2025 October 10;6(10):1415-1423. doi: 10.37871/jbres2197.
Molecular Description of the Glideosomatic Complex in the Parasitic Protozoans Toxoplasma and Plasmodium: A Sequence Comparison with Apicomplexans of Interest in the Health of Arthropods of Agricultural Importance
Edison Pascal*, Lenin González-Paz, Aníbal Méndez and Helimar Vásquez
- Glideosoma
- Apicomplexa
- Myosin A (Myo A)
- Gregarins
- Glidant motility
Abstract
This research aimed to molecularly analyze the glideosome complex in the parasites Toxoplasma and Plasmodium, comparing it with apicomplexans of interest in arthropods of agricultural importance (such as gregarines), to identify relevant structural and functional similarities and differences in their motility and potential therapeutic targets. Through a systematic review of databases (PubMed, Google Scholar, NCBI, PDB) and comparative analysis of amino acid sequences, we evaluated whether the molecular conformation of the glideosome in arthropod apicomplexans bears similarities to those of medical relevance. The glideosome, essential for gliding locomotion, a movement without morphological changes that facilitates cell invasion, is composed of proteins such as Myosin A (MyoA), its light chain (MLC1/MTIP), and the proteins GAP45 and GAP50. Post-translationally modified (myristoylation, palmitoylation), the latter act as anchors between the motor complex and the parasite's inner membrane. The study revealed notable differences: Toxoplasma gondii MyoA has 1,810 amino acids, while in Gregarina polymorpha it reaches 2,846, suggesting evolutionary adaptations linked to their niches (invasion of mammalian cells vs. extracellular environments in arthropods). Despite these divergences, conserved domains were identified in MyoA, indicating a common functional core for motility. These findings support that the glideosome is an ancestral system optimized for invasion, but with structural variations that reflect ecological pressures. The comparison among apicomplexans positions this complex as a key model for studying pathogenicity mechanisms and developing broad or specific control strategies, depending on the differences identified. This work underscores the importance of comparative approaches for understanding the evolution of molecular systems in parasites with a medical and agricultural impact.
Introduction
Parasitic protozoans are microparasites (due to their small size, ranging from 5 to 200 µm in length) and reproduce within the host organism. These organisms have various habitats, where symbionts present diverse morphologies, mobility modes, reproductive patterns, and nutritional and metabolic needs. Most of them are motile and heterotrophic, with some organelles absent or modified, and their structure and function vary according to the habitat in different groups [1].
Among the enteroparasitosis that affects humans and animals, those caused by intestinal apicomplexans stand out, which are endoparasitic protozoans. The parasites above have an apical complex that allows them to penetrate the cells (enterocytes) of their hosts; In addition, their biological cycle is notable for being monoxenic with an alternation in their reproductive cycles: schizogony (asexual) and sporogony (sexual), the final stage of which is the oocyst that is excreted in the feces, and some have a zoonotic component [2].
The presence of gregarines of the genera Nematopsis spp., Paraophioidina spp., and Cephalolobus spp., which are Apicomplexan protozoans that infect the intestine of some arthropods of agricultural interest, such as the white shrimp Penaeus vannamei, causing losses in the aquaculture industry. These parasites live in the body cavities of invertebrates, such as arthropods, annelids, and mollusks, feeding by osmosis. Gregarines can be joined end to end (syzygy) or solitary, with the anterior segment known as protomerite and the posterior as satellite [3].
An innovative and largely unknown method of cell movement is the gliding motility exhibited by protozoan parasites of the phylum Apicomplexa. These parasites are of great interest due to their medical relevance as disease-causing agents in humans and animals. For example, Plasmodium is responsible for malaria in humans, while Toxoplasma and Cryptosporidium are significant opportunistic pathogens in immunocompromised individuals. Recent research has shed light on the molecular basis of this mode of movement and has revealed how parasites employ gliding motility to invade host cells [4].
These parasitic protozoans must penetrate the host organism's cells to complete their life cycle. For example, in the case of Plasmodium spp., the apicomplexan must enter the liver and red blood cells of the human host and the midgut of the culicid (mosquito) vector. To carry out this function, the parasite uses an actomyosin mechanism located at the core of a larger structure called the invasion machinery or glideosome [5].
When we talk about parasitic apicomplexans of interest in arthropod health (of agricultural importance), we are referring to intestinal gregarines, which are usually found in the midgut of crustaceans and belong to the Porosporidea family, specifically to the Nematopsis genus. These parasites infect the intestine of penaeid shrimp bred in captivity and in the wild in various parts of the world, harming the production of farmed shrimp [6].
Gregarines are considered the oldest and most fundamental apicomplexans in apicomplexan phylogenies. Although the study of their biology using advanced methods is in its infancy, it is crucial to understand the evolution of parasitism in this group. A comprehensive understanding of aspects such as biology, genetics, host-parasite interactions, and the holobiome of gregarine hosts is necessary to unravel this evolution. Although apicomplexan gregarines are closely related to parasites such as Plasmodium, Toxoplasma, and Cryptosporidium, which have significant impacts on health and the economy, gregarines, because they primarily infect invertebrates and lack direct medical relevance are often overlooked in "omics" studies. However, being the most primitive apicomplexans, they play a fundamental role in understanding the evolution of parasitism in Apicomplexa from free-living ancestors [7,8].
The objective of this research is to describe and molecularly analyze the glideosome complex in the parasitic protozoans Toxoplasma and Plasmodium, by comparing sequences with other apicomplexans of sanitary interest present in arthropods of agricultural importance, such as Gregarinas, to understand structural and functional similarities and differences that may provide knowledge about the motility mechanisms and potential targets for the control of diseases transmitted by these organisms.
Materials and Methods
This article corresponds to an exhaustive bibliographic review, supported by the methodological principles described by Hernández R, et al. [9], who define this type of study as a systematic process of compilation, evaluation, and critical synthesis of scientific information available in bibliographic and newspaper sources. The documentary review is a crucial technique to base research, identify knowledge gaps, and consolidate the state of the art in a specific area.
Research Question Statement
The review aimed to evaluate the following question: Does the molecular conformation of the glideosome complex in apicomplexans of interest in the health of agriculturally important arthropods present significant similarities with that of apicomplexans of medical relevance?
Search Strategy and Sources
A descriptive and comparative approach was adopted to analyze the collected information. To this end, a systematic search was conducted in recognized scientific databases, including PUBMED and Google Scholar, using a search strategy with key terms related to the topic. The keywords used were: "Gliding," "Glideosome," "Apicomplexa," "Gregarins," and "Molecular Functioning in Apicomplexa."
Filters were applied to include only articles published in indexed, high-impact journals, peer-reviewed documents, and relevant texts addressing molecular, structural, and functional aspects of the glideosome complex in apicomplexans. Of the 380 articles returned by the search engines, 31 documents were selected and subjected to a thorough analysis. In addition, the review was complemented by consulting the bibliographic references cited in the selected documents, in order to expand and strengthen the information collection.
Comparative Molecular Analysis
To explore potential molecular similarities between the studied apicomplexans, a comparative analysis of the amino acid sequences of the proteins comprising the glideosome complex was performed. This search was conducted in the NCBI (National Center for Biotechnology Information) database, part of the National Library of Medicine (NIH) [10], and the Protein Data Bank (PDB) [11]. Glideosome-related protein sequences from Plasmodium, Toxoplasma, and representatives of gregarines, apicomplexans that parasitize arthropods and are relevant to public health and aquaculture, were selected.
Results and Discussion
Gliding or sliding movement in apicomplexans of agricultural interest (Gregarinas)
Microscopic observations conducted by the author and other researchers have demonstrated that gregarines, specifically those of the genus Nematopsis, exhibit a "gliding" movement within the intestinal epithelium of shrimp, particularly the farmed white shrimp Penaeus vannamei arthropod of significant agricultural importance in aquaculture [3,4].
Within this conceptual framework, "gliding" can be defined as a form of locomotion in which the apicomplexan parasite moves without altering its apparent shape. This type of movement is crucial for the cellular invasion and propagation of these protozoans and is associated with specialized structures such as the subpellicular cytoskeleton, the conoid, subpellicular microtubules, and the inner membrane complex (IMC). Understanding the structural organization of the parasite, including its cytoskeleton, is essential for elucidating its capacity for invasion and transmission between species. This knowledge, in turn, is critical for the development of effective therapeutic strategies against these parasites [12].
Other studies also confirm that gregarine trophozoites exhibit unidirectional movement, with the protomerite advancing along straight or curved paths depending on the individual observed, while the entire body (deutomerite) follows the same trajectory as the apical protomerite. The movement speed of the trophozoites has been estimated at approximately 60 μm/s. This characterization of trophozoite locomotion provides valuable insights into their behavior and may prove useful in future studies of these parasitic organisms [13].
However, gregarines share similarities with Toxoplasma and Plasmodium, responsible for toxoplasmosis and malaria. These parasites use a set of proteins similar to those used by gregarines to glide toward the cells they infect. This similarity makes them a promising model for investigating their mobility and developing treatments against the parasitic diseases they cause [4].
Gliding facilitates the entry and movement of Apicomplexan parasites between host cells. This type of motility relies on the interaction of actin and myosin, proteins also involved in muscle contraction in humans and other vertebrates. Myosin's molecular structure features "legs" or "appendages" that move along actin filaments, generating movement. In Apicomplexa, myosin combines with various proteins to form the glideosome, a crucial complex whose precise mechanism remains incompletely understood due to limited data on the molecular structure of many of its constituent proteins. However, comprehensive knowledge of this process could be key to developing drugs that interfere with glideosome formation, thereby halting the spread of diseases such as malaria and toxoplasmosis [14].
In this context, we can state that gliding motility is generated by a motor mechanism composed of actin and myosin (the Glideosoma), which is located in the complex cortical domain of the parasite. In this system, myosin is bound to an internal membrane complex and promotes the backward movement of actin-associated cell surface adhesins, resulting in the advancement of the parasite. We can say that this process results in the advancement of the parasite, which allows its movement and infection of host cells. This knowledge about the mechanism of parasite mobility can be useful in the development of therapeutic strategies to combat parasitic diseases and prevent their spread [15].
Unfortunately, our current understanding of the evolution, genetics, and adaptive capacity of Apicomplexa, unicellular parasites that infect a variety of animals, focuses primarily on species that infect humans. This is due to the paucity of data on the earliest diverging lineages of apicomplexans that parasitize invertebrates, including gregarines.
Recently, genome mining has been conducted to identify genes responsible for facilitating gliding, a crucial function in apicomplexan parasites, which is currently being investigated through this molecular model, or glideosome. Sequence analysis reveals that proteins associated with actin and regulators show high conservation across apicomplexans. In contrast, the sequences of predicted core and adhesion glideosome proteins show significant variation among different apicomplexan lineages, especially in gregarines [16].
Within the context of the apicomplexans group of organisms that includes parasites such as Toxoplasma, Plasmodium, and gregarines of aquatic interest significant molecular relationship can be observed among the structures involved in the gliding process. This mode of movement is fundamental to the mobility and invasion of host cells by these parasites.
Molecular structure of the glideosoma
The glideosoma allows apicomplexans to cross biological barriers that are normally prohibited, allowing them to invade host cells actively [17].
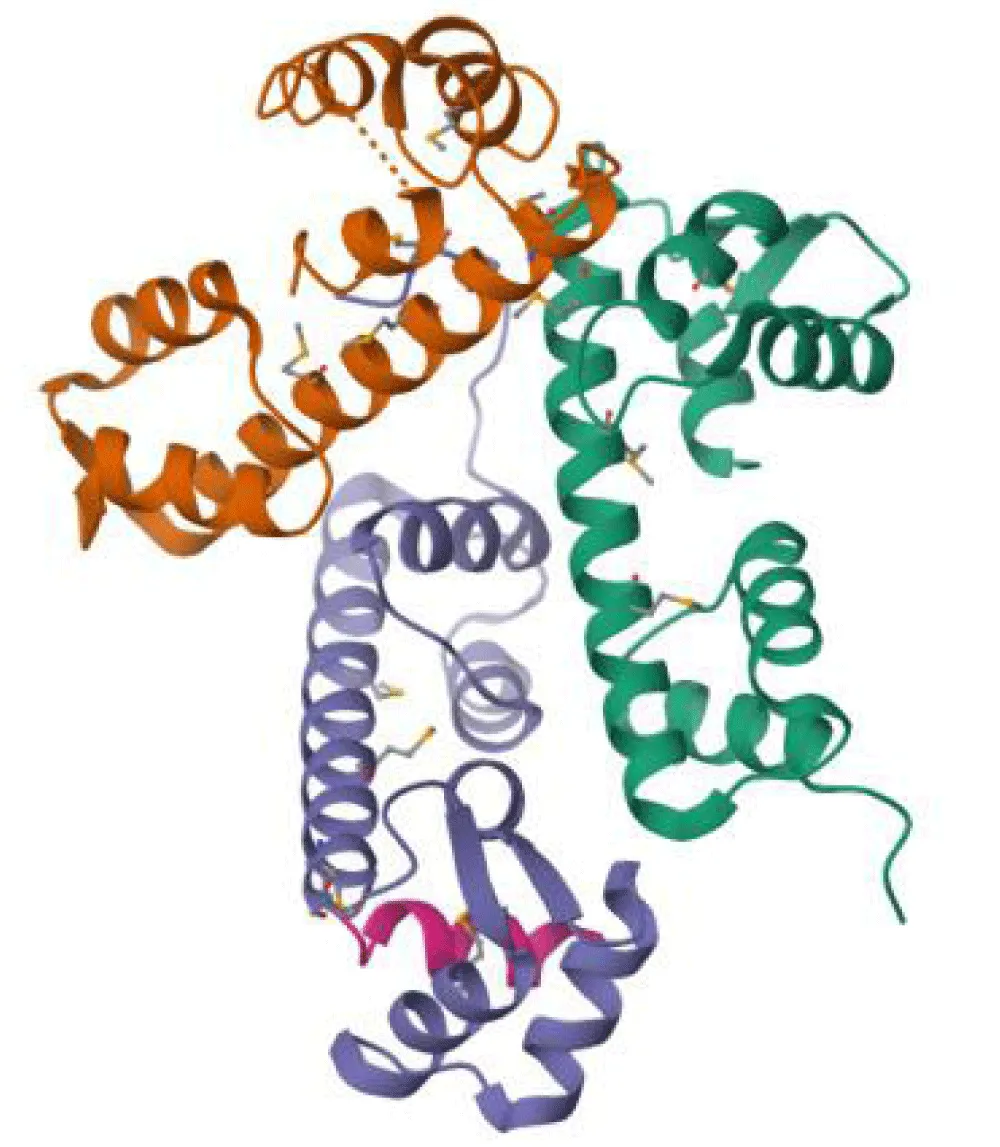
Gliding motility is driven by an actomyosin motor and requires the coordinated action of a complex protein structure known as the "glideosome." Glideosomatic assembly is precisely coordinated in time and space, confined to the narrow region between the Plasma Membrane (PM) and the IMC. This gliding process depends on actin formation and a motor complex composed of class XIV Myosin A (MyoA), its associated myosin light chain 1 (MLC1 or MTIP) (Figure 1), along with two gliding-related proteins, GAP45 and GAP50. GAP45 undergoes modifications by myristoylation and palmitoylation, whereas GAP50 is an integral membrane protein of the BMI, suggesting that both proteins anchor MyoA to the BMI. The preformed MyoA-MLC1-GAP45 complex binds to the IMC through interaction with GAP50 and acylation of GAP45. GAP50 is firmly anchored to the IMC and is postulated to act as a stable anchor point for the dynamic complex [18,19].
In other words, the preassembled MyoA-MLC1-GAP45 complex in apicomplexan protozoans is a protein scaffold essential for gliding motility in these organisms. This complex includes MyoA, the MLC1 protein, and the GAP45 protein. MyoA acts as a motor protein that interacts with actin to move, while MLC1 is a transmembrane protein associated with MyoA. GAP45, meanwhile, is a membrane-adhering protein modified by myristoylation and palmitoylation. Together, this preassembled complex plays an essential role in gliding motility by enabling the interaction between actin filaments and cell surface adhesion proteins, facilitating the efficient transmission of MyoA-generated force to the cell substrate in apicomplexan protozoans.
However, myristoylation and palmitoylation are post-translational protein modification processes that involve the addition of fatty acid chains to cysteine residues in specific proteins. These modifications play a crucial role in protein subcellular localization, stability, and function. Myristoylation involves the addition of a myristoyl group (14 carbon atoms) to the protein, while palmitoylation involves the addition of a palmitoyl group (16 carbon atoms). These processes are important for the proper function of many proteins, including the regulation of cell signaling and protein-lipid interactions in various biological pathways [20,21].
Kumar A, et al. [22] worked with the apicomplexan Toxoplasma gondii, where they established that the Gliding Associated Connector (GAC), is an essential conserved protein important for the function of the glideosome, which facilitates the union of actin filaments with transmembrane adhesion proteins on the cell surface and allows the efficient transmission of the force produced by the displacement of actin by myosin towards the cell substrate.
Dos Santos PN, et al. [23] researched the apicomplexan parasite T. gondii, specifically with the cellular structure called Conoid, said structure or organelle is made up of tubulin fibers and Preconoidal Rings (PCR). It is a dynamic organelle whose function is not clearly defined. Through expanded ultrastructure microscopy, it has been shown that the PCRs act as centers for the components of the Glideososome, including Formin1 (a protein that regulates actin dynamics in eukaryotic cells). Conserved components in Apicomplexans, Pcr4 and Pcr5, have also been identified, which form a heterodimer and are essential for the structure, containing B-box zinc finger domains. The function of Pcr6 is crucial in binding the PCRs to the tubulin fiber cone. Myosin H uses F-actin generated by Formin1 to exert the force necessary for conoid extrusion, directing the flow of F-actin into the pellicular space and thus controlling parasite motility.
Alder A, et al. [24] investigated the role of Essential Light Chains (ELCs) in Toxoplasma gondii and Plasmodium falciparum, resolving their crystal structures as components of trimeric subcomplexes. Their findings revealed that, despite ELCs binding to a conserved MyoA sequence, P. falciparum ELCs adopt distinct structural conformations in their free state compared to their MyoA-bound state. The researchers propose that ELCs optimize MyoA efficiency by modulating its secondary structure, thereby strengthening its lever arm. Structural and biophysical analyses further demonstrated that calcium binding has no discernible impact on ELC conformation. This work marks a critical advancement in elucidating the glideosome-mediated gliding mechanism in apicomplexan protozoans.
Dynamic MyoA-MTIP-GAP motor complex in apicomplexans
The MyoA-MTIP-GAP motor complex (Figure 1) plays a fundamental role in the motility and cellular invasion of apicomplexan parasites. This complex is composed of MyoA, the myosin light chain MTIP (also known as MLC1 in Toxoplasma), and the glideosome-associated proteins GAP45 and GAP50. MTIP is essential for linking the actin-myosin motor to the Inner Membrane Complex (IMC) through its interaction with GAP45, while GAP50 anchors the complex to the IMC membrane. The coordinated interactions among these proteins suggest a high degree of functional conservation of the motor complex in merozoites, contributing significantly to our understanding of apicomplexan invasion and propagation mechanisms [25]. The assembly and function of this molecular motor are tightly regulated by post-translational modifications, particularly phosphorylation and lipid acylation, which are triggered by calcium-dependent signaling cascades. The spatial organization and polymerization of actin filaments are also critical determinants of motility efficiency. Furthermore, the proximity of glycolytic enzymes to the glideosome may enhance local ATP production, ensuring a sufficient energy supply for sustained motor activity [26,27]. Collectively, these findings highlight the intricate regulation and structural organization of the MyoA-MTIP-GAP motor complex, underscoring its central role in the pathogenicity of apicomplexan parasites and its potential as a target for therapeutic intervention.
Baum J, et al. [28] showed that elements of the internal part of the motor complex are found in apicomplexan merozoites. These authors performed immunoprecipitations using radiolabeled schizont samples collected from late-stage cultures (40-48 h post-invasion) with rabbit polyclonal antibodies against PfMTIP. These immunoprecipitations consistently showed protein bands with approximate molecular masses of 90, 45, 40, 35, and 25 kDa. Western blots with subunit-specific antibodies revealed products with expected molecular weights, indicating that these proteins were PfMyoA, PfGAP50, PfGAP45, and PfMTIP, respectively. Using antibodies against PfGAP45 and PfGAP50 in further immunoprecipitations, an interaction was observed with anti-PfMyoA, PfMTIP, and the two PfGAP proteins. Although PfGAP50 precipitation with anti-PfGAP45 was minimal, suggesting a possible instability of the complex under isolation conditions. This lack of precipitation with antibodies against PfMyoA may indicate the need to optimize experimental conditions. Specifically, the interaction between these four components of the motor complex in merozoites supports the functional conservation of the internal portion of this complex under in vivo conditions [29,30].
Comparison of the amino acid sequences of the protein complex constituting the glidesome in apicomplexans
A detailed search was conducted in the National Library of Medicine (NIH) database at the National Center for Biotechnology Information (NCBI) to compare the protein sequences of the glidesome complex in Plasmodium, Toxoplasma, and the Gregarinasin group.
In table 1, it can be observed that the highest percentage of similarity corresponded to Myosin (MyoA, Chain A) of Toxoplasma gondii, comparing the amino acid sequence with Gregarina polymorpha, with 60.16% similarity (Table 1).
| Table 1: Percentages of similarity of the amino acid sequences of the proteins that make up the Glideosoma complex, compared to some Gregarinosins. | |||
| Protein or Structure | Apicomplex of origin | Percentage of identity | Gregarious comparison |
| MyoA | Plasmodium | 54.05% | Gregarina polymorpha |
| Class XIV Miosina | Plasmodium | 54.13% | Gregarina polymorpha |
| Plasmodium | 53.42% | Gregarina niphandrodes | |
| MyoA | Toxoplasma gondii | 0 | 0 |
| MyoA, cadena A | Toxoplasma gondii GTI | 60.16% | Gregarina polymorpha |
| Toxoplasma gondii GTI | 59.37% | Gregarina niphandrodes | |
| GP45 | Plasmodium malariae | 0 | 0 |
| Plasmodium relictum | 0 | 0 | |
| GAP50 | Plasmodium malariae | 38.38% | Gregarina niphandrodes |
| Plasmodium relictum | 38.38% | Gregarina niphandrodes | |
| Plasmodium gallinaceum | 37.57% | Gregarina niphandrodes | |
| Plasmodium ovale | 35.77% | Gregarina niphandrodes | |
| Toxoplasma gondii ME49 | 39.37% | Gregarina niphandrodes | |
| MTIP | Plasmodium knowlesi | 27.47% | Gregarina niphandrodes |
| Toxoplasma | 0 | 0 | |
MASKTTSEELKTATALKKRSSDVHAVDHSGNVYKGFQIWTDLAPSVKEEPDLMFAKCIVQAGTDKGNLTC VQIDPPGFDEPFEVPQANAWNVNSLIDPMTYGDIGMLPHTNIPCVLDFLKVRFMKNQIYTTADPLVVAIN PFRDLGNTTLDWIVRYRDTFDLSKLAPHVFYTARRALDNLHAVNKSQTIIVSGESGAGKTEATKQIMRYF AAAKTGSMDLRIQNAIMAANPVLEAFGNAKTIRNNNSSRFGRFMQLDVGREGGIKFGSVVAFLLEKSRVL TQDEQERSYHIFYQMCKGADAAMKERFHILPLSEYKYINPLCLDAPGIDDVAEFHEVCESFRSMNLTEDE VASVWSIVSGVLLLGNVEVTATKDGGIDDAAAIEGKNLEVFKKACGLLFLDAERIREELTVKVSYAGNQE IRGRWKQEDGDMLKSSLAKAMYDKLFMWIIAVLNRSIKPPGGFKIFMGMLDIFGFEVFKNNSLEQFFINI TNEMLQKNFVDIVFDRESKLYRDEGVSSKELIFTSNAEVIKILTAKNNSVLAALEDQCLAPGGSDEKFLS TCKNALKGTTKFKPAKVSPNINFLISHTVGDIQYNAEGFLFKNKDVLRAEIMEIVQQSKNPVVAQLFAGI VMEKGKMAKGQLIGSQFLSQLQSLMELINSTEPHFIRCIKPNDTKKPLDWVPSKMLIQLHALSVLEALQL RQLGYSYRRPFKEFLFQFKFIDLSASENPNLDPKEAALRLLKSSKLPSEEYQLGKTMVFLKQTGAKELTQ IQRECLSSAAAENLYFQ Amino acid sequence of Gregarina polymorpha of the MyoA, A chain MPPKSKAKAKAKVGANKKLTRPGSKIHALDASGAVFKAFYVWSVDAPIVKKSPDQLYGKCLVLNGSNEDT FKLKQVEPEEDDDSFESSASRCYNANSNIDPMAFPDIGMLPHTNDACVLDYMRARLVMGNIFVTAEPLLV VQNPFKNVGNATDDWIRKYRDVPDVGTLGPHVFSIARTALENLHSVNKSQTIIVSGESGAGKTEATKQAM RFFAAAKSGAMDLRVQKAILAANPVLEAFGNAKTIRNNNSSRFGRYMQLLVAANGGIQNGSVQGFLLEKS RILCQEDNERSYHIFYQILKGVNEDQKKKFKCKTLQQYKYINPKCLDSPGINDLDEWKDVCESFDSMMMS AEERDSVLSICSGVLLMGNVNVVAVAKQGLDDAAEIEEASRATFEDACALMFIDPKAVEEGITVKISIAG GNEIRSVWKKDEGDMLKASLVKGMYDATFQYIIRKLNSNIEPEGGWPAYMGMLDIFGFEVFKNNSLEQLF INVTNEMLQKNFTDTVFERESKLYKDEGIPITDLIFTSNAEVIEALSGRRGSLFATLEDQCLAPGGSDEK FLSAVYSTLKGSTKVYAAKVGGNQNFIVAHTIGEIQYLVEGFLFKNKDVLRAELVEVVQASTNVVSKELF AGVVVEKGKSAKGQLIGSQFMNQLSRLMELINATEAHFIRCVKPNEEKLPLTFTPSKVLIQLQSLSILEA LQLRNLGYSYRRPFSDFLYQYKFVNLGITEDKSLEPMDAAKKLLISSKIPESDWKIGKTMTFMKPAAMKA MQHKQREALASWQPLISLIEALYKRMALKKEYLKLRSGVVRAQALTRRRLAGKIPSPPPDVAVGF
The notable difference in MyoA sequence length between Toxoplasma gondii (1,810 amino acids) and Gregarina polymorpha (2,846 amino acids) suggests that although both proteins belong to the glideosome complex essential for cell motility and penetration, they have undergone distinct structural adaptations. This disparity could reflect specific evolutionary pressures associated with their ecological niches and invasion mechanisms. For example, the larger size of MyoA in Gregarina polymorpha could be linked to the need to optimize motility in extracellular or intertwined environments of arthropods, while the more compact version in T. gondii could favor energetic efficiency during invasion of nucleated mammalian cells [4,18].
Despite these differences, the similarity detected in key domains (such as the myosin motor or actin-binding sites) supports the hypothesis of evolutionary conservation in elements necessary for glideosome assembly and function. This is consistent with previous findings that point to the glideosome complex as a "core molecular module" in Apicomplexa, whose functional core is maintained even in phylogenetically distant groups [5]. The conservation of these regions could explain the ability of both parasites to perform gliding movements, essential for host cell invasion [15].
However, differences in regulatory domains or intrinsically disordered regions (more extensive in gregarins) could modulate interactions with accessory proteins specific to each organism. For example, in T. gondii, MyoA associates with proteins such as the Essential Light Chain protein (ELC) and MTIP (Myosin Tail Interacting Protein), whereas in gregarins, the presence of additional domains could facilitate binding to components of the arthropod extracellular matrix [14,13]. These divergences underscore the plasticity of the glideosome to adapt to different infection scenarios, from insect epithelial tissues to vertebrate immune cells.
Evolutionary and applied implications
The partial conservation of MyoA suggests that the glideosome could be a "hot spot" for the development of broad-spectrum antiparasitic compounds. However, the identified structural differences indicate that therapeutic strategies should consider specific adaptations, such as inhibitors targeting variable domains in gregarines of aquacultural importance (e.g., Nematopsis spp.), without affecting the homologous machinery in medical apicomplexans [26]. Furthermore, this finding reinforces the need for further comparative proteomics and crystallography studies to map critical, commonly conserved regions.
Relationship with previous studies
The results discussed here are consistent with work such as that of Treeck M, et al. [31], who highlighted the conservation of motility-associated signaling pathways in Apicomplexa, but also with recent research identifying differences in the regulation of glideosome assembly between Plasmodium and gregarines [23]. This suggests that, although the functional core of the glideosome is ancestral, its regulation and accessory components have diverged to optimize infection in different hosts.
Limitations and Perspectives
This analysis focused on primary sequences, so future studies should integrate structural (cryo-EM) and functional (in vitro motility assays) data to validate whether length differences directly affect actin-myosin interaction dynamics. It would also be relevant to incorporate comparative transcriptomics to assess whether MyoA variants in gregarins are differentially expressed during specific invasion stages.
Conclusions
Potential similarities among apicomplexans such as Toxoplasma, Plasmodium, and gregarines in their use of a conserved set of proteins to move toward the cells they infect position them as valuable models for studying parasite motility and developing treatments for the diseases they cause. This molecular connection opens new opportunities to deepen our understanding of the biology of these organisms and to identify innovative therapeutic targets, as gliding motility is essential for both cellular invasion and the propagation and virulence of these parasites.
In the specific case of gregarines, parasites relevant to the health of arthropods of agricultural importance, understanding the molecular basis of gliding is crucial for addressing aquatic pathology issues and improving control strategies in aquaculture systems. Identifying similarities in gliding mechanisms between gregarines and other apicomplexans enables the design of more effective interventions to prevent and control infections in these productive systems. Finally, the data reinforce that, although the glideosome is a highly conserved complex within Apicomplexa, its structural and functional variations may be key to understanding differential pathogenicity across diverse hosts, thereby opening opportunities for the design of specific pharmacological strategies tailored to the ecological niche and host of each parasite.
References
- Unzaga J, Zonta M. Protozoos parásitos de importancia sanitaria: un abordaje transdisciplinar. La Plata: Editorial de la Universidad Nacional de La Plata (EDULP). 2023. doi: 10.35537/10915/154565
- Cazorla Perfetti D, Leal Rojas G, Escalona Nelo Á, Hernández Nava J, Acosta Quintero M, Morales Moreno P. Aspectos clínicos y epidemiológicos de la infección por coccidios intestinales en Urumaco, estado Falcón, Venezuela. Bol Mal Salud Ambient. 2014;54(2):159-73.
- Pascal E, Portillo E, Mendez A, Vasquez H. Relationship between Infection Caused by the Apicomplex Protozoan Nematopsis sp and the Weight of White Shrimp Litopenaeus vannamei in a Cultivation System. J Biomed Res Environ Sci. 2023:13;4(10):1405-1411. doi: 10.37871/jbres1810.
- Sibley LD, Hâkansson S, Carruthers VB. Gliding motility: an efficient mechanism for cell penetration. Curr Biol. 1998 Jan 1;8(1):R12-4. doi: 10.1016/s0960-9822(98)70008-9. PMID: 9427622.
- Boucher LE, Bosch J. The apicomplexan glideosome and adhesins - Structures and function. J Struct Biol. 2015 May;190(2):93-114. doi: 10.1016/j.jsb.2015.02.008. Epub 2015 Mar 9. PMID: 25764948; PMCID: PMC4417069.
- Aguado-García N, Cousin J. Relacion longitud-peso y factor de condición del “camarón rosa” Farfantepenaeus paulensis parasitados con Nematopsis paulensis (Protozoa: Apicomplexa: Porosporidae). Boletín del Instituto Oceanográfico de Venezuela. 2004;43(1&2).
- Rueckert S, Betts EL, Tsaousis AD. The Symbiotic Spectrum: Where Do the Gregarines Fit? Trends Parasitol. 2019 Sep;35(9):687-694. doi: 10.1016/j.pt.2019.06.013. Epub 2019 Jul 22. PMID: 31345767.
- Robert-Paganin J, Robblee JP, Auguin D, Blake TCA, Bookwalter CS, Krementsova EB, Moussaoui D, Previs MJ, Jousset G, Baum J, Trybus KM, Houdusse A. Plasmodium myosin A drives parasite invasion by an atypical force generating mechanism. Nat Commun. 2019 Jul 23;10(1):3286. doi: 10.1038/s41467-019-11120-0. PMID: 31337750; PMCID: PMC6650474.
- Hernández R, Fernández C, Baptista P. Metodología de la investigación. Editorial McGraw-Hill; 2014.
- National centre for biotechnology information. Currently Available Genomic Sequences. National Library of Medicine (NIH). 2025.
- Structure of the plasmodium MTIP-MyoA complex, a key component of the parasite invasion motor. Protein Data Bank. 2006.
- Spencer LM, Gómez A, Collovini E. Mecanismos de invasion del esporozoíto y merozoíto de Plasmodium. Bionatura. 2016;1(2):39-44. doi: 10.21931/RB/2016.01.02.9.
- King C, Sleep J. Modelling the mechanism of gregarine gliding using bead translocation. J Eukaryot Microbiol. 2005;52(2):7S-27S. doi: 10.1111/j.1550-7408.2005.05202003_1_41.x.
- Matuschewski K, Schüler H. Actin/myosin-based gliding motility in apicomplexan parasites. Subcell Biochem. 2008;47:110-20. doi: 10.1007/978-0-387-78267-6_9. PMID: 18512346.
- Heintzelman MB. Gliding motility in apicomplexan parasites. Semin Cell Dev Biol. 2015 Oct;46:135-42. doi: 10.1016/j.semcdb.2015.09.020. Epub 2015 Sep 30. PMID: 26428297.
- Boisard J, Duvernois-Berthet E, Duval L, Schrével J, Guillou L, Labat A, Le Panse S, Prensier G, Ponger L, Florent I. Marine gregarine genomes reveal the breadth of apicomplexan diversity with a partially conserved glideosome machinery. BMC Genomics. 2022 Jul 2;23(1):485. doi: 10.1186/s12864-022-08700-8. PMID: 35780080; PMCID: PMC9250747.
- Pazicky S, Dhamotharan K, Kaszuba K, Mertens HDT, Gilberger T, Svergun D, Kosinski J, Weininger U, Löw C. Structural role of essential light chains in the apicomplexan glideosome. Commun Biol. 2020 Oct 13;3(1):568. doi: 10.1038/s42003-020-01283-8. PMID: 33051581; PMCID: PMC7555893.
- Frénal K, Polonais V, Marq JB, Stratmann R, Limenitakis J, Soldati-Favre D. Functional dissection of the apicomplexan glideosome molecular architecture. Cell Host Microbe. 2010 Oct 21;8(4):343-57. doi: 10.1016/j.chom.2010.09.002. PMID: 20951968.
- Wetzel DM, Håkansson S, Hu K, Roos D, Sibley LD. Actin filament polymerization regulates gliding motility by apicomplexan parasites. Mol Biol Cell. 2003 Feb;14(2):396-406. doi: 10.1091/mbc.e02-08-0458. PMID: 12589042; PMCID: PMC149980.
- Chen Y, Li Y, Wu L. Protein S-palmitoylation modification: implications in tumor and tumor immune microenvironment. Front Immunol. 2024 Feb 13;15:1337478. doi: 10.3389/fimmu.2024.1337478. PMID: 38415253; PMCID: PMC10896991.
- Das T, Yount JS, Hang HC. Protein S-palmitoylation in immunity. Open Biol. 2021 Mar;11(3):200411. doi: 10.1098/rsob.200411. Epub 2021 Mar 3. PMID: 33653086; PMCID: PMC8061762.
- Kumar A, Vadas O, Dos Santos Pacheco N, Zhang X, Chao K, Darvill N, Rasmussen HØ, Xu Y, Lin GM, Stylianou FA, Pedersen JS, Rouse SL, Morgan ML, Soldati-Favre D, Matthews S. Structural and regulatory insights into the glideosome-associated connector from Toxoplasma gondii. Elife. 2023 Apr 4;12:e86049. doi: 10.7554/eLife.86049. PMID: 37014051; PMCID: PMC10125020.
- Dos Santos Pacheco N, Brusini L, Haase R, Tosetti N, Maco B, Brochet M, Vadas O, Soldati-Favre D. Conoid extrusion regulates glideosome assembly to control motility and invasion in Apicomplexa. Nat Microbiol. 2022 Nov;7(11):1777-1790. doi: 10.1038/s41564-022-01212-x. Epub 2022 Sep 15. PMID: 36109645.
- Alder A, Wilcke L, Pietsch E, von Thien H, Pazicky S, Löw C, Mesen-Ramirez P, Bachmann A, Burda PC, Kunick C, Sondermann H, Wilson D, Gilberger TW. Functional inactivation of Plasmodium falciparum glycogen synthase kinase GSK3 modulates erythrocyte invasion and blocks gametocyte maturation. J Biol Chem. 2022 Sep;298(9):102360. doi: 10.1016/j.jbc.2022.102360. Epub 2022 Aug 10. PMID: 35961464; PMCID: PMC9478393.
- Guerra Vega AP. Estudio de la miosina B de Plasmodium falciparum y su posible papel en la invasión del parásito al glóbulo rojo [doctoral thesis]. 2019.
- Daher W, Soldati-Favre D. Mechanisms controlling glideosome function in apicomplexans. Curr Opin Microbiol. 2009 Aug;12(4):408-14. doi: 10.1016/j.mib.2009.06.008. Epub 2009 Jul 3. PMID: 19577950.
- Santos JM, Lebrun M, Daher W, Soldati D, Dubremetz JF. Apicomplexan cytoskeleton and motors: key regulators in morphogenesis, cell division, transport and motility. Int J Parasitol. 2009 Jan;39(2):153-62. doi: 10.1016/j.ijpara.2008.10.007. Epub 2008 Nov 6. PMID: 19028497.
- Baum J, Papenfuss AT, Baum B, Speed TP, Cowman AF. Regulation of apicomplexan actin-based motility. Nat Rev Microbiol. 2006 Aug;4(8):621-8. doi: 10.1038/nrmicro1465. PMID: 16845432.
- Quevillon E, Spielmann T, Brahimi K, Chattopadhyay D, Yeramian E, Langsley G. The Plasmodium falciparum family of Rab GTPases. Gene. 2003 Mar 13;306:13-25. doi: 10.1016/s0378-1119(03)00381-0. PMID: 12657463.
- Chattopadhyay N, Wang Z, Ashman LK, Brady-Kalnay SM, Kreidberg JA. alpha3beta1 integrin-CD151, a component of the cadherin-catenin complex, regulates PTPmu expression and cell-cell adhesion. J Cell Biol. 2003 Dec 22;163(6):1351-62. doi: 10.1083/jcb.200306067. PMID: 14691142; PMCID: PMC2173722.
- Treeck M, Sanders JL, Elias JE, Boothroyd JC. The phosphoproteomes of Plasmodium falciparum and Toxoplasma gondii reveal unusual adaptations within and beyond the parasites' boundaries. Cell Host Microbe. 2011 Oct 20;10(4):410-9. doi: 10.1016/j.chom.2011.09.004. PMID: 22018241; PMCID: PMC3254672.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.