Medicine Group. 2024 September 30;5(9):1238-1252. doi: 10.37871/jbres2011.
Unplanned Excision of Soft Tissue Sarcoma - The Impact of Residual Disease in Re-Excision Specimen
Julien Montreuil MD-MSc1, Moses Markowitz MD2, Eric Kholodovsky2, Sergio J. Torralbas Fitz1, Dominic Campano MD1, Erik J. Geiger MD1, Francis Hornicek MD-PHD1, Brooke Crawford MD1 and H. Thomas Temple MD1
2University of Miami Miller School of Medicine, USA
Abstract
Background: This study aims to assess the oncologic outcomes of unplanned versus planned excision in patients with high-grade soft tissue sarcoma.
Methods: A retrospective review was conducted, including 147 patients surgically treated for non-metastatic high-grade soft tissue sarcoma at a single institution between 2010 and 2021. Perioperative, clinical, and surveillance data were collected. Outcomes were compared between patients with unplanned (n = 53) and planned (n = 94) excisions. Multivariate analyses controlled for tumor characteristics and adjuvant therapies.
Results: The cohort included 53 patients treated with unplanned excision and 94 patients with planned excision, with a mean follow-up of 65.3 months postoperatively. Demographic variables, tumor size, type, histological grade, and margin status were statistically comparable between the planned and unplanned excision groups based on univariate analysis. Unplanned excisions were associated with significantly higher rates of local recurrence (39.6% vs 7.4%, p < 0.001) and metastasis (18.9% vs 5.3%, p = 0.009) compared to planned excisions. On multivariate analysis, planned excision conferred a decreased risk for local recurrence (HR 0.12, 95% CI 0.05-0.31, p < 0.001) and metastasis (OR 0.22, 95% CI 0.05-1.02, p = 0.05). Final positive margins were strongly associated with local recurrence (HR 18.26, 95% CI 6.80-48.99, p < 0.001). Over 90% of re-excision specimens after unplanned excision contained residual disease. Patients with microscopic or macroscopic disease identified in the re-excision specimen had a 42% and 44% risk of local recurrence, respectively, in comparison to those patients with no residual disease identified as having no incidence of local recurrence.
Conclusion: This study highlights the importance of a proper primary excision by an orthopedic oncologist for patients with high-grade soft tissue sarcoma. Unplanned excisions often result in tumor beds with either gross or residual microscopic disease and have a significantly increased risk of local recurrence, distant metastasis, and shortened survival.
Introduction
Soft tissue sarcomas are rare tumors that are associated with significant morbidity and mortality. Despite being rare, in 2021 alone, 13,400 people in the United States were newly diagnosed [1]. At ten years, 25% of patients develop metastases with an overall survival of 70% following treatment consisting of surgery and radiotherapy with or without chemotherapy [2]. Tumor grade and stage impact patient survival [3-6], whereas radiation therapy and surgical margin status affect local recurrence [7-9]. Local recurrence increases disease morbidity, often requiring re-excision, additional radiotherapy, soft tissue reconstructive procedures and sometimes amputation; however, the relationship between local recurrence and overall survival is unclear.
Unplanned excision of soft tissue sarcomas occurs in 18-49% of patients [10-12]. When patients with soft tissue sarcomas undergo excision without adequate pre-operative clinical and radiologic evaluation and discussion in a Multidisciplinary Tumor Board (MTB), inadequate excision margins, wound complications, and contamination of normal adequate tissue are potential consequences. Potter, et al. [13] retrospectively assessed the outcomes of patients with high-grade soft tissue sarcoma and demonstrated that unplanned excisions resulted in increased rates of local recurrence but had no significant effect on disease-specific survival. Liang Y, et al. [14] demonstrated similar results even when controlling for various baseline covariates (including size, depth, and grade). Other studies have demonstrated worse oncologic outcomes in unplanned excision despite subsequent tumor bed excision [9,11,15,16]. Zaidi, et al. [17] in a multi-institutional study of patients with high-grade soft tissue sarcomas, concluded that unplanned excision resulted in higher rates of local recurrence but did not appear to affect overall survival compared to a cohort who had planned excision. Most studies suggest a higher rate of local recurrence in the setting of unplanned excision but do not uniformly control for tumor size, grade, stage, and the effect of adjuvant therapies.
The purpose of this study is to assess the oncologic outcomes of unplanned versus planned excision in patients with high-grade soft tissue sarcomas utilizing a multivariant analysis to control for tumor characteristics and the presence or absence of gross or microscopic residual disease after unplanned excision.
Methods
This is a retrospective cohort study of 147 consecutive patients treated surgically for non-metastatic high-grade soft tissue sarcoma of the extremities at the same institution from 2010 to 2021. High-grade was defined as grade 2 or 3 (based on mitotic activity, necrosis and tumor differentiation). Tumors in the pelvis, retroperitoneum, head, and neck were excluded. Patients with metastatic disease at presentation, low-grade sarcoma, and patients managed nonoperatively were also excluded from this study. The study was approved by the institutional review board. Unplanned excision was defined as excision of a soft tissue mass with or without appropriate imaging studies but without prior biopsy or review by a multidisciplinary panel. Planned excision was defined as surgical excision following appropriate staging, biopsy, and multidisciplinary review.
Demographics and Clinical Data
Demographic and clinical data were collected for each patient, including age, gender, race, ethnicity, Body Mass Index (BMI), presence of diabetes, smoking status, treatment with preoperative or postoperative chemotherapy, and radiotherapy in the form of external beam radiation or brachytherapy. Additionally, tumor characteristics were recorded for each patient as documented by a trained and experienced musculoskeletal pathologist, including tumor type, size, grade, location viability, tumor size, excision margins, and the presence of gross or microscopic tumor in the final specimen in the context of a previously unplanned excision. Patients with synovial sarcoma, clear-cell sarcoma, angiosarcoma, rhabdomyosarcoma, and epithelioid sarcoma (SCARE) were identified and specifically assessed, as these subtypes are most likely to metastasize to locoregional lymph nodes [18]. For patients with these subtypes, treatment involved sentinel node biopsy for any lymph node that was clinically or radiographically suspicious. Patients with Myxofibrosarcoma (MFS) were also assessed with subgroup analysis due to the high risk of local recurrence in these patients [19]. Percent viability of the excised tumor was grouped by < 5%, 5-50%, and > 50% as previously described by Lucas DR, et al. [20]. The need for complex soft tissue reconstructive procedures, including locoregional or free flaps, was documented in the case of large soft tissue defects after the excision. Any postoperative complications, including wound dehiscence, deep infection, deep vein thrombosis, or pulmonary embolism, were documented. Clinical outcomes were surveyed and documented by the primary musculoskeletal oncology attending surgeon including local recurrence, distant metastasis, treatment-related complications and mortality.
Statistical Analysis
Descriptive statistics were utilized to compare planned and unplanned excision of soft tissue sarcomas and provide a comprehensive overview of patient demographics, tumor characteristics, and treatment modalities. Quantitative variables were expressed as mean and standard deviations or median and interquartile ranges depending on their distribution. Categorical variables were expressed as absolute values or percentages. Comparative analyses, including chi-square or Fisher's exact tests and t-tests or Mann-Whitney U tests, were applied to explore associations and differences in categorical and continuous variables, respectively. Subgroup analyses further stratified the data based on tumor subtype, grade, and patient demographics to investigate potential variations on the impact of unplanned excision. Multivariate regression, using Cox models and logistic regression, were utilized to identify independent predictors of overall survival and assess factors associated with specific outcomes. Survival analysis techniques, such as Kaplan-Meier curves, log-rank tests, and Cox Proportional-Hazards models, were employed to estimate overall survival rates and assess differences between patients with and without unplanned excision, while adjusting for relevant covariates. Data were analyzed using STATA version 18.0 (STATA Corp, USA) and R version 3.5.1 (R Foundation for Statistical Computing). Statistical significance was defined as a p value less than 0.05.
Results
The study comprised 147 participants: 53 individuals (36%) undergoing re-excision for unplanned surgeries and 94 (64%) undergoing planned wide excision. The mean age at the time of surgery was comparable between the unplanned and planned groups; 54.60 (SD = 15.69) and 54.49 (SD = 17.79) years, respectively (p = 0.94). Gender distribution also showed no significant difference, with 58.5% males and 41.5% females in the unplanned group, and 55.3% males and 44.7% females in the planned group (p = 0.71). Body Mass Index (BMI) was not significantly different between the two groups, with mean values of 28.24 (SD = 6.162) for the unplanned group and 28.62 (SD = 7.200) for the planned group (p = 0.74). Similarly, the prevalence of diabetes was not significantly different, with rates of 30.2% in the unplanned group and 20.2% in the planned group (p = 0.17) (Table 1).
| Table 1: Descriptive statistics. | ||||
| Variable | Total n = 147 Mean (std) / n (%) | Unplanned n = 53 Mean (std) / n (%) | Planned n = 94 Mean (std) / n (%) | p-value |
| Age | 54.5(17.0) | 54.6(15.7) | 54.5(17.8) | 0.94 |
| Gender | 0.71 | |||
| M | 83(56.5) | 31(58.5) | 52(55.3) | |
| F | 64(43.5) | 22(41.5) | 42(44.7) | |
| BMI | 28.5(6.8) | 28.2(6.2) | 28.6(7.2) | 0.74 |
| Diabetes | 35(23.8) | 16(30.2) | 19(20.2) | 0.17 |
| Size | 0.41 | |||
| < 5 cm | 26(17.7) | 11(20.8) | 15(16.0) | |
| 5-10 cm | 55(37.4) | 22(41.5) | 33(35.1) | |
| > 10 cm | 66(44.9) | 20(37.7) | 46 (48.9) | |
| Depth | < 0.001 | |||
| Deep | 45(30.6) | 5(9.4) | 40(42.6) | |
| Superficial | 102(69.4) | 48(90.6) | 54(57.4) | |
| Specific types | 0.23 | |||
| SCARE | 27(18.4) | 13(24.5) | 14(14.9) | |
| MFS | 27(18.4) | 11(20.8) | 16(17.0) | |
| Grade | 0.41 | |||
| II | 42(28.6) | 13(24.5) | 29(30.9) | |
| III | 105(71.4) | 40(75.5) | 65(69.1) | |
| Viability | 0.23 | |||
| < 5% | 24(16.3) | 5(9.4) | 19(20.2) | |
| 5-50% | 96(65.3) | 38(71.7) | 58(61.7) | |
| > 50% | 27(18.4) | 10(18.9) | 17(18.1) | |
| Fungating | 0.75 | |||
| Yes | 12(8.2) | 5(9.6) | 7(7.4) | |
| Margins | 0.28 | |||
| Positive | 9(6.1) | 5(9.4) | 4(4.3) | |
| Residual Disease | ||||
| None | 5(9.4) | N/A | ||
| Microscopic | N/A | 12(22.6) | N/A | |
| Macroscopic | N/A | 36(67.9) | N/A | |
| Radiation | ||||
| Any EBRT | 110(74.8) | 36(67.9) | 74(78.7) | 0.15 |
| Brachy | 46(31.3) | 20(37.7) | 26(27.7) | 0.21 |
| Chemo | ||||
| Neoadjuvant Chemo | 60(40.8) | 19(35.8) | 41(43.6) | 0.36 |
| Soft tissues | ||||
| Complex Recon | 20(13.6) | 13(24.5) | 7(7.4) | 0.004 |
| Wound complication | 58(39.5) | 20(37.6) | 38(40.4) | 0.75 |
| SSI | 21(14.3) | 7(13.2) | 14(14.9) | 0.78 |
| I&D needed | 23(15.6) | 6(11.3) | 17(18.1) | 0.10 |
| Recurrence | ||||
| LR | 28 (19.0) | 21 (39.6) | 7(7.4) | < 0.001 |
| Metastasis | 15 (10.2) | 10(18.9) | 5(5.3) | 0.009 |
| Status | < 0.001 | |||
| NED | 112(76.2) | 31(58.5) | 81(86.2) | |
| AWD | 13(8.8) | 11(20.8) | 2(2.1) | |
| DOD | 10(6.8) | 7(13.2) | 3(3.2) | |
| DOC | 12(8.2) | 4(7.5) | 8(8.5) | |
| Follow-up (Months) | ||||
| 65.3(31.9) | 66.3(30.9) | 64.7(32.5) | 0.78 | |
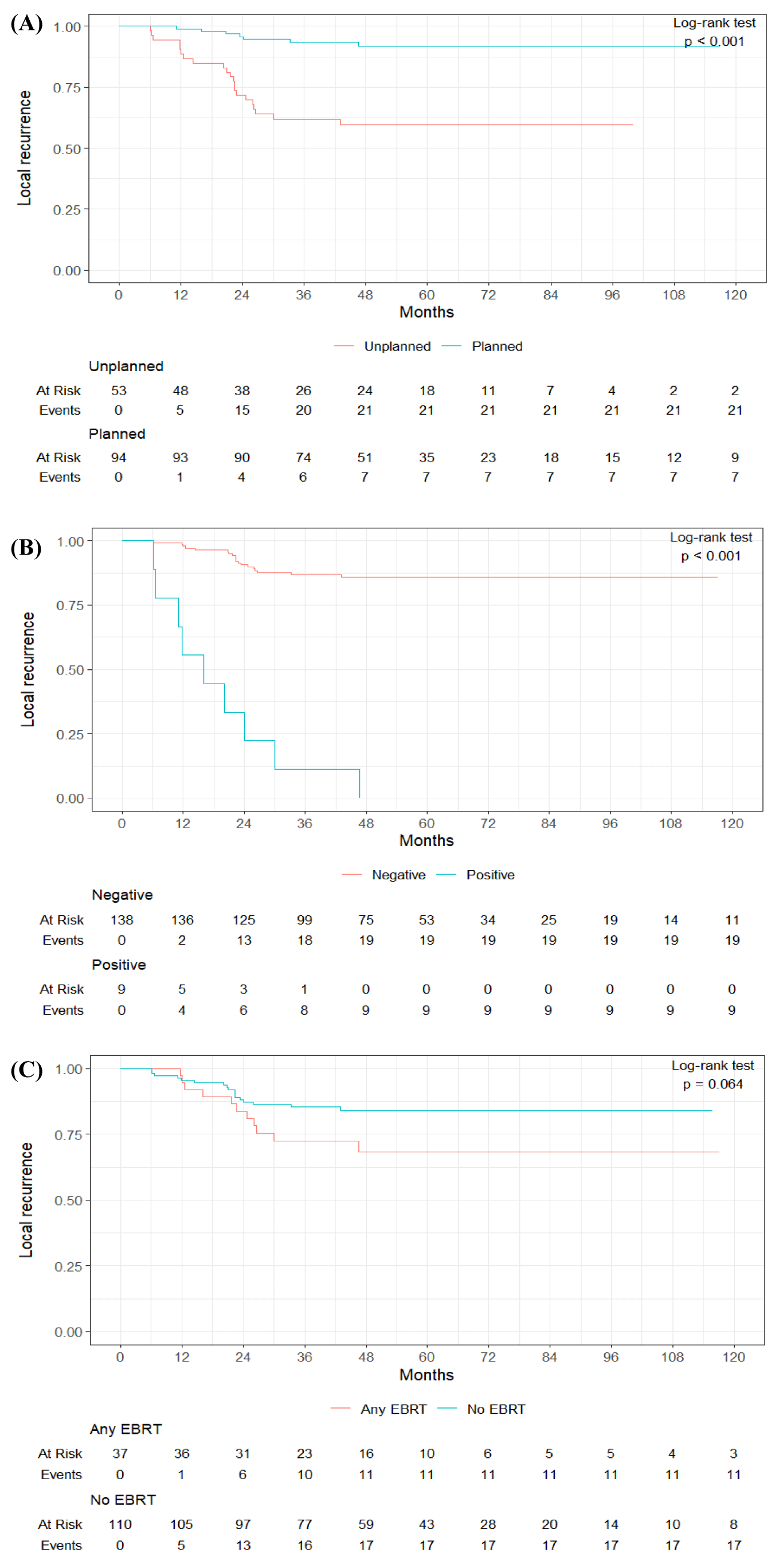
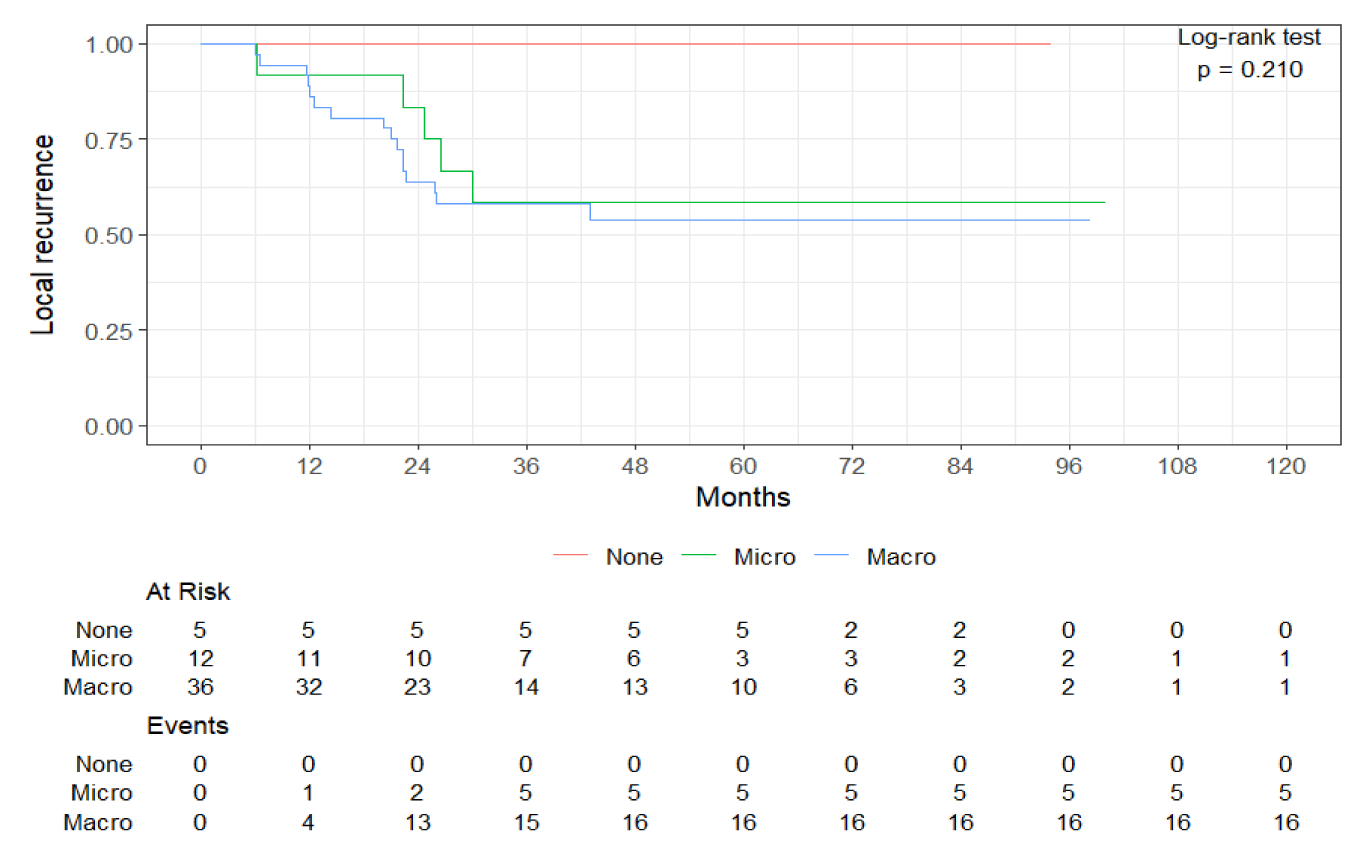
Tumor size, categorized as less than 5 cm, 5-10 cm, and more than 10 cm, did not show significant differences between unplanned and planned surgeries (p = 0.41). Depth of tumor location, however, demonstrated a significant distinction (p < 0.001), with only 9.4% of the unplanned group having deep seated tumors compared to 42.6% in the planned group. 90.6% of unplanned excisions were performed for superficial tumors. The distribution of special tumor types (SCARE and MFS) and tumor grade (2 and 3) did not reveal significant differences between the unplanned and planned groups (p = 0.23 and p = 0.41, respectively). Viability, categorized as < 5%, 5-50%, and > 50%, also did not differ significantly between the groups (p = 0.23). Fungating tumors were observed in 9.6% of the unplanned group and 7.4% of the planned group, with no significant difference (p = 0.75). Margins status showed no significant difference between groups (p = 0.28), with 9.4% positive margins in the unplanned group at the time of re-excision and 4.3% in the planned group. Residual disease in re-excision specimens of unplanned surgeries was microscopic in 22.6% of cases, and macroscopic in 67.9%. The presence of residual disease after unplanned excision significantly increases the risk of local recurrence. Patients with microscopically involved margins in the re-excision specimen had a 42% LR, and the same risk was 44% when the macroscopic disease was identified in the re-excised specimen. In comparison, of the patients without residual disease in tumor bed at the time of the re-excision, none had a local recurrence. All patients with unplanned excision of myxofibrosarcoma had residual disease in the re-excised specimen. The incidence of local recurrence for that group was 55% (n = 6).
Regarding adjuvant therapies, there was no significant difference in treatment with External Beam Radiation Therapy (EBRT) between the two groups (p = 0.15), with 67.9% in the unplanned group and 78.7% in the planned group receiving this treatment, though the timing of radiotherapy as well as the choice of total radiation dose and field of therapy were different in those treated with prior unplanned excision. Treatment with brachytherapy did not show a significant difference when comparing patients treated with unplanned excision to planned (p = 0.21). Treatment with neoadjuvant chemotherapy also was not significantly different between groups (p = 0.36), with 35.8% in the unplanned group and 43.6% in the planned group receiving neoadjuvant chemotherapy.
In the unplanned group, 24.5% of cases required complex soft tissue closure and/or reconstruction compared to 7.4% in the planned group (p = 0.004). 37.6% of the unplanned surgery re-excision group experienced a wound complication, while the planned surgery group had a slightly higher rate of 40.4%, with no statistically significant difference observed (p = 0.75). More specifically, 13.2% of the unplanned group and 14.9% of the planned group experienced surgical site infection (SSI) (p = 0.78).
Recurrence patterns, including Local Recurrence (LR) and metastasis, demonstrated significant differences (LR; p < 0.001, Mets; p = 0.009) between the unplanned and planned groups, with higher rates in the unplanned group (LR: 39.6%, Mets: 18.9%) compared to the planned group (LR: 7.4%, Mets: 5.3%). The overall status of participants at the last follow-up demonstrated significant differences (p < 0.001) in the distribution of participants categorized as No Evidence of Disease (NED), Alive With Disease (AWD), Dead Of Disease (DOD), or Dead of Other Causes (DOC) between the unplanned and planned groups. The average follow-up duration in months did not significantly differ between the two groups, with 66.27 months (SD = 30.86) for the unplanned group, 64.73 months (SD = 32.54) for the planned group, p-(p = 0.78.)
Univariate analysis demonstrated that unplanned excisions are associated with a significantly higher risk of local recurrence compared to planned excisions. (Hazard Ratio HR= 0.15, 95% [0.06, 0.36], p < 0.001) Tumor size did not demonstrate a significant association with local recurrence (p = 0.32) Deep-seated tumors had a significant association with lower local recurrence risk compared to superficial tumors (HR = 6.45, 95% C.I. [1.53, 27.21], p = 0.01). The type of STS showed no significant association with local recurrence (p = 0.17). Tumor grade (grade II or III) did not significantly correlate with local recurrence (p = 0.69). Tumor viability, stratified into the following groups: 5%, 5-50%, and more than 50%, did not exhibit a significant association with local recurrence (p = 0.12). Fungating tumors were associated with a higher risk of local recurrence (HR = 3.91, 95% C.I. [1.58, 9.69], p = 0.003).
Positive margins significantly increased the risk of local recurrence. (HR = 17.3, 95% C.I. [7.7, 39.0], p < 0.001). The use of External Beam Radiation Therapy (EBRT) compared to no radiation approached significance in demonstrating a protective effect of adjuvant radiation on local recurrence (HR = 0.50, 95% C.I. [0.23, 1.06], p = 0.07). There was no significant difference between brachytherapy and EBRT in local recurrence rates in the cohort of 46 patients treated with brachytherapy and 110 treated with EBRT. Neoadjuvant chemotherapy did not have a significant effect on local recurrence (p = 0.24). The presence of Surgical Site Infections (SSI), wound complications, and the need for complex initial reconstructive strategies did not show significant associations with local recurrence (Respectively: p = 0.58, p = 0.36, p = 0.25) (Table 2).
| Table 2: Local recurrence univariate regression. | ||||
| Variable | HR | 95% C.I | p-value | |
| Setting | < 0.001 | |||
| Planned | 0.15 | 0.06 | 0.36 | |
| Size | ||||
| < 5 cm | 1 | 0.32 | ||
| 5-10 cm | 1.41 | 0.38 | 5.22 | |
| > 10 cm | 2.23 | 0.65 | 7.65 | |
| Depth | 0.01 | |||
| Deep | 1 | |||
| Superficial | 6.45 | 1.53 | 27.21 | |
| Type of STS | 0.17 | |||
| SCARE | 1 | |||
| MFS | 4.26 | 0.90 | 20.1 | |
| Others | 2.67 | 0.62 | 11.5 | |
| Grade | 0.69 | |||
| II | 1 | |||
| III | 0.85 | 0.38 | 1.88 | |
| Viability | 0.12 | |||
| < 5% | 1 | |||
| 5-50% | 1.53 | 0.53 | 4.43 | |
| > 50% | 0.21 | 0.02 | 1.84 | |
| Fungating | 0.003 | |||
| Yes | 3.91 | 1.58 | 9.69 | |
| Margins | < 0.001 | |||
| Positive | 17.3 | 7.7 | 39.0 | |
| Radiation | ||||
| EBRT vs none | 0.50 | 0.23 | 1.06 | 0.07 |
| Brachy vs EBRT | 0.87 | 0.38 | 1.97 | 0.74 |
| Chemotherapy | ||||
| Any Chemo | 0.88 | 0.40 | 1.94 | 0.75 |
| Soft tissues | ||||
| Complex reconstruction | 1.77 | 0.67 | 4.67 | 0.25 |
| SSI | 0.72 | 0.22 | 2.37 | 0.58 |
| Wounds | 1.41 | 0.67 | 2.97 | 0.36 |
In the multivariable Cox regression analysis, we explored the independent associations of several key variables with the hazard of local recurrence in this context. When controlling for grade, size and utilization of radiation therapy, or chemotherapy, the status of surgical margins was a robust predictor of the event, with a substantial hazard ratio of 18.26 (95% CI: 6.80; 48.99) for cases with positive margins compared to those with negative margins (p < 0.001). The type of excision, whether unplanned or planned, also demonstrated a significant impact on the hazard. Planned excisions had a HR of 0.12 (95% CI: 0.05; 0.31) compared to unplanned excisions (p < 0.001), indicating a substantially lower risk of LR in cases where excisions were planned when controlling for all other variables (Table 3).
| Table 3: Local recurrence multivariate regression model. | ||||
| VARIABLE | HR | (95%CI) | p-value | |
| Grade | 2 | 1 | 0.18 | |
| 3 | 0.53 | ( 0.21;1.33) | ||
| Size | < 5 cm | 1 | 0.44 | |
| 5-10 cm | 0.85 | ( 0.22;3.34) | ||
| > 10 cm | 1.57 | ( 0.40;6.19) | ||
| Margins (0,1) | No | 1 | < 0.001 | |
| Yes | 18.26 | ( 6.80;48.99) | ||
| Unplanned vs Planned Excision (0,1) | 0 | 1 | < 0.001 | |
| 1 | 0.12 | ( 0.05;0.31) | ||
| Any external beam radiation | No | 1 | 0.59 | |
| Yes | 0.80 | ( 0.36;1.80) | ||
| Any Chemotherapy | No | 1 | 0.31 | |
| Yes | 1.60 | ( 0.65;3.95) | ||
In the multivariable Cox regression analysis for metastasis occurrence, the type of excision, whether unplanned or planned, emerged as a significant predictor. Planned excisions had an odds ratio (OR) of 0.22 (95% CI: 0.05; 1.02) compared to unplanned excisions (p = 0.05), suggesting a substantially lower likelihood of metastasis in cases where excisions were planned. The status of surgical margins was also a robust predictor of metastasis, with cases having positive margins exhibiting a strikingly high odds ratio of 65.56 (95% CI: 8.27; 519.89) compared to cases with negative margins (p < 0.001) (Table 4) (Figure 1(A-C)).
| Table 4: Metastasis multivariate analysis. | ||||
| VARIABLE | OR | (95%CI) | p-value | |
| Unplanned vs Planned Excision (0,1) | 0 | 1 | 0.05 | |
| 1 | 0.22 | ( 0.05;1.02) | ||
| Grade | 2 | 1 | 0.11 | |
| 3 | 0.31 | ( 0.07;1.30) | ||
| Depth (Deep = 1, Superficial = 2) | 1 | 1 | 0.58 | |
| 2 | 1.89 | ( 0.20;18.21) | ||
| Margins | Negative | 1 | < 0.001 | |
| Positive | 65.56 | ( 8.27;519.89) | ||
| Size | Less than 5 cm | 1 | 0.89 | |
| Between 5 and 10 cm | 0.70 | ( 0.10;5.10) | ||
| Greater than 10 cm | 1.01 | ( 0.15;6.56) | ||
The surgical setting emerged as a significant predictor of disease-specific survival. Patients who underwent planned excisions had an HR of 0.24 (95% CI: 0.06; 0.93) compared to those in unplanned settings (p = 0.04). Larger tumor sizes were associated with higher hazards of disease-specific mortality. Specifically, tumors larger than 10 cm had 7 mortality events and tumors larger then 5 cm had 3 events. With a HR of 1.1 (95% CI: 1.00; 1.20), indicating a trend toward increased mortality risk compared to smaller tumors p = 0.04). The type of STS did not show a statistically significant association with disease-specific survival. Tumor grade did not exhibit a significant association with disease-specific survival. Tumor viability at the time of excision did not significantly impact disease-specific survival (p = 0.62). The presence of fungating tumors however significantly influenced disease-specific survival (p < 0.001). Fungating tumors had a substantially higher HR of 8.66 (95% CI: 2.42; 30.92) compared to non-fungating tumors. The status of surgical margins significantly impacted disease-specific survival (p < 0.001). Tumors with positive margins had a substantially higher HR of 24.13 (95% CI: 6.77; 86.06) compared to those with negative margins. Neither External Beam Radiation (EBRT) nor chemotherapy, whether neoadjuvant or combined, showed statistically significant associations with disease-specific survival (p > 0.05) (Table 5) (Figure 2).
| Table 5: Overall disease-specific survival univariate analysis. | ||||
| Variable | HR | 95% C.I | p-value | |
| Setting | 0.04 | |||
| Planned | 0.24 | 0.06 | 0.93 | |
| Size | ||||
| 1.1 | 1.00 | 1.20 | 0.04 | |
| > 5 cm | 3 events | |||
| > 10 cm | 7 events | |||
| Type of STS | 0.62 | |||
| SCARE | 1 | |||
| MFS | 2.35 | 0.24 | 22.69 | |
| Others | 1.26 | 0.15 | 10.54 | |
| Grade | 0.50 | |||
| II | 1 | |||
| III | 1.44 | 0.53 | 3.91 | |
| Viability | 0.62 | |||
| Less 5 | 1 | |||
| 5-50 | 2.80 | 0.35 | 22.18 | |
| More 50 | 0.00 | |||
| Fungating | < 0.001 | |||
| Yes | 8.66 | 2.42 | 30.92 | |
| Margins | < 0.001 | |||
| Positive | 24.13 | 6.77 | 86.06 | |
| Radiation | ||||
| EBRT vs none | 1.45 | 0.31 | 6.87 | 0.64 |
| Chemo | ||||
| Neoadjuvant | 0.66 | 0.17 | 2.54 | 0.54 |
| Combined | 1.96 | 0.42 | 9.24 | 0.40 |
| Soft tissues | ||||
| Complex ST | 2.93 | 0.61 | 13.98 | 0.18 |
| Subsequent Sx | 19.1 | 2.42 | 150.83 | 0.005 |
| Metastasis | < 0.001 | |||
| Presence | 68.51 | 8.67 | 541.48 | |
Discussion
The impact of a previous unplanned excision on the local recurrence, distant metastasis, and overall survival following re-excision is a subject of great interest in musculoskeletal oncology. This study assessed the impact of various tumor and treatment characteristics using a multivariate analysis on these outcome measures in patients with high-grade soft tissue sarcomas.
The literature is equivocal regarding the impact of unplanned excision on the risk of local recurrence. Studies by Qureshi YA, et al. [11] and Potter BK, et al. [13] demonstrate in their respective cohorts that patients treated with unplanned excision of a soft tissue sarcoma are at an increased risk of local recurrence, while Sacchetti F, et al. [21], showed that treatment following unplanned excision does not independently impact the risk of local recurrence. This meta-analysis included soft tissue sarcomas of various grades and did not explicitly control for tumor size and depth in its primary analyses, though the authors did acknowledge these factors as potential sources of bias in their discussion. In the current study cohort, 53 cases had unplanned excision, and they had a significantly increased risk of local recurrence when controlling for tumor grade, size, margins at the time of re-excision, and utilization of radiation therapy. The variability present in the literature may be due to several factors. The geographic and institutional rates of unplanned excision vary widely. Relatively low rates of unplanned excision in some series may influence the analysis of differences in outcomes between planned and unplanned patient cohorts. Tumor and patient variability and failure to control covariates may conspire to mask real differences between these patient groups. In this study cohort, there were significantly more superficial tumors in the unplanned group than in the planned excision cohort. This observation is not surprising and has been attributed to the surgeon performing the initial procedure more often mistaking these more superficial tumors to be benign and performing, in hindsight, an unsatisfactory excision [9,10,22,23]. This is notable as these more superficial tumors excised by non-orthopedic oncology surgeons were associated in this study with an increased risk of recurrence (p = 0.01). In this cohort, the increased risk of recurrence in patients with unplanned excisions is also related to the extent of the previous excision and residual tumor cells in the tumor bed. Also, 67.9% of patients treated with unplanned excision had residual macroscopic disease, and 22.6% had residual microscopic disease in the tumor bed, amounting to over 90% of patients with evidence of residual disease at the time of re-excision. This can be considered in the context of other studies that demonstrate 47 to 70% of patients with residual disease at the time of re-excision in the tumor bed of a patient that underwent previously unplanned excision [13, 24-27]. Patients with microscopic residual disease at the time of re-excision have an almost 50% risk of local recurrence. The current study demonstrated that the risk of local recurrence is increased by previously unplanned excision, particularly when controlling for tumor grade, size, and utilization of radiation therapy. In this cohort, having postoperative SSI, wound complications, or the need for complex reconstructive strategies did not pose an increased risk of local recurrence. This is in contrast to other investigations where patients with wound-related complications have increased tumor-related events [28,29]. These studies, however, do not control for variables such as tumor size, depth, grade, and the utilization of radiotherapy that may confound this association. Additionally, in this study cohort, patients treated previously with unplanned excision for MFS demonstrated a 55% risk of local recurrence despite a wide margin re-excision. Special considerations must be made for these patients when considering re-excision and radiotherapy regimens to address the increased risk of local recurrence.
In the current cohort, 10.2% of all patients developed metastatic disease, consistent with the literature demonstrating the risk of metastatic disease after oncologic excision for soft tissue sarcoma ranging from 15% to 24% in cohorts of patients not restricted by tumor grade [11,23,24,30,31]. What is surprising is that in patients with previously unplanned excision, there was a significantly increased incidence of metastatic disease (18.9%) compared to planned excision (5.3%). The effect of a previous unplanned excision on metastasis is controversial, with some studies interestingly demonstrating a decreased risk of metastasis when these tumors are initially excised in an unplanned procedure [14,32]. This protective effect of an unplanned excision demonstrated must be understood in consideration of the characteristics of the patients making up each study group. Studies such as those by Fiore M, et al. [33] and Lewis JJ, et al. [34] demonstrated protective effects of a previous unplanned excision. However, in the study by Fiore M, et al. [33] the groups of patients treated with a primary planned excision had higher grade tumors and deeper tumors, both well-documented significant predictors of distant metastasis [35-37]. Similarly, in the study by Lewis JJ, et al. [34], significantly more patients had negative margins and smaller tumors when treated with a re-excision procedure than when treated with one planned procedure. Negative margins and smaller size tumors have also been identified as predictors of a reduced risk of distant metastasis [5,37,38]. In this study cohort, there was no significant difference in size, grade, or surgical margins between patients treated with re-excision versus those treated with a planned primary excision. Additionally, size, grade, surgical margins, and depth were controlled for in multivariate analysis. Thus, even when controlling for these tumor characteristics, prior unplanned excision of soft tissue sarcoma is associated with an increased risk of metastasis. Notably, in this cohort of patients treated with re-excision, over 90% of patients had evidence of residual disease at the time of the re-excision procedure. Evidence of residual disease has been demonstrated to increase the risk of distant metastasis in various studies [23,39]. Thus, the increased risk of distant metastasis in patients with a prior unplanned excision may be attributable to the significant proportion of patients with active residual disease in the tumor bed. To understand the true cause and effect relationship that an unplanned excision has on distant metastasis, large prospective studies should be performed. When patients present with a high likelihood of residual disease following an unplanned excision, the increased risk of metastasis must be considered.
The effect of a previously unplanned excision on overall survival also remains controversial [9,13,34]. In this study, the disease-specific mortality at 5 years is 3.2% after planned excision compared to 13.2% after excision for an unplanned excision. The significant detriment of a previously unplanned excision on disease-specific survival demonstrated in this study, just as in the increased risk of metastasis in this study, may be explained by the large proportion of patients with residual disease at the time of re-excision. Various retrospective studies have demonstrated residual disease to be associated with worse overall survival [13,39,40]. Fortunately, recent treatment regimens have improved disease and disease-free survival, therefore limiting the ability to have statistical analysis based on large numbers of deaths. Additional large studies are warranted to more accurately assess the impact of unplanned excision on disease-specific survival, including multivariate analyses to control for disease characteristics as confounding variables.
In tandem with surgical treatment, adjuvant radiotherapy remains standard practice in treating high-grade soft tissue sarcoma. Various studies demonstrated that adjuvant radiation decreases local recurrence in patients with high-grade sarcoma without a significant effect on overall survival [8,41]. Some retrospective analyses, as well as early randomized trials, suggest that when the primary surgery results in negative margins, radiotherapy does not confer a reduction in the risk of recurrence [42-45]. Thus, in the current cohort of patients treated both with unplanned and planned excisions, with nearly 96% treated with negative surgical margins, the effect of adjuvant radiation in reducing local recurrence approached but did not achieve significance (p = .07). This study additionally assessed postoperative brachytherapy as an adjuvant treatment modality. The cohort demonstrated that brachytherapy compared to external beam therapy was no different when considering the risk of local recurrence (p = .74), similar to various retrospective and prospective trials that have validated it in reducing local recurrence [46,47]. Brachytherapy offers a shorter, lower cost radiotherapy treatment that reduces local recurrence similarly to external beam radiotherapy [46,48]. With a rapid dose drop from the site of treatment, brachytherapy should be considered to spare normal tissue, while external beam therapy can cover a larger volume with increased homogeneity. Various studies have assessed the impact of brachytherapy on postoperative complications [47,49]. The effect of brachytherapy on these postoperative complications still requires further research, while controlling for various confounding factors that may contribute to each complication. Additionally, in MFS, radiotherapy requires larger fields due to the proximal and distal tumor extension commonly seen in this disease [50,51]. For this reason, our group does not recommend utilization of brachytherapy in patients with this sarcoma subtype. Though the efficacy of brachytherapy was assessed as a secondary objective in this study, it should be assessed in a larger cohort study with more detailed statistical analysis targeted at this specific technique.
The use of chemotherapy in the treatment of soft tissue sarcoma has been limited to patients presenting with large high-grade tumors or in the context of metastatic disease. The efficacy of systemic chemotherapy in reducing local recurrence, distant metastasis, and improving survival, is still debated [52-55]. This study indicates no significant benefit of chemotherapy administration and local disease recurrence risk (p = .75) or overall survival (p = 0.40). Randomized prospective trials including large patient cohorts are warranted to evaluate the utility of modern chemotherapy regimens for various types of soft tissue sarcoma.
The effect of a previously unplanned excision on wound-related complications is an important consideration. In a retrospective series of 67 patients treated with re-excision for unplanned excision of soft tissue sarcoma by Traweek RS, et al. [56], 45% of patients experienced wound complications, 15% of which being considered major wound complications. In this study, the cohort of patients with previous unplanned excision more often required a complex soft tissue reconstruction including skin grafts and tissue flaps, however, there was no significant effect on wound-related complications or surgical site infections. This is likely influenced by the cohort of patients treated with re-excision having more superficial tumors, likely increasing the need for locoregional flaps. Additionally, having more superficial and smaller tumors in the unplanned cohort may reduce any added risk of seroma or deep wound related complications. Further studies are warranted to more closely evaluate the effect of unplanned excision on soft tissue complications and assess any added risk that postoperative external beam radiotherapy versus brachytherapy may introduce for wound related complications in various regimens.
There are several limitations in this study that must be considered. The study cohort includes only patients from a single sarcoma center, therein limiting the generalizability to all institutions. Additionally, during the study period, some variables in hospital practice and treatment protocols could have changed. Finally, the cohort assessed in this study is small in number, and a larger cohort would be beneficial, specifically when assessing the influence of more rare events such as mortality, brachytherapy treatment and certain sarcoma subtypes of interest like myxofibrosarcoma.
The study cohort of patients with unplanned excision truly highlights the significance of residual disease at the time of re-excision when considering any additional risk associated with unplanned excision. Identification of patients with residual disease is crucial, as these patients in our study, are at an increased risk of local recurrence, distant metastasis, and worse overall survival. Thus, treatment should be individualized and optimized for these patients. Patients with an unplanned excision with either microscopic or macroscopic residual disease had greater than a 40% incidence of local recurrence compared to patients with no residual disease at the time of re-excision who had no cases of local recurrence. The findings of this study underscore the critical need for continued education and improved clinical practices regarding soft tissue sarcomas. Future efforts should focus on developing comprehensive educational initiatives for non-oncology surgeons. These programs should emphasize the recognition of suspicious soft tissue masses, and the risks associated with unplanned excisions. Concurrently, optimizing referral pathways from primary care and non-oncology specialties to sarcoma centers is crucial. This could involve implementing alerts and establishing electronic platforms for immediate consultation and rapid access clinics for urgent evaluations of patients with uncertain soft tissue tumors. Enhancing multidisciplinary collaboration through regular tumor boards that include community surgeons can foster better communication and awareness. Finally, developing and widely disseminating updated best practice guidelines and protocols for managing soft tissue masses, with an emphasis on pre-excisional biopsy and imaging, can be helpful in reducing the incidence of unplanned excisions and improving patient outcomes.
Conclusion
This study highlights the importance of a proper primary excision by an orthopedic oncologist for patients with high-grade soft tissue sarcoma. Unplanned excisions often result in tumor beds with either gross or residual microscopic disease and have a significantly higher risk of local recurrence and poorer overall clinical outcomes.
References
- Society AC. About soft tissue sarcoma. 2021.
- Posch F, Leitner L, Bergovec M, Bezan A, Stotz M, Gerger A, Pichler M, Stöger H, Liegl-Atzwanger B, Leithner A, Szkandera J. Can Multistate Modeling of Local Recurrence, Distant Metastasis, and Death Improve the Prediction of Outcome in Patients With Soft Tissue Sarcomas? Clin Orthop Relat Res. 2017 May;475(5):1427-1435. doi: 10.1007/s11999-017-5232-x. Epub 2017 Jan 12. PMID: 28083752; PMCID: PMC5384928.
- Callegaro D, Miceli R, Bonvalot S, Ferguson PC, Strauss DC, van Praag VVM, Levy A, Griffin AM, Hayes AJ, Stacchiotti S, Pèchoux CL, Smith MJ, Fiore M, Tos APD, Smith HG, Catton C, Szkandera J, Leithner A, van de Sande MAJ, Casali PG, Wunder JS, Gronchi A. Development and external validation of a dynamic prognostic nomogram for primary extremity soft tissue sarcoma survivors. EClinicalMedicine. 2019 Nov 22;17:100215. doi: 10.1016/j.eclinm.2019.11.008. PMID: 31891146; PMCID: PMC6933187.
- Fan Z, Chi C, Tong Y, Huang Z, Song Y, You S. Score for the Risk and Overall Survival of Lung Metastasis in Patients First Diagnosed With Soft Tissue Sarcoma: A Novel Nomogram-Based Risk Assessment System. Technol Cancer Res Treat. 2022 Jan-Dec;21:15330338211066240. doi: 10.1177/15330338211066240. PMID: 35006028; PMCID: PMC8753250.
- LeVay J, O'Sullivan B, Catton C, Bell R, Fornasier V, Cummings B, Hao Y, Warr D, Quirt I. Outcome and prognostic factors in soft tissue sarcoma in the adult. Int J Radiat Oncol Biol Phys. 1993 Dec 1;27(5):1091-9. doi: 10.1016/0360-3016(93)90529-5. PMID: 8262833.
- Pisters PW, Leung DH, Woodruff J, Shi W, Brennan MF. Analysis of prognostic factors in 1,041 patients with localized soft tissue sarcomas of the extremities. J Clin Oncol. 1996 May;14(5):1679-89. doi: 10.1200/JCO.1996.14.5.1679. PMID: 8622088.
- Bilgeri A, Klein A, Lindner LH, Nachbichler S, Knösel T, Birkenmaier C, Jansson V, Baur-Melnyk A, Dürr HR. The Effect of Resection Margin on Local Recurrence and Survival in High Grade Soft Tissue Sarcoma of the Extremities: How Far Is Far Enough? Cancers (Basel). 2020 Sep 8;12(9):2560. doi: 10.3390/cancers12092560. PMID: 32911853; PMCID: PMC7563962.
- Saxby NE, An Q, Miller BJ. Local Recurrence of Soft Tissue Sarcoma Revisited: Is there a Role for "Selective" Radiation? Iowa Orthop J. 2022 Jun;42(1):239-248. PMID: 35821921; PMCID: PMC9210402.
- Larios F, Gonzalez MR, Ruiz-Arellanos K, Aquilino E Silva G, Pretell-Mazzini J. Is Unplanned Excision of Soft Tissue Sarcomas Associated with Worse Oncological Outcomes?-A Systematic Review and Meta-Analysis. Cancers (Basel). 2024 Jan 19;16(2):443. doi: 10.3390/cancers16020443. PMID: 38275885; PMCID: PMC10814439.
- Smolle MA, Tunn PU, Goldenitsch E, Posch F, Szkandera J, Bergovec M, Liegl-Atzwanger B, Leithner A. The Prognostic Impact of Unplanned Excisions in a Cohort of 728 Soft Tissue Sarcoma Patients: A Multicentre Study. Ann Surg Oncol. 2017 Jun;24(6):1596-1605. doi: 10.1245/s10434-017-5776-8. Epub 2017 Jan 20. PMID: 28108827; PMCID: PMC5413518.
- Qureshi YA, Huddy JR, Miller JD, Strauss DC, Thomas JM, Hayes AJ. Unplanned excision of soft tissue sarcoma results in increased rates of local recurrence despite full further oncological treatment. Ann Surg Oncol. 2012 Mar;19(3):871-7. doi: 10.1245/s10434-011-1876-z. Epub 2011 Jul 27. PMID: 21792512.
- Gage MM, Nagarajan N, Ruck JM, Canner JK, Khan S, Giuliano K, Gani F, Wolfgang C, Johnston FM, Ahuja N. Sarcomas in the United States: Recent trends and a call for improved staging. Oncotarget. 2019 Mar 29;10(25):2462-2474. doi: 10.18632/oncotarget.26809. PMID: 31069009; PMCID: PMC6497437.
- Potter BK, Adams SC, Pitcher JD Jr, Temple HT. Local recurrence of disease after unplanned excisions of high-grade soft tissue sarcomas. Clin Orthop Relat Res. 2008 Dec;466(12):3093-100. doi: 10.1007/s11999-008-0529-4. Epub 2008 Sep 26. PMID: 18818981; PMCID: PMC2628223.
- Liang Y, Guo TH, Xu BS, Hong DC, Qiu HB, Zhou ZW, Zhang X. The Impact of Unplanned Excision on the Outcomes of Patients With Soft Tissue Sarcoma of the Trunk and Extremity: A Propensity Score Matching Analysis. Front Oncol. 2021 Jan 22;10:617590. doi: 10.3389/fonc.2020.617590. PMID: 33552992; PMCID: PMC7862753.
- Saeed H, King DM, Johnstone CA, Charlson JA, Hackbarth DA, Neilson JC, Bedi M. Preoperative Radiation Therapy Followed by Reexcision May Improve Local Control and Progression-Free Survival in Unplanned Excisions of Soft Tissue Sarcomas of the Extremity and Chest-Wall. Int J Surg Oncol. 2016;2016:5963167. doi: 10.1155/2016/5963167. Epub 2016 Oct 10. PMID: 27803813; PMCID: PMC5075633.
- Shemesh SS, Garbrecht EL, Rutenberg TF, Conway SA, Rosenberg AE, Pretell-Mazzini J. Unplanned excision of soft tissue sarcoma: does it impact the accuracy of intra-operative pathologic assessment at time of re-excision? Int Orthop. 2021 Nov;45(11):2983-2991. doi: 10.1007/s00264-021-05187-y. Epub 2021 Aug 20. PMID: 34415419.
- Zaidi MY, Ethun CG, Liu Y, Poultsides G, Howard JH, Mogal H, Tseng J, Votanopoulos K, Fields RC, Cardona K. The impact of unplanned excisions of truncal/extremity soft tissue sarcomas: A multi-institutional propensity score analysis from the US Sarcoma Collaborative. J Surg Oncol. 2019 Sep;120(3):332-339. doi: 10.1002/jso.25521. Epub 2019 Jun 6. PMID: 31172536.
- Gilbert NF, Cannon CP, Lin PP, Lewis VO. Soft-tissue sarcoma. J Am Acad Orthop Surg. 2009 Jan;17(1):40-7. doi: 10.5435/00124635-200901000-00006. PMID: 19136426.
- Yurtbay A, Coşkun HS, Say F, Dabak N. Is the Thickness of the Margin Associated With Local Recurrence and Survival in Patients With Myxofibrosarcoma? Clin Orthop Relat Res. 2023 Nov 1;481(11):2125-2136. doi: 10.1097/CORR.0000000000002709. Epub 2023 May 29. PMID: 37249339; PMCID: PMC10567052.
- Lucas DR, Kshirsagar MP, Biermann JS, Hamre MR, Thomas DG, Schuetze SM, Baker LH. Histologic alterations from neoadjuvant chemotherapy in high-grade extremity soft tissue sarcoma: clinicopathological correlation. Oncologist. 2008 Apr;13(4):451-8. doi: 10.1634/theoncologist.2007-0220. PMID: 18448561.
- Sacchetti F, Alsina AC, Morganti R, Innocenti M, Andreani L, Muratori F, Scoccianti G, Totti F, Campanacci DA, Capanna R. Re-excision after unplanned excision of soft tissue sarcoma: A systematic review and metanalysis. The rationale of systematic re-excision. J Orthop. 2021 May 14;25:244-251. doi: 10.1016/j.jor.2021.05.022. PMID: 34099954; PMCID: PMC8165293.
- Dyrop HB, Safwat A, Vedsted P, Maretty-Kongstad K, Hansen BH, Jørgensen PH, Baad-Hansen T, Keller J. Characteristics of 64 sarcoma patients referred to a sarcoma center after unplanned excision. J Surg Oncol. 2016 Feb;113(2):235-9. doi: 10.1002/jso.24137. Epub 2016 Jan 18. PMID: 26776152.
- Fiore M, Casali PG, Miceli R, Mariani L, Bertulli R, Lozza L, Collini P, Olmi P, Mussi C, Gronchi A. Prognostic effect of re-excision in adult soft tissue sarcoma of the extremity. Ann Surg Oncol. 2006 Jan;13(1):110-7. doi: 10.1245/ASO.2006.03.030. Epub 2006 Jan 1. PMID: 16372156.
- Chandrasekar CR, Wafa H, Grimer RJ, Carter SR, Tillman RM, Abudu A. The effect of an unplanned excision of a soft-tissue sarcoma on prognosis. J Bone Joint Surg Br. 2008 Feb;90(2):203-8. doi: 10.1302/0301-620X.90B2.19760. PMID: 18256089.
- Scoccianti G, Innocenti M, Frenos F, Muratori F, Sacchetti F, Beltrami G, Capanna R, Campanacci DA. Re-excision after unplanned excision of soft tissue sarcomas: Long-term results. Surg Oncol. 2020 Sep;34:212-217. doi: 10.1016/j.suronc.2020.04.026. Epub 2020 Apr 27. PMID: 32891333.
- Gingrich AA, Elias A, Michael Lee CY, Nakache YN, Li CS, Shah DR, Boutin RD, Canter RJ. Predictors of residual disease after unplanned excision of soft tissue sarcomas. J Surg Res. 2017 Feb;208:26-32. doi: 10.1016/j.jss.2016.08.096. Epub 2016 Sep 8. PMID: 27993214; PMCID: PMC5180207.
- Mihara A, Iwanaga R, Muramatsu K, Ihara K, Sakai T. Oncological and functional outcomes of planned and unplanned excision of soft tissue sarcoma: A retrospective study. J Orthop Sci. 2023 Jul;28(4):867-873. doi: 10.1016/j.jos.2022.04.007. Epub 2022 Apr 28. PMID: 35491297.
- Nakamura T, Matsumine A, Matsubara T, Asanuma K, Uchida A, Sudo A. Clinical characteristics of patients with large and deep soft tissue sarcomas. Oncol Lett. 2015 Aug;10(2):841-844. doi: 10.3892/ol.2015.3289. Epub 2015 May 28. PMID: 26622580; PMCID: PMC4509109.
- Van Geel AN, Eggermont AM, Hanssens PE, Schmitz PI. Factors Influencing Prognosis After Initial Inadequate Excision (IIE) for Soft Tissue Sarcoma. Sarcoma. 2003;7(3-4):159-65. doi: 10.1080/13577140310001650321. PMID: 18521381; PMCID: PMC2395531.
- Noria S, Davis A, Kandel R, Levesque J, O'Sullivan B, Wunder J, Bell R. Residual disease following unplanned excision of soft-tissue sarcoma of an extremity. J Bone Joint Surg Am. 1996 May;78(5):650-5. doi: 10.2106/00004623-199605000-00003. PMID: 8642020.
- Decanter G, Stoeckle E, Honore C, Meeus P, Mattei JC, Dubray-Longeras P, Ferron G, Carrere S, Causeret S, Guilloit JM, Fau M, Rosset P, Machiavello JC, Delhorme JB, Regenet N, Gouin F, Blay JY, Coindre JM, Penel N, Bonvalot S. Watch and Wait Approach for Re-excision After Unplanned Yet Macroscopically Complete Excision of Extremity and Superficial Truncal Soft Tissue Sarcoma is Safe and Does Not Affect Metastatic Risk or Amputation Rate. Ann Surg Oncol. 2019 Oct;26(11):3526-3534. doi: 10.1245/s10434-019-07494-6. Epub 2019 Jul 23. PMID: 31338771.
- Alsina AC, Sacchetti F, Kaya H, Yaman B, Tamsel İ, Sabah D. Impact of the unplanned excision on the oncological outcomes of patients with soft tissue sarcomas: a single-center retrospective review of 490 patients. Acta Orthop Traumatol Turc. 2022 Jul;56(4):272-277. doi: 10.5152/j.aott.2022.21373. PMID: 35968619; PMCID: PMC9612672.
- Fiore M, Ford S, Callegaro D, Sangalli C, Colombo C, Radaelli S, Frezza AM, Renne SL, Casali PG, Gronchi A. Adequate Local Control in High-Risk Soft Tissue Sarcoma of the Extremity Treated with Surgery Alone at a Reference Centre: Should Radiotherapy Still be a Standard? Ann Surg Oncol. 2018 Jun;25(6):1536-1543. doi: 10.1245/s10434-018-6393-x. Epub 2018 Feb 22. PMID: 29470819.
- Lewis JJ, Leung D, Espat J, Woodruff JM, Brennan MF. Effect of reresection in extremity soft tissue sarcoma. Ann Surg. 2000 May;231(5):655-63. doi: 10.1097/00000658-200005000-00005. PMID: 10767786; PMCID: PMC1421052.
- Stefanovski PD, Bidoli E, De Paoli A, Buonadonna A, Boz G, Libra M, Morassut S, Rossi C, Carbone A, Frustaci S. Prognostic factors in soft tissue sarcomas: a study of 395 patients. Eur J Surg Oncol. 2002 Mar;28(2):153-64. doi: 10.1053/ejso.2001.1242. PMID: 11884051.
- von Konow A, Ghanei I, Styring E, Vult von Steyern F. Late Local Recurrence and Metastasis in Soft Tissue Sarcoma of the Extremities and Trunk Wall: Better Outcome After Treatment of Late Events Compared with Early. Ann Surg Oncol. 2021 Nov;28(12):7891-7902. doi: 10.1245/s10434-021-09942-8. Epub 2021 Apr 16. PMID: 33861406; PMCID: PMC8519908.
- Takemori T, Kawamoto T, Hara H, Fukase N, Fujiwara S, Kitayama K, Yahiro S, Miyamoto T, Mifune Y, Hoshino Y, Kakutani K, Matsumoto T, Matsushita T, Niikura T, Kuroda R, Akisue T. Clinical Outcomes and Prognostic Factors in Soft Tissue Sarcoma Patients After Unplanned Excision. Cancer Manag Res. 2022 May 25;14:1815-1824. doi: 10.2147/CMAR.S364912. PMID: 35642242; PMCID: PMC9148585.
- Zagars GK, Ballo MT, Pisters PW, Pollock RE, Patel SR, Benjamin RS, Evans HL. Prognostic factors for patients with localized soft-tissue sarcoma treated with conservation surgery and radiation therapy: an analysis of 1225 patients. Cancer. 2003 May 15;97(10):2530-43. doi: 10.1002/cncr.11365. PMID: 12733153.
- Bianchi G, Sambri A, Cammelli S, Galuppi A, Cortesi A, Righi A, Caldari E, Ferrari S, Donati D. Impact of residual disease after "unplanned excision" of primary localized adult soft tissue sarcoma of the extremities: evaluation of 452 cases at a single Institution. Musculoskelet Surg. 2017 Dec;101(3):243-248. doi: 10.1007/s12306-017-0475-y. Epub 2017 Apr 25. PMID: 28444540.
- Rehders A, Stoecklein NH, Poremba C, Alexander A, Knoefel WT, Peiper M. Reexcision of soft tissue sarcoma: sufficient local control but increased rate of metastasis. World J Surg. 2009 Dec;33(12):2599-605. doi: 10.1007/s00268-009-0262-5. Erratum in: World J Surg. 2010 Aug;34(8):1991. PMID: 19838751.
- Yang JC, Chang AE, Baker AR, Sindelar WF, Danforth DN, Topalian SL, DeLaney T, Glatstein E, Steinberg SM, Merino MJ, Rosenberg SA. Randomized prospective study of the benefit of adjuvant radiation therapy in the treatment of soft tissue sarcomas of the extremity. J Clin Oncol. 1998 Jan;16(1):197-203. doi: 10.1200/JCO.1998.16.1.197. PMID: 9440743.
- Khanfir K, Alzieu L, Terrier P, Le Péchoux C, Bonvalot S, Vanel D, Le Cesne A. Does adjuvant radiation therapy increase loco-regional control after optimal resection of soft-tissue sarcoma of the extremities? Eur J Cancer. 2003 Sep;39(13):1872-80. doi: 10.1016/s0959-8049(03)00426-x. PMID: 12932665.
- Baldini EH, Goldberg J, Jenner C, Manola JB, Demetri GD, Fletcher CD, Singer S. Long-term outcomes after function-sparing surgery without radiotherapy for soft tissue sarcoma of the extremities and trunk. J Clin Oncol. 1999 Oct;17(10):3252-9. doi: 10.1200/JCO.1999.17.10.3252. PMID: 10506627.
- Pisters PW, Pollock RE, Lewis VO, Yasko AW, Cormier JN, Respondek PM, Feig BW, Hunt KK, Lin PP, Zagars G, Wei C, Ballo MT. Long-term results of prospective trial of surgery alone with selective use of radiation for patients with T1 extremity and trunk soft tissue sarcomas. Ann Surg. 2007 Oct;246(4):675-81; discussion 681-2. doi: 10.1097/SLA.0b013e318155a9ae. PMID: 17893504.
- Ahmad R, Jacobson A, Hornicek F, Haynes AB, Choy E, Cote G, Nielsen GP, Chen YL, DeLaney TF, Mullen JT. The Width of the Surgical Margin Does Not Influence Outcomes in Extremity and Truncal Soft Tissue Sarcoma Treated With Radiotherapy. Oncologist. 2016 Oct;21(10):1269-1276. doi: 10.1634/theoncologist.2015-0534. Epub 2016 Jul 20. PMID: 27440063; PMCID: PMC5061529.
- Naghavi AO, Fernandez DC, Mesko N, Juloori A, Martinez A, Scott JG, Shah C, Harrison LB. American Brachytherapy Society consensus statement for soft tissue sarcoma brachytherapy. Brachytherapy. 2017 May-Jun;16(3):466-489. doi: 10.1016/j.brachy.2017.02.004. Epub 2017 Mar 23. PMID: 28342738.
- Neugebauer J, Blum P, Keiler A, Süß M, Neubauer M, Moser L, Dammerer D. Brachytherapy in the Treatment of Soft-Tissue Sarcomas of the Extremities-A Current Concept and Systematic Review of the Literature. Cancers (Basel). 2023 Feb 10;15(4):1133. doi: 10.3390/cancers15041133. PMID: 36831476; PMCID: PMC9954233.
- Janjan NA, Yasko AW, Reece GP, Miller MJ, Murray JA, Ross MI, Romsdahl MM, Oswald MJ, Ochran TG, Pollock RE. Comparison of charges related to radiotherapy for soft-tissue sarcomas treated by preoperative external-beam irradiation versus interstitial implantation. Ann Surg Oncol. 1994 Sep;1(5):415-22. doi: 10.1007/BF02303815. PMID: 7850543.
- Pisters PW, Harrison LB, Leung DH, Woodruff JM, Casper ES, Brennan MF. Long-term results of a prospective randomized trial of adjuvant brachytherapy in soft tissue sarcoma. J Clin Oncol. 1996 Mar;14(3):859-68. doi: 10.1200/JCO.1996.14.3.859. PMID: 8622034.
- Imanishi J, Slavin J, Pianta M, Jackett L, Ngan SY, Tanaka T, Charoenlap C, DI Bella C, Choong PF. Tail of Superficial Myxofibrosarcoma and Undifferentiated Pleomorphic Sarcoma After Preoperative Radiotherapy. Anticancer Res. 2016 May;36(5):2339-44. PMID: 27127141.
- Kikuta K, Kubota D, Yoshida A, Morioka H, Toyama Y, Chuuman H, Kawai A. An analysis of factors related to the tail-like pattern of myxofibrosarcoma seen on MRI. Skeletal Radiol. 2015 Jan;44(1):55-62. doi: 10.1007/s00256-014-1992-5. Epub 2014 Aug 30. PMID: 25172220.
- Sarcoma Meta-analysis Collaboration (SMAC). Adjuvant chemotherapy for localised resectable soft tissue sarcoma in adults. Cochrane Database Syst Rev. 2000;(4):CD001419. doi: 10.1002/14651858.CD001419. PMID: 11034717.
- Woll PJ, Reichardt P, Le Cesne A, Bonvalot S, Azzarelli A, Hoekstra HJ, Leahy M, Van Coevorden F, Verweij J, Hogendoorn PC, Ouali M, Marreaud S, Bramwell VH, Hohenberger P; EORTC Soft Tissue and Bone Sarcoma Group and the NCIC Clinical Trials Group Sarcoma Disease Site Committee. Adjuvant chemotherapy with doxorubicin, ifosfamide, and lenograstim for resected soft-tissue sarcoma (EORTC 62931): a multicentre randomised controlled trial. Lancet Oncol. 2012 Oct;13(10):1045-54. doi: 10.1016/S1470-2045(12)70346-7. Epub 2012 Sep 4. PMID: 22954508.
- Rizk VT, Naghavi AO, Brohl AS, Joyce DM, Binitie O, Kim Y, Hanna JP, Swank J, Gonzalez RJ, Reed DR, Druta M. Chemotherapy improves distant control in localized high-grade soft tissue sarcoma of the extremity/trunk. Clin Sarcoma Res. 2020 Jul 9;10:11. doi: 10.1186/s13569-020-00132-w. PMID: 32670544; PMCID: PMC7350709.
- Haussmann J, Matuschek C, Bölke E, Tamaskovics B, Corradini S, Wessalowski R, Maas K, Schmidt L, Orth K, Peiper M, Keitel V, Feldt T, Jensen BO, Luedde T, Fischer J, Knoefel WT, Ashmawy H, Pedotoa A, Kammers K, Budach W. Comparison of Different Systemic Therapeutic Regimes in Resectable Soft-Tissue Sarcoma-Results of a Network Meta-Analysis. Cancers (Basel). 2021 Nov 11;13(22):5631. doi: 10.3390/cancers13225631. PMID: 34830786; PMCID: PMC8615898.
- Traweek RS, Martin AN, Rajkot NF, Guadagnolo BA, Bishop AJ, Lazar AJ, Keung EZ, Torres KE, Hunt KK, Feig BW, Roland CL, Scally CP. Re-excision After Unplanned Excision of Soft Tissue Sarcoma is Associated with High Morbidity and Limited Pathologic Identification of Residual Disease. Ann Surg Oncol. 2023 Jan;30(1):480-489. doi: 10.1245/s10434-022-12359-6. Epub 2022 Sep 9. PMID: 36085392.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.