. Comprehensive analysis of reported adverse events of COVID-19 vaccines. Vaccines. 2021;9:926.
- Widge AT, Rouphael NG, Jackson LA, Anderson EJ, Roberts PC, Makhene M, Chappell JD, Denison MR, Stevens LJ, Pruijssers AJ, McDermott AB, Flach B, Lin BC, Doria-Rose NA, O'Dell S, Schmidt SD, Neuzil KM, Bennett H, Leav B, Makowski M, Albert J, Cross K, Edara VV, Floyd K, Suthar MS, Buchanan W, Luke CJ, Ledgerwood JE, Mascola JR, Graham BS, Beigel JH; mRNA-1273 Study Group. Durability of Responses after SARS-CoV-2 mRNA-1273 Vaccination. N Engl J Med. 2021 Jan 7;384(1):80-82. doi: 10.1056/NEJMc2032195. Epub 2020 Dec 3. PMID: 33270381; PMCID: PMC7727324.
- Logunov DY, Dolzhikova IV, Shcheblyakov DV, Tukhvatulin AI, Zubkova OV, Dzharullaeva AS, Kovyrshina AV, Lubenets NL, Grousova DM, Erokhova AS, Botikov AG, Izhaeva FM, Popova O, Ozharovskaya TA, Esmagambetov IB, Favorskaya IA, Zrelkin DI, Voronina DV, Shcherbinin DN, Semikhin AS, Simakova YV, Tokarskaya EA, Egorova DA, Shmarov MM, Nikitenko NA, Gushchin VA, Smolyarchuk EA, Zyryanov SK, Borisevich SV, Naroditsky BS, Gintsburg AL; Gam-COVID-Vac Vaccine Trial Group. Safety and efficacy of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine: an interim analysis of a randomised controlled phase 3 trial in Russia. Lancet. 2021 Feb 20;397(10275):671-681. doi: 10.1016/S0140-6736(21)00234-8. Epub 2021 Feb 2. Erratum in: Lancet. 2021 Feb 20;397(10275):670. doi: 10.1016/S0140-6736(21)00386-X. PMID: 33545094; PMCID: PMC7852454.
- McDonald I. Comparative systematic review and meta-analysis of reactogenicity, immunogenicity and efficacy of COVID-19 vaccines. NPJ Vaccines. 2021;6:1-14.
- De Vuyst H. Vaccine safety surveillance in the context of COVID- 19: WHO approaches. Vaccine. 2021;39:6670-6675.
- Singh A. Myocarditis and pericarditis after mRNA COVID-19 vaccination. Heart, Lung and Circulation 31 (2022): 532-541.
- Barda N. Effectiveness and safety of third dose of BNT162b2 mRNA COVID-19 vaccine. The Lancet. 2021;398:2093-2100.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.