2025 October 10;6(10):1424-1431. doi: 10.37871/jbres2198.
Zebrafish as a Versatile Model for Cancer Research: Insights from In vitro and In vivo Studies
Saili Paul1 and Anisur Rahman Khuda-Bukhsh2*
2Retired Professor and Emeritus Fellow of UGC, Department of Zoology, University of Kalyani, Kalyani-741235, W.B., India
- Zebrafish
- Cancer model
- Xenograft
- Angiogenesis
- nanoparticles
- Carcinogen
Abstract
Rodent models have provided critical insights into the developmental biology of cancer cells and host responses to transformation. However, mortality from several malignancies remains high, underscoring the need for alternative animal models that allow the integrated study of cancer cell biology, developmental processes, and therapeutic interventions. The zebrafish has emerged as a powerful model owing to its rapid development, tractable genetics, suitability for in vivo imaging, and compatibility with chemical screening.
Multiple approaches-including genetic, xenograft, and chemical models- have been established in zebrafish to investigate alterations in molecular pathways, gene functions during cancer progression, and to evaluate novel anticancer agents. In particular, zebrafish xenograft models provide a low-cost, high-throughput platform that can be rapidly established with minimal sample requirements, making them highly attractive materials for cancer research. These models have been instrumental in exploring tumor proliferation, metastasis, and angiogenesis, while also enabling the study of drug pharmacokinetics, tumor–microenvironment interactions, and the prediction of personalized treatment efficacy.
Collectively, zebrafish models represent a versatile and translationally relevant system for studying tumor biology and accelerating the discovery and evaluation of anticancer therapeutics.
Introduction
Zebrafish have become an established tool for both drug discovery and toxicity evaluation [1]. They possess numerous characteristics that make them an an ideal model for cancer drug development. In contrast to mice, zebrafish are highly fecund and display rapid development. Mating can yield hundreds of fertilized eggs that quickly develop from embryos into larval fish, hatching at approximately 72 hours post-fertilization (hpf) [2]. Zebrafish reach breeding maturity by 90 days post-fertilization. Although they are not mammals, zebrafish have a comparable vertebrate anatomy to humans and possess orthologs for 70% of human proteins [3]. With current technology, it is relatively easy to generate zebrafish transgenic models with altered gene expression. The use of transgenic zebrafish cancer models has enabled the study of numerous malignancies [4], and transgenic models with vascular endothelial cell-specific expression of fluorescent proteins have facilitated discoveries related to developmental and tumor-induced neo-angiogenesis and vascularization [5].
The zebrafish (Danio rerio) has become one of the most important vertebrate model organisms in biomedical research. Since the initial studies into genetics and vertebrate development, the zebrafish model has evolved into a powerful tool for studying many human diseases, including in cancer biology.
Zebrafish are small in size and are easy to breed and maintain in large numbers at low cost. Laboratory zebrafish are housed in purpose-built aquaculture systems, maintained at 28.5°C with a controllable light–dark cycle. They are highly fecund, and a gravid female can produce hundreds of eggs each week through natural spawning. Fertilization and embryo development occur externally to the female. Embryonic development is rapid; within 24 hours, the single-celled zygote develops into a motile, transparent embryo with a classical vertebrate body plan [6]. Maintained at 28.5°C, the majority of morphogenesis is complete by 3 days post-fertilization (dpf), and the embryo hatches from the protective chorion, marking the transition from the embryonic to the larval stage. The larval zebrafish will continue to grow, and between 5 and 6 dpf, the digestive system and mouth become functional. The yolk sac, which has sustained the animal throughout the embryonic and early larval stages, is rapidly depleted and is completely absorbed by 7 dpf [6].
The zebrafish animal model has many well-known characteristics and advantages that make it an excellent model for biological research and the study of human diseases [7].
As zebrafish provide a robust model for molecular studies of many human diseases, cancers are among the most studied disorders using this model due to the simplicity of designing experiments through genetic engineering, xenotransplantation, and chemical exposures [8-10].
Materials and Methods
Cancer models
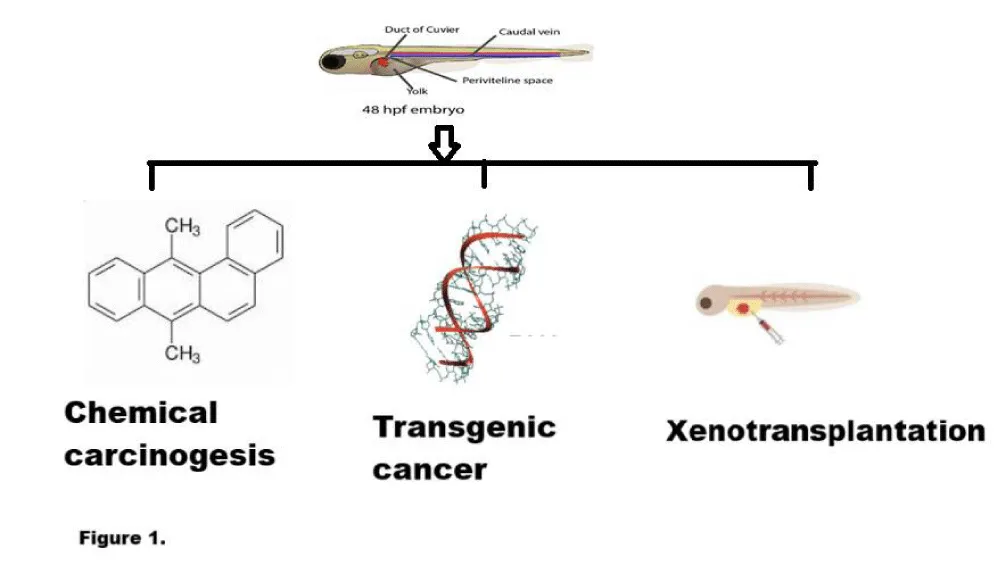
Danio rerio have been used as different types of models for various types of studies. The major types have been summarized in figure 1.
Xenograft model
Xenografted zebrafish embryos are one approach utilized for cancer studies by transplanting human cancer cells into the animal model. The transplantation of cancer cells is possible at around 2 days post-fertilization (dpf), when the zebrafish adaptive immune response is not yet fully established. Therefore, using 2 dpf embryos allows for the spread, metastasis, and survival of cancer cells without the need for prior immunosuppressive treatment [11]. Diverse engraftment assays can be carried out in these zebrafish. Allogenic transplantation is the transfer of cells, organs, or tissues from one individual to another of the same species.
Xenografting is the process of implanting living tumor cells from one species into another. Lee and colleagues performed the first xenotransplant of human cancer cells into zebrafish embryos to model melanoma [12]. Orthotopic transplantation is a xenotransplatation of tumor cells, which involves cell implantation into the same site or organ where cancer has developed in the donor.
Transgenic model
Researchers generate transgenic zebrafish models by microinjecting exogenous DNA into one-cell-stage embryos, resulting in the misexpression of wild-type or constitutively active oncogenes under the control of zebrafish tissue-specific promoters [13]. The plasmid construct used for zebrafish transgenesis typically includes a promoter region, the target gene, and flanking regulatory sequences essential for stable gene expression.
These transgenic and mutant zebrafish models are powerful tools for investigating various diseases, particularly cancer, and are developed using either forward or reverse genetic approaches [14]. Forward genetics often employs Ethylnitrosourea (ENU) mutagenesis to induce random mutations, whereas reverse genetics utilizes targeted genome-editing tools such as CRISPR/Cas9 and TAL Effector Nucleases (TALENs) [15].
The high evolutionary conservation of oncogenes such as NRAS^Q61K and BRAF^V600E in zebrafish cancer models, combined with the microinjection of human tissue-specific promoters, enables the development of tumors that closely resemble human cancer phenotypes [16]. Moreover, reverse genetic approaches involving the knockdown or knockout of specific genes provide rapid insights into gene function and their roles in tumorigenesis.
Notably, Ju B, et al. [17] demonstrated that expression of human oncogenic KRAS, driven by the zebrafish krt5 or gfap promoter, induces malignant tumors of the nervous system.
Carcinogenic model
The use of chemical carcinogens was one of the first approaches to induce mutations for tumor formation or developmental defects in the zebrafish model. In this method, the animals are exposed to carcinogenic chemicals dissolved or suspended in their cistern water, allowing for long-term exposure. There are various ways to induce cancer in zebrafish, including chemical mutagenesis, irradiation mutagenesis, and insertional mutagenesis, which can be transposon-based or viral vector-based. Researchers have successfully induced several cancer types by adding carcinogens to the water, such as Dibenzo(a,l)Pyrene (DBP), 7,12-Dimethylbenz(a)Anthracene (DMBA), N-methyl-N′-nitro-N-nitrosoguanidine (MNNG), N-Dimethylnitrosamine (DEN), and N-nitrosodiethylamine (NDMA) (Table 1).
| Table 1: Types of cancer models. | ||
| Model | Approaches | References |
| Xenograft Models | Xenografted zebrafish embryos are one of the approaches for modeling cancer through transplanting human cancer cells into the animal model. | [11] |
| Genetic Models | Transgenic and mutant zebrafish models are major and powerful utilizing models for diseases and cancer research as these models are generated by different techniques, mainly through forward or reverse genetic approaches. | [15] |
| Chemical Models | The use of chemical carcinogens was one of the first approaches to induce cancer mutations for tumor formation or developmental defects in the zebrafish animal model. | [15] |
According to their role in developing different cancer types, the cancer models have been summarized in table 1.
Monitoring cancer processes in zebrafish
In vivo monitoring of tumor processes in zebrafish requires specific and often expensive imaging techniques, as well as qualified personnel. Some of the approaches used for cancer monitoring in zebrafish are shown in table 2. For many applications, imaging fluorescently labeled tumors in whole zebrafish is advantageous. For example, whole-animal imaging can be used to assess growth kinetics and overall dissemination. Additionally, cell transplantation can be employed to determine whether fluorescently labelled cell populations are fully transformed-a hallmark of cancer. The different imaging tools used in different types of cancer studies are shown in table 2.
| Table 2: Types of imaging tools utilised for the analysis of tumor size or tumor growth. | ||
| Microscopy type | Application | Reference |
| Stereo-Miscroscopy | The (fli:GFP) Casper zebrafish embryo can be used as an efficient animal model to study metastatic behavior of human CM cells. | [18] [19] |
| PET imaging (Positron emission tomography) | PET imaging technique is used to visualize and quantify metabolic and physiological process in zebrafish cancer xenograft models. | [18] [20] |
| Fluorescence microscopy | Perform immunofluorescence of caspase-3 on CRC zebrafish xenograft models | [21] [22] |
| Confocal microscopy | Melanoma cell positioned between the zebrafish endothelial cells, indicating that this cell is directly within the blood vessel. | [23] [24] |
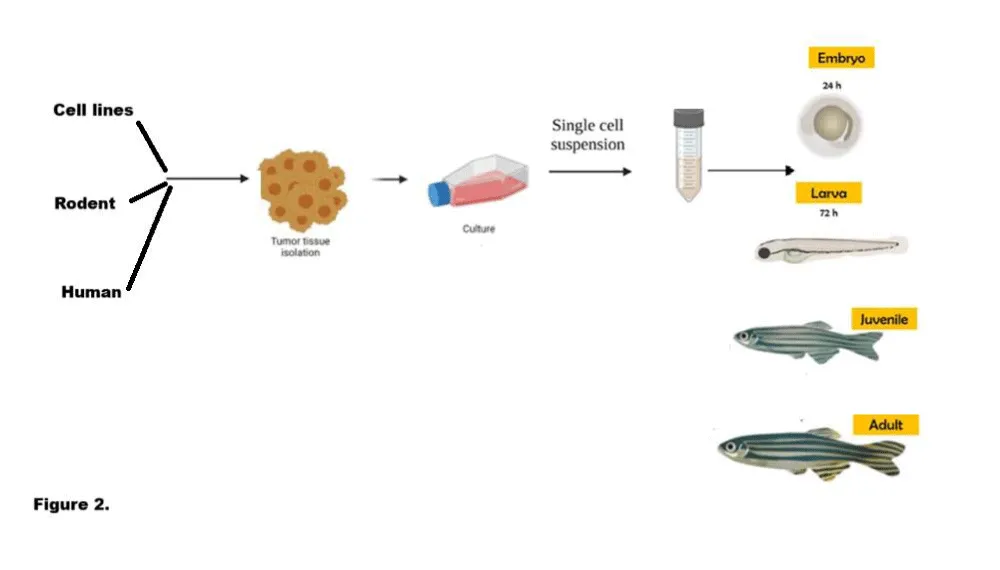
Zebrafish xenotransplantation model for evaluation of cancer progression
Zebrafish serve as a valuable tool in human cancer xenotransplantation studies, potentially overcoming some of the drawbacks associated with murine models. The main benefits of using zebrafish are most prominent when employing embryonic stages for xenotransplantation (Figure 2). The site of transplantation may vary, but it is typically the yolk sac, cardinal vein, duct of Cuvier, or hindbrain (Table 3). Depending on the site of transplantation, different phenotypes of tumorigenesis can be observed, such as cancer cells invasion, extravasation, and metastasis.
| Table 3: Human cancer xenograft transplantation models in zebrafish. | |||
| Transplanted cancer type | Developmental stage | Injection site | References |
| Melanoma | Blastula | Blastodisc | [12] |
| Colorectal | 48 hpf | Yolk sac | [25] |
| Prostate | 48 hpf | Yolk sac | [26] |
| Breast | 48hpf | Yolk sac | [27] |
| Glioblastoma | 72hpf | Brain | [28] |
| Non-small lung cancer | 48hpf | Yolk sac | [29] |
Tumor angiogenesis in zebrafish embryos
Angiogenesis, the process of forming new blood vessels from pre-existing ones, plays a key role in tumor growth and metastasis. The transparent body of zebrafish allows for clear observation of tumor metastasis under a microscope. Thus, identifying anti-angiogenic drugs and angiogenesis-related targets has significant implications for the development of anti-neoplastic therapies. In this respect, tumor cell engrafting in zebrafish embryos may provide a valid alternative to other in vivo vertebrate animal models. The application of transgenic angio-fluorescent strains of zebrafish enables direct, in vivo, real-time evaluation of angiogenesis-inhibiting drugs on the neovascular system of tumor tissues, facilitating rapid and precise screening and effect analysis of anti-angiogenic drugs [30].
Tumor invasiveness and metastasis in zebrafish embryos
The vast majority of cancer deaths are caused by the metastatic expansion of primary tumors as a consequence of cancer cell dissemination into the body, primarily via the lymphatic and blood vessels. Several anti-metastatic drugs have been identified through studies using zebrafish models [31]. Briefly, human cancer cells labeled with a fluorescent marker, such as Enhanced Green Fluorescent Protein (EGFP), are inoculated into the larvae or embryos of zebrafish. This approach is effective because zebrafish embryos are transparent and do not have a fully developed adaptive immune system until 21 days post-fertilization (dpf) [32]. Zebrafish are maintained for a few days with or without the drug, and the effects of the drug are evaluated by observing the behavior of the cells during tumor invasion, migration, extravasation, and angiogenesis. Compounds that suppress the metastatic dissemination of cancer cells in zebrafish xenograft models are listed in table 4.
| Table 4: List of anti-metastatic drugs identified in zebrafish xenograft model. | ||||||
| Drug | Target molecule | Targeting molecular event | Recipient fish | Cancer cell | Inoculation site | Reference |
| Osimertinib | EGFR | Angiogenesis | Tg(Fli1: EGFP) | H1975 | Duct of Cuvier | [33] |
| Sunitinib | VGFR | Angiogenesis | Tg(Fli1: EGFP) | T241 (Thyroid) | Perivitelline space | [34] |
| Tenovin-6 | SIRT1/2 | Migration | Tg(Fli1: EGFP) | TC252 (Ewing sarcoma) and A673 (Ewing sarcoma) | Duct of Cuvier | [35] |
| Lipofermata | FATP | Invasion | Casper | ZMEL1 (Skin) | Subcutaneous tissue | [36] |
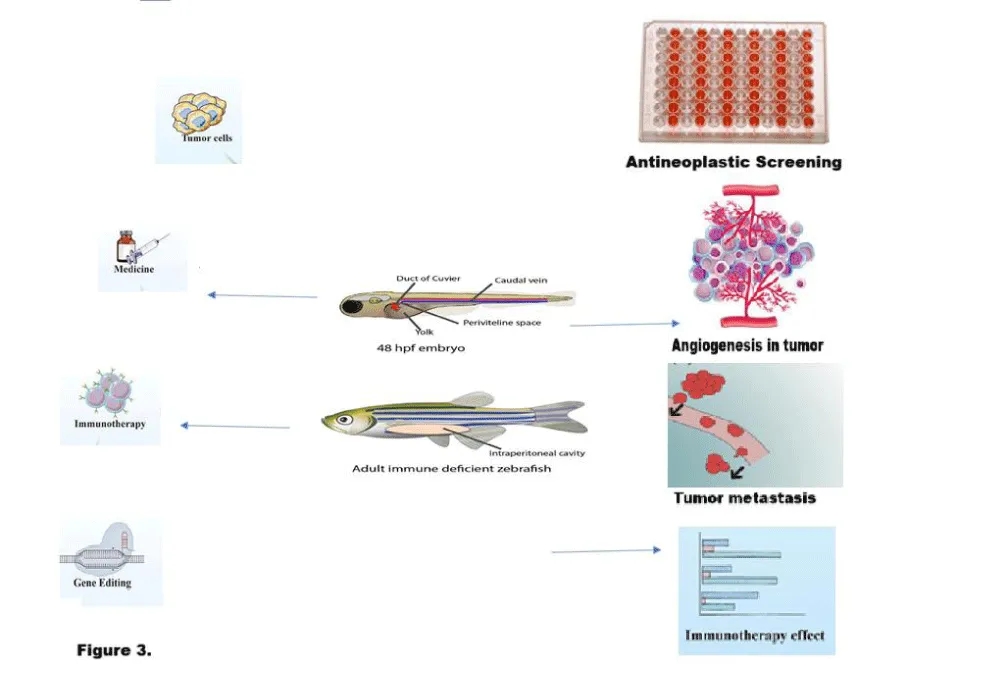
The zebrafish field has seen significant growth over the past 10 years, as the rapid application of transgenic and chemical screening techniques has placed this model in a unique category of cancer research (Figure 3).
Preclinical Testing of Nanoparticles Using Zebrafish Models of Disease
Anti-cancer nanoparticles tested on zebrafish
Developing Nanoparticles (NPs) that can identify, target, and eliminate cancer cells is a significant goal in the field of nanomedicine. Several studies have also demonstrated the use of zebrafish to evaluate drug delivery efficacy for increasing bioavailability or for targeted drug transfer. However, to test these particles, an ideal animal model must be selected to determine the efficacy and toxicity of the NPs (Table 5). Therefore, zebrafish have been designated as an alternative model for testing these particles due to the several advantages previously mentioned.
| Table 5: Evaluation of nanoparticle-based approaches for cancer treatment using xenograft zebrafish models. | ||||||
| Nano system | Drug | Mechanism | Tumor Type/ Cell Line |
Zebrafish Stage | Injection Site | Reference |
| Zinc oxide NPs | - | Apoptosis and ROS induction | Gingival squamous cell carcinoma/ Ca9-22-DiL |
48 hpf | Yolk sac | [37] |
| NanogelDOX | DOX | Hydrazone sensitivity to pH accelerate drug release | Melanoma/ B6-RFP or GPF |
48 hpf | Neural tube | [38] |
Zebrafish xenografts enable drug discovery validation and screening
The zebrafish xenograft assay has frequently been used as an in vivo reporter for validating the efficacy of anticancer drugs. These studies demonstrate the utility of zebrafish xenograft models for investigating a wide variety of cancer mechanisms, often leading to the identification of drug targets for anticancer treatments (Table 6).
Application in Tumor Drug Screening
The research and development of novel drugs are lengthy and expensive processes. Drugs are initially developed through in vitro tests, where the effects of small-molecule therapies on cells are assessed by measuring cell proliferation, cytotoxicity, marker expression, migration, signaling pathway activation, and morphological changes. Following this, in vivo tests are conducted to evaluate the half-life of a drug and perform final drug screening. Zebrafish are aquatic organisms that can absorb small-molecule compounds directly from the water [42], making them more suitable for small-molecule drug screening and drug delivery pathway research than mice.
Conclusion
From the abovementioned information on various aspects and insights, Zebrafish have proven to be reliable models for studying and visualizing human cancer cell biology and dynamics, including metastasis and tumor tissue neo-angiogenesis, in vivo. Furthermore, the involvement of epigenetic modulators in tumor biology could enhance our understanding of complex diseases such as cancer. The availability of transgenic and mutant models, along with the ability to transplant cancer cells into zebrafish, provides a wide array of options for studying human cancer. Although zebrafish are non-mammalian model organisms, they exhibit significant evolutionary conservation of disease-related genes and pathways with humans. While searching for novel drugs can be conducted in vitro on a large scale, the effects on a whole living organism may differ strikingly. Screening for targeted treatments in zebrafish xenografts could offer new opportunities for personalized anticancer therapy in the future, as recent research has demonstrated that zebrafish studies are reliable for modelling human cancer.
Acknowledgement
Saili Paul thanks the Principal of Kanchrapara College for his kind encouragements.
References
- Cassar S, Adatto I, Freeman JL, Gamse JT, Iturria I, Lawrence C, Muriana A, Peterson RT, Van Cruchten S, Zon LI. Use of Zebrafish in Drug Discovery Toxicology. Chem Res Toxicol. 2020 Jan 21;33(1):95-118. doi: 10.1021/acs.chemrestox.9b00335. Epub 2019 Nov 16. PMID: 31625720; PMCID: PMC7162671.
- Longkumer S, Jamir A, Pankaj PP. Maintenance and Breeding of Zebrafish under Laboratory Conditions for Animal Research. Agricultural Science Digest, 2024 June 44(3): 551-555. doi: 10.18805/ag.D-5599.
- Howe K, Clark MD, Torroja CF, Torrance J, Berthelot C, Muffato M, Collins JE, Humphray S, McLaren K, Matthews L, McLaren S, Sealy I, Caccamo M, Churcher C, Scott C, Barrett JC, Koch R, Rauch GJ, White S, Chow W, Kilian B, Quintais LT, Guerra-Assunção JA, Zhou Y, Gu Y, Yen J, Vogel JH, Eyre T, Redmond S, Banerjee R, Chi J, Fu B, Langley E, Maguire SF, Laird GK, Lloyd D, Kenyon E, Donaldson S, Sehra H, Almeida-King J, Loveland J, Trevanion S, Jones M, Quail M, Willey D, Hunt A, Burton J, Sims S, McLay K, Plumb B, Davis J, Clee C, Oliver K, Clark R, Riddle C, Elliot D, Threadgold G, Harden G, Ware D, Begum S, Mortimore B, Kerry G, Heath P, Phillimore B, Tracey A, Corby N, Dunn M, Johnson C, Wood J, Clark S, Pelan S, Griffiths G, Smith M, Glithero R, Howden P, Barker N, Lloyd C, Stevens C, Harley J, Holt K, Panagiotidis G, Lovell J, Beasley H, Henderson C, Gordon D, Auger K, Wright D, Collins J, Raisen C, Dyer L, Leung K, Robertson L, Ambridge K, Leongamornlert D, McGuire S, Gilderthorp R, Griffiths C, Manthravadi D, Nichol S, Barker G, Whitehead S, Kay M, Brown J, Murnane C, Gray E, Humphries M, Sycamore N, Barker D, Saunders D, Wallis J, Babbage A, Hammond S, Mashreghi-Mohammadi M, Barr L, Martin S, Wray P, Ellington A, Matthews N, Ellwood M, Woodmansey R, Clark G, Cooper J, Tromans A, Grafham D, Skuce C, Pandian R, Andrews R, Harrison E, Kimberley A, Garnett J, Fosker N, Hall R, Garner P, Kelly D, Bird C, Palmer S, Gehring I, Berger A, Dooley CM, Ersan-Ürün Z, Eser C, Geiger H, Geisler M, Karotki L, Kirn A, Konantz J, Konantz M, Oberländer M, Rudolph-Geiger S, Teucke M, Lanz C, Raddatz G, Osoegawa K, Zhu B, Rapp A, Widaa S, Langford C, Yang F, Schuster SC, Carter NP, Harrow J, Ning Z, Herrero J, Searle SM, Enright A, Geisler R, Plasterk RH, Lee C, Westerfield M, de Jong PJ, Zon LI, Postlethwait JH, Nüsslein-Volhard C, Hubbard TJ, Roest Crollius H, Rogers J, Stemple DL. The zebrafish reference genome sequence and its relationship to the human genome. Nature. 2013 Apr 25;496(7446):498-503. doi: 10.1038/nature12111. Epub 2013 Apr 17. Erratum in: Nature. 2014 Jan 9;505(7482):248. Cooper, James [corrected to Cooper, James D]; Eliott, David [corrected to Elliot, David]; Mortimer, Beverly [corrected to Mortimore, Beverley]; Begum, Sharmin [added]; Lloyd, Christine [added]; Lanz, Christa [added]; Raddatz, Günter [added]; Schuster, Ste. PMID: 23594743; PMCID: PMC3703927.
- Stoletov K, Klemke R. Catch of the day: zebrafish as a human cancer model. Oncogene. 2008 Jul 31;27(33):4509-20. doi: 10.1038/onc.2008.95. Epub 2008 Mar 31. PMID: 18372910.
- Tobia C, De Sena G, Presta M. Zebrafish embryo, a tool to study tumor angiogenesis. Int J Dev Biol. 2011;55(4-5):505-9. doi: 10.1387/ijdb.103238ct. PMID: 21858773.
- Kimmel CB, Ballard WW, Kimmel SR, Ullmann B, Schilling TF. Stages of embryonic development of the zebrafish. Dev Dyn. 1995 Jul;203(3):253-310. doi: 10.1002/aja.1002030302. PMID: 8589427.
- Teame T, Zhang Z, Ran C, Zhang H, Yang Y, Ding Q, Xie M, Gao C, Ye Y, Duan M, Zhou Z. The use of zebrafish (Danio rerio) as biomedical models. Anim Front. 2019 Jun 25;9(3):68-77. doi: 10.1093/af/vfz020. PMID: 32002264; PMCID: PMC6951987.
- Konantz M, Balci TB, Hartwig UF, Dellaire G, André MC, Berman JN, Lengerke C. Zebrafish xenografts as a tool for in vivo studies on human cancer. Ann N Y Acad Sci. 2012 Aug;1266:124-37. doi: 10.1111/j.1749-6632.2012.06575.x. PMID: 22901264.
- Huang H, Vogel SS, Liu N, Melton DA, Lin S. Analysis of pancreatic development in living transgenic zebrafish embryos. Mol Cell Endocrinol. 2001 May 25;177(1-2):117-24. doi: 10.1016/s0303-7207(01)00408-7. PMID: 11377827.
- Feitsma H, Cuppen E. Zebrafish as a cancer model. Mol Cancer Res. 2008 May;6(5):685-94. doi: 10.1158/1541-7786.MCR-07-2167. PMID: 18505914.
- Lam SH, Chua HL, Gong Z, Lam TJ, Sin YM. Development and maturation of the immune system in zebrafish, Danio rerio: a gene expression profiling, in situ hybridization and immunological study. Dev Comp Immunol. 2004 Jan;28(1):9-28. doi: 10.1016/s0145-305x(03)00103-4. PMID: 12962979.
- Lee LM, Seftor EA, Bonde G, Cornell RA, Hendrix MJ. The fate of human malignant melanoma cells transplanted into zebrafish embryos: assessment of migration and cell division in the absence of tumor formation. Dev Dyn. 2005 Aug;233(4):1560-70. doi: 10.1002/dvdy.20471. PMID: 15968639.
- Huiting LN, Laroche F, Feng H. The Zebrafish as a Tool to Cancer Drug Discovery. Austin J Pharmacol Ther. 2015;3(2):1069. Epub 2015 May 4. PMID: 26835511; PMCID: PMC4731041.
- Mimeault M, Batra SK. Emergence of zebrafish models in oncology for validating novel anticancer drug targets and nanomaterials. Drug Discov Today. 2013 Feb;18(3-4):128-40. doi: 10.1016/j.drudis.2012.08.002. Epub 2012 Aug 10. PMID: 22903142; PMCID: PMC3562372.
- Xie X, Ross JL, Cowell JK, Teng Y. The promise of zebrafish as a chemical screening tool in cancer therapy. Future Med Chem. 2015;7(11):1395-405. doi: 10.4155/fmc.15.73. PMID: 26230879.
- White R, Rose K, Zon L. Zebrafish cancer: the state of the art and the path forward. Nat Rev Cancer. 2013 Sep;13(9):624-36. doi: 10.1038/nrc3589. PMID: 23969693; PMCID: PMC6040891.
- Ju B, Chen W, Orr BA, Spitsbergen JM, Jia S, Eden CJ, Henson HE, Taylor MR. Oncogenic KRAS promotes malignant brain tumors in zebrafish. Mol Cancer. 2015 Feb 3;14(1):18. doi: 10.1186/s12943-015-0288-2. PMID: 25644510; PMCID: PMC4320811.
- White RM, Sessa A, Burke C, Bowman T, LeBlanc J, Ceol C, Bourque C, Dovey M, Goessling W, Burns CE, Zon LI. Transparent adult zebrafish as a tool for in vivo transplantation analysis. Cell Stem Cell. 2008 Feb 7;2(2):183-9. doi: 10.1016/j.stem.2007.11.002. PMID: 18371439; PMCID: PMC2292119.
- Pontes KCS, Groenewoud A, Cao J, Ataide LMS, Snaar-Jagalska E, Jager MJ. Evaluation of (fli:GFP) Casper Zebrafish Embryos as a Model for Human Conjunctival Melanoma. Invest Ophthalmol Vis Sci. 2017 Dec 1;58(14):6065-6071. doi: 10.1167/iovs.17-22023. PMID: 29204645.
- Yan C, Brunson DC, Tang Q, Do D, Iftimia NA, Moore JC, Hayes MN, Welker AM, Garcia EG, Dubash TD, Hong X, Drapkin BJ, Myers DT, Phat S, Volorio A, Marvin DL, Ligorio M, Dershowitz L, McCarthy KM, Karabacak MN, Fletcher JA, Sgroi DC, Iafrate JA, Maheswaran S, Dyson NJ, Haber DA, Rawls JF, Langenau DM. Visualizing Engrafted Human Cancer and Therapy Responses in Immunodeficient Zebrafish. Cell. 2019 Jun 13;177(7):1903-1914.e14. doi: 10.1016/j.cell.2019.04.004. Epub 2019 Apr 25. PMID: 31031007; PMCID: PMC6570580.
- Blackburn JS, Liu S, Raimondi AR, Ignatius MS, Salthouse CD, Langenau DM. High-throughput imaging of adult fluorescent zebrafish with an LED fluorescence macroscope. Nat Protoc. 2011 Feb;6(2):229-41. doi: 10.1038/nprot.2010.170. Epub 2011 Feb 3. PMID: 21293462; PMCID: PMC3362200.
- Cross LM, Cook MA, Lin S, Chen JN, Rubinstein AL. Rapid analysis of angiogenesis drugs in a live fluorescent zebrafish assay. Arterioscler Thromb Vasc Biol. 2003 May 1;23(5):911-2. doi: 10.1161/01.ATV.0000068685.72914.7E. PMID: 12740225.
- Zhang L, Alt C, Li P, White RM, Zon LI, Wei X, Lin CP. An optical platform for cell tracking in adult zebrafish. Cytometry A. 2012 Feb;81(2):176-82. doi: 10.1002/cyto.a.21167. Epub 2011 Dec 7. PMID: 22162445; PMCID: PMC6383206.
- Hill D, Chen L, Snaar-Jagalska E, Chaudhry B. Embryonic zebrafish xenograft assay of human cancer metastasis. F1000Res. 2018 Oct 22;7:1682. doi: 10.12688/f1000research.16659.2. PMID: 30473782; PMCID: PMC6234738.
- Roel M, Rubiolo JA, Guerra-Varela J, Silva SB, Thomas OP, Cabezas-Sainz P, Sánchez L, López R, Botana LM. Marine guanidine alkaloids crambescidins inhibit tumor growth and activate intrinsic apoptotic signaling inducing tumor regression in a colorectal carcinoma zebrafish xenograft model. Oncotarget. 2016 Dec 13;7(50):83071-83087. doi: 10.18632/oncotarget.13068. PMID: 27825113; PMCID: PMC5347754.
- Ghotra VP, He S, van der Horst G, Nijhoff S, de Bont H, Lekkerkerker A, Janssen R, Jenster G, van Leenders GJ, Hoogland AM, Verhoef EI, Baranski Z, Xiong J, van de Water B, van der Pluijm G, Snaar-Jagalska BE, Danen EH. SYK is a candidate kinase target for the treatment of advanced prostate cancer. Cancer Res. 2015 Jan 1;75(1):230-40. doi: 10.1158/0008-5472.CAN-14-0629. Epub 2014 Nov 11. PMID: 25388286.
- Naber HP, Drabsch Y, Snaar-Jagalska BE, ten Dijke P, van Laar T. Snail and Slug, key regulators of TGF-β-induced EMT, are sufficient for the induction of single-cell invasion. Biochem Biophys Res Commun. 2013 May 24;435(1):58-63. doi: 10.1016/j.bbrc.2013.04.037. Epub 2013 Apr 22. PMID: 23618854.
- Zeng A, Ye T, Cao D, Huang X, Yang Y, Chen X, Xie Y, Yao S, Zhao C. Identify a Blood-Brain Barrier Penetrating Drug-TNB using Zebrafish Orthotopic Glioblastoma Xenograft Model. Sci Rep. 2017 Oct 30;7(1):14372. doi: 10.1038/s41598-017-14766-2. PMID: 29085081; PMCID: PMC5662771.
- Chou HL, Lin YH, Liu W, Wu CY, Li RN, Huang HW, Chou CH, Chiou SJ, Chiu CC. Combination Therapy of Chloroquine and C2-Ceramide Enhances Cytotoxicity in Lung Cancer H460 and H1299 Cells. Cancers (Basel). 2019 Mar 15;11(3):370. doi: 10.3390/cancers11030370. PMID: 30884764; PMCID: PMC6468447.
- Cha YR, Weinstein BM. Visualization and experimental analysis of blood vessel formation using transgenic zebrafish. Birth Defects Res C Embryo Today. 2007 Dec;81(4):286-96. doi: 10.1002/bdrc.20103. PMID: 18228261.
- Brown HK, Schiavone K, Tazzyman S, Heymann D, Chico TJ. Zebrafish xenograft models of cancer and metastasis for drug discovery. Expert Opin Drug Discov. 2017 Apr;12(4):379-389. doi: 10.1080/17460441.2017.1297416. PMID: 28277839.
- Lieschke GJ, Trede NS. Fish immunology. Curr Biol. 2009 Aug 25;19(16):R678-82. doi: 10.1016/j.cub.2009.06.068. PMID: 19706273.
- Li XY, Huang LT, Wu JQ, He MF, Zhu SH, Zhan P, Lv TF, Song Y. Zebrafish Xenograft Model of Human Lung Cancer for Evaluating Osimertinib Resistance. Biomed Res Int. 2019 Jun 27;2019:3129748. doi: 10.1155/2019/3129748. PMID: 31346515; PMCID: PMC6620834.
- Lee SL, Rouhi P, Dahl Jensen L, Zhang D, Ji H, Hauptmann G, Ingham P, Cao Y. Hypoxia-induced pathological angiogenesis mediates tumor cell dissemination, invasion, and metastasis in a zebrafish tumor model. Proc Natl Acad Sci U S A. 2009 Nov 17;106(46):19485-90. doi: 10.1073/pnas.0909228106. Epub 2009 Nov 3. PMID: 19887629; PMCID: PMC2780785.
- Ban J, Aryee DN, Fourtouna A, van der Ent W, Kauer M, Niedan S, Machado I, Rodriguez-Galindo C, Tirado OM, Schwentner R, Picci P, Flanagan AM, Berg V, Strauss SJ, Scotlandi K, Lawlor ER, Snaar-Jagalska E, Llombart-Bosch A, Kovar H. Suppression of deacetylase SIRT1 mediates tumor-suppressive NOTCH response and offers a novel treatment option in metastatic Ewing sarcoma. Cancer Res. 2014 Nov 15;74(22):6578-88. doi: 10.1158/0008-5472.CAN-14-1736. Epub 2014 Oct 3. PMID: 25281719.
- Zhang M, Di Martino JS, Bowman RL, Campbell NR, Baksh SC, Simon-Vermot T, Kim IS, Haldeman P, Mondal C, Yong-Gonzales V, Abu-Akeel M, Merghoub T, Jones DR, Zhu XG, Arora A, Ariyan CE, Birsoy K, Wolchok JD, Panageas KS, Hollmann T, Bravo-Cordero JJ, White RM. Adipocyte-Derived Lipids Mediate Melanoma Progression via FATP Proteins. Cancer Discov. 2018 Aug;8(8):1006-1025. doi: 10.1158/2159-8290.CD-17-1371. Epub 2018 Jun 14. PMID: 29903879; PMCID: PMC6192670.
- L SW, Lee CH, Lin MS, Chi CW, Chen YJ, Wang GS, Liao KW, Chiu LP, Wu SH, Huang DM, Chen L, Shen YS. ZnO Nanoparticles Induced Caspase-Dependent Apoptosis in Gingival Squamous Cell Carcinoma through Mitochondrial Dysfunction and p70S6K Signaling Pathway. Int J Mol Sci. 2020 Feb 26;21(5):1612. doi: 10.3390/ijms21051612. PMID: 32111101; PMCID: PMC7084801.
- Van Driessche A, Kocere A, Everaert H, Nuhn L, Van Herck S, Griffiths G, Fenaroli F, De Geest BG. pH-sensitive hydrazone-linked doxorubicin nanogels via polymeric-activated ester scaffolds: Synthesis, assembly, and in vitro and in vivo evaluation in tumor-bearing zebrafish. Chem Mater 2018;30:8587-8596. doi: 10.1021/acs.chemmater.8b03702.
- Welker AM, Jaros BD, An M, Beattie CE. Changes in tumor cell heterogeneity after chemotherapy treatment in a xenograft model of glioblastoma. Neuroscience. 2017 Jul 25;356:35-43. doi: 10.1016/j.neuroscience.2017.05.010. Epub 2017 May 17. PMID: 28526577; PMCID: PMC5555152.
- van der Ent W, Burrello C, Teunisse AF, Ksander BR, van der Velden PA, Jager MJ, Jochemsen AG, Snaar-Jagalska BE. Modeling of human uveal melanoma in zebrafish xenograft embryos. Invest Ophthalmol Vis Sci. 2014 Sep 23;55(10):6612-22. doi: 10.1167/iovs.14-15202. PMID: 25249605.
- Zheng MW, Zhang CH, Chen K, Huang M, Li YP, Lin WT, Zhang RJ, Zhong L, Xiang R, Li LL, Liu XY, Wei YQ, Yang SY. Preclinical Evaluation of a Novel Orally Available SRC/Raf/VEGFR2 Inhibitor, SKLB646, in the Treatment of Triple-Negative Breast Cancer. Mol Cancer Ther. 2016 Mar;15(3):366-78. doi: 10.1158/1535-7163.MCT-15-0501. Epub 2015 Dec 31. PMID: 26721945.
- Peterson RT, Link BA, Dowling JE, Schreiber SL. Small molecule developmental screens reveal the logic and timing of vertebrate development. Proc Natl Acad Sci U S A. 2000 Nov 21;97(24):12965-9. doi: 10.1073/pnas.97.24.12965. PMID: 11087852; PMCID: PMC27161.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.