Medicine Group. 2024 September 27;5(9):1214-1228. doi: 10.37871/jbres2009.
Origanum species are known to be powerful antioxidants due to the secondary metabolites they contain.
Sema Yiyit Doğan1, Seçil Kaya2 and Ebru Kondolot Solak1*
2Gazi University, Vocational School of Technical Sciences, Department of Material and Material Processing Technologies, 06374, Ankara, Turkey
- Green synthesis
- Nanoparticles
- Antioxidant activity
- Antimicrobial
Abstract
Origanum species are known to be powerful antioxidants due to the secondary metabolites they contain. It is therefore hypothesized that nanoparticles synthesized by origanum species may have applications in medicine, cosmetics, and food production. This study aimed to investigate the antioxidant and antimicrobial activities of silver nanoparticles obtained at different times (60, 120, and 180 min) and temperatures (30, 60 and 90°C) using O. onites extract. The initial synthesis of Ag Nanoparticles (AgNps) was characterized by UV- spectrophotometry and the surface plasmon resonance band was observed at 430-450 nm under all conditions. The highest absorbance was observed at a wavelength of 425 nm for 180 min at 90°C. The FTIR spectrum showed a peak in the 3.000-3.500 cm-1 range compared to the control, confirming the reduction of silver nitrate. When the particle distribution of AgNPs prepared at three different reaction times was examined by SEM, it was observed that AgNPs with very similar particle sizes were formed. The average particle size was determined to be 171 nm, 169 nm, and 152 nm, respectively, for reaction times of 60, 120, and 180 minutes at 90°C, based on the results of the EDX analysis. The highest DPPH scavenging activity was observed in nanoparticles obtained at the third hour of the 60°C and 90°C reactions. AgNPs obtained at all 3 temperatures showed antimicrobial activity on S. aureus, E. coli, and C. albicans, however, the highest activity was found in AgNPs synthesized at 60°C and 90°C at the 3rd hour. Nanoparticles synthesized from natural materials should not display toxic properties. The AgNPs produced in this study did not exhibit toxicity towards healthy human fibroblasts. The results demonstrated that the AgNPs synthesized within the scope of this study have potential applications in biomedical fields. It was determined that the AgNPs with the highest activity were synthesized at temperatures of 60°C and 90°C at the 3rd hour.
Introduction
Nanoparticles (NPs), which are an important material class in the field of nanotechnology in recent years with sizes ranging from 1 to 1000 nm, have significant applications in the pharmaceutical industries, biomedical, textiles, wastewater treatment, and biosensors [1-4]. Today, researchers' studies on the applications of metal nanoparticles such as silver, copper, selenium, zinc, gold, titanium, and magnesium among nanoparticles reach enormous dimensions. Due to their unique morphology, controllable geometric structure, high specific surface area, surface energy, catalytic performance, and stability, Silver Nanoparticles (AgNPs) attract a lot of attention among researchers and are applied in many fields from electronics to diagnosis and therapy [3-6]. AgNPs have higher antibacterial activity, lower drug resistance, and many improved properties than ordinary silver. The synthesis of AgNPs has a wide variety of sub-methods according to the application area, mainly physical, chemical, and biological (green) methods [7]. The method of obtaining AgNP through green synthesis, which has become a subject of intense research in recent years, has low energy consumption and separation units, is environmentally friendly, economical, and sustainable, and is highly preferred [8-10]. In this approach, AgNPs are synthesized using raw materials (such as microorganisms, plants, and templates) and reagents suitable for green chemistry [5,8].
Plants have a wide variety of phytochemical compounds (polyphenols, flavonoids, terpenes, saponins, terpenoids, alkaloids, tannins, saccharides, vitamins, chelating proteins, etc.) that have an antioxidant effect [11,12]. Since the first article on the green synthesis of AgNPs via the alfalfa (Medicago sativa) plant was published by Gardea-Torresdey, et al. [13] in 2003, the mechanism of silver nanoparticle synthesis via plant extracts remains a mystery [14]. Studies have shown that the formation mechanism of silver nanoparticles depends on the type of plant extract used as a reducing agent, and also that many components present in the plant are responsible for the formation of metal nanoparticles [11,15]. Also, various studies show that silver ions in plant material extracts are formed in plants by reduction via proteins [16], polysaccharides [17], vitamins [18], and secondary metabolites [19]. Sharma, et al. [16] reported that secondary amine in the protein was considered responsible for the reduction16. Ag+ ions adhere to the protein surface and are reduced, forming silver nuclei. The resulting silver nuclei are reduced and grow repeatedly and accumulate in the nuclei, forming AgNPs [14]. In other studies, the presence of some functional groups (such as -C–O–C–, –C=O–, –C–O–, –C=C-), polyol components, and water-soluble heterocyclic components in phytochemicals serve as reducing and stabilizing agents in metal synthesis [20-22].
The O. onites, belonging to the Lamiaceae family, is a group of plants that grow in the Eastern Mediterranean region and are widely used in the treatment of various diseases in traditional medicine [23,24]. O. onites, is a perennial and herbaceous plant species [25]. O. onites attracts attention as an important potential in the prevention of antimicrobial, antiviral, antioxidant, anticancer, antidiabetic, anti-inflammatory, antimycotic, and neurodegenerative disorders, especially due to its strong phenolic content [26]. Essential oil products of O. onites have “Generally Recognized as Safe (GRAS)” (ESO, GRAS-182.20) status [27]. Also; AgNPs synthesis via O. onites is limited in the literature. This makes it an environmentally friendly and valuable natural resource that finds many applications, especially in the food and pharmaceutical industries. In 2021, Genc reported the synthesis of AgNPs with methanol extract of O. onites for the first time; It has been determined that phytochemicals play an important role in the reduction of silver nanoparticles and that the synthesized approximately 52 nm nanoparticles have high antioxidant activity. The absorption peak was observed in the UV-Vis spectrum at 450 nm. By FTIR analysis, it was observed that secondary metabolites were also present in silver nanoparticles, and by XRD analysis, the synthesized nanoparticles were observed to have a crystal structure [28]. In another study in 2023, Geçer reported the cytotoxic, apoptotic, and necrotic processes of AgNPs synthesized by O. onites. The absorption peak of AgNP formation was observed at 433 nm, dimensional analysis of AgNPs was determined at 18.1 nm, characteristic peaks were revealed by FTIR, and the face-centered cubic unit structure was indexed by XRD. Additionally, in the study, cytotoxic activity was performed by MTT test on Capan-1, L929, and Caco-2 cell lines. While AgNPs showed cytotoxic activity in the Capan-1 and Caco-2 cell lines, no activity was observed in the L929 cell line [29].
In the present study, the synthesis of Silver Nanoparticles (AgNPs) through environmentally friendly methods, particularly employing O. onites extract, demonstrates a sustainable approach, underscoring the potential versatility of these nanoparticles in various applications. Furthermore, the research extensively investigated the antioxidant properties of the synthesized AgNPs using the DPPH● radical scavenging method, revealing a significant temperature-dependent increase in scavenging capacity, notably at 60°C and 90°C. The observed results imply potential advantages for biomedical and therapeutic applications. Additionally, the evaluation of cytotoxicity on human fetal lung fibroblast (MRC-5) cells demonstrated the biocompatibility of AgNPs over a 48-hour incubation period, further emphasizing their suitability for safe utilization in medical contexts. In conclusion, the eco-friendly synthesis of AgNPs utilizing O. onites extract, coupled with their verified antioxidant activity and biocompatibility, underscores their pivotal role in advancing sustainable nanomaterials for diverse applications.
This study examines the efficacy of O. onites extract as a model for environmentally friendly and sustainable methods in the synthesis of Silver Nanoparticles (AgNPs). The results obtained from UV-spectrophotometry, FT-IR, and SEM analysis indicated that O. onites has the potential to be a highly effective method for synthesizing nanoparticles in a green manner, although the results did vary depending on the temperature and time. The results demonstrate that the synthesized AgNPs exhibit antimicrobial activity against S. aureus, E. coli, and C. albicans. Furthermore, the DPPH scavenging activity of AgNPs demonstrated that they possess antioxidant properties. In addition to exhibiting antioxidant and antimicrobial activity, the AgNPs were found to have no toxic effect on healthy human cells (MRC-5) in a cytotoxicity study. Overall, the synthesis of AgNPs at temperatures of 60°C and 90°C is more effective. Consequently, it can be assumed that O. onites-mediated AgNPs obtained through green synthesis may possess the effective, reliable and biocompatible potential for biomedical and therapeutic applications.
Through our literature review, we identified Origanum onites as an under-researched plant, despite its known potent phenolic content and significant antioxidant, antimicrobial, and therapeutic properties, particularly within its native Eastern Mediterranean region. Notably, the application of O. onites in the synthesis of Silver Nanoparticles (AgNPs) has been scarcely explored in existing literature.
In this study, we introduce a novel approach that leverages the bioactive compounds of O. onites to synthesize biocompatible and environmentally friendly AgNPs, potentially offering distinct advantages for various biomedical and therapeutic applications. Our findings demonstrate that AgNPs synthesized with O. onites extract exhibit enhanced antioxidant and antimicrobial activities, particularly at synthesis temperatures of 60°C and 90°C with a reaction time of 3 hours. This enhancement is attributed to the unique phytochemical profile of O. onites, which we hypothesize plays a crucial role in the superior properties of the synthesized nanoparticles. Furthermore, the biocompatibility of these AgNPs, confirmed by their non-toxicity in MRC-5 human fetal lung fibroblast cells, underscores their potential for safe application in medical contexts, thereby distinguishing our method from conventional approaches.
In, the use of O. onites in the green synthesis of AgNPs presents a sustainable, effective, and innovative alternative to existing methods, with broad potential applications in nanotechnology and biomedicine.
Materials and Methods
Materials
The plant material was collected from Denizli, Turkiye. Silver Nitrate (AgNO3) was purchased from Chem Pure (U.S.A). Ethanol was obtained from Isolab (Turkiye). Gallic acid, Folin-Ciocalteu’s phenol reagent, and sodium carbonate were obtained from Merck KGaA Chemical Co. (Germany). Dimethyl sulfoxide (DMSO) was purchased from Carlo Erba (France). DPPH (2,2-Diphenyl-1-picrylhydrazyl) and ethanol were supplied from Sigma–Aldrich (Germany). MTT (3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) was obtained from Biotium (USA). Antimicrobial Susceptibility Testing discs; Tetracycline (30 µg), fluconazole (25 µg), and blank were purchased from Bioanalyse (Turkey).
Preparation of plant extract
O. onites was collected in March 2022, from Denizli Turkiye. The leaves were washed with distilled water and completely dried plants were ground using mechanical mortar. 20 grams of sample were extracted in water/ethanol solution (30/70) at 100°C in a Soxhlet Apparatus for 6h. The extracted solution was evaporated at 40°C. After the solvent was removed, the extract was stored at +4°C for further use.
Green synthesis of silver nanoparticles
Briefly, 1 mm silver nitrate was dissolved in 50 ml of distilled water. 10 mg extract was added to the silver nitrate solution under continuous stirring for 3h at 30-60-90°C. At the optimum reaction temperature, the same experimental process was followed for the formation of AgNPs at different reaction times (60-120-180 min). After incubation, the reaction mixture color transformed from light yellow to a dim brown color. After the end of the reaction time, the mixture was allowed to cool and then centrifuged (Hitachi/CR22GII, Japan) at 11000 rpm for 30 minutes. The resulting product was then washed several times with deionized water and centrifuged again under the same conditions (Figure 1). The resulting black precipitate was kept in an oven at 40°C until completely dry [29,30].
Mechanism of nanoparticle formation
The green synthesis approach leverages the phytochemical compounds present in O. onites, such as polyphenols, flavonoids, terpenes, and carvacrol, which act as reducing and stabilizing agents. These compounds are crucial in the reduction of silver ions (Ag⁺) to elemental silver (Ag⁰), leading to the nucleation and growth of nanoparticles. Specifically, the phenolic groups present in carvacrol and other compounds facilitate the donation of electrons to silver ions, reducing them and allowing the formation of silver nuclei. These nuclei further aggregate and stabilize due to the presence of other phytochemicals, resulting in the formation of AgNPs with distinct size and shape characteristics.
Characterization
The surface plasmon resonance of NPs was performed using a UV–Vis Spectrophotometer (Thermo Scientific Genesys 150, USA) with a 325-800 nm spectral range. The maximum absorbance was observed at 425 nm. SEM (FEI Company QUANTA 400F Field Emission, USA) was used for morphological analysis. The elemental composition of the obtained samples and the presence of silver metal in the samples were analyzed by Energy Dispersive X-ray analysis (EDX) combined with FE-SEM during the SEM study. The interpretation of chemical bonds and functional groups was conducted using FT-IR spectroscopy (Bruker Vertex 80/80v, Germany). Particle size distribution was performed by dynamic light scattering (DLS) using a particle size analyzer (Zetasizer Nano ZS90, Malvern Panalytical Ltd., UK).
Antioxidant activity
We investigated the antioxidant activity of the samples using the DPPH● (2,2-diphenyl-1-picrylhydrazyl) radical scavenging method, as developed by Blois in 1958. This assay relies on measuring the scavenging capacity of antioxidants against the stable free radical DPPH●. The odd electron of the nitrogen atom in DPPH● is reduced as it receives a hydrogen atom from antioxidants, forming the corresponding hydrazine. The deep violet color of DPPH● transforms into yellow when mixed with the antioxidants, and the absorbance is measurable at a wavelength of 517 nm [31]. 0.004% DPPH● solution was prepared in ethanol. The prepared DPPH● solution was added to different concentrations of silver nanoparticles in a 1:1 ratio and incubated for 30 minutes in the dark. At the end of incubation, absorbances were measured at 517 nm wavelength (Thermo Scientific Genesys 150, the U.S.A), and percentage DPPH● removal was calculated according to the following formula (Equation 1) [32]:
(1)
Antimicrobial activity
In this study, the antibacterial activity of silver nanoparticles obtained by green synthesis on gram-positive (Gr +) S. aureus ATCC 25923, gram-negative (Gr-) E. coli ATCC 25922 bacteria and antifungal activity on C. albicans ATCC 10231 fungus were investigated. S. aureus and E. coli were grown in Mueller-Hinton Broth (MHB), and C. albicans was grown in Sabouraud (2%) Dextrose Broth (SDB). The antimicrobial activity of AgNPs was investigated by the disc diffusion method [33]. Two times activated microorganisms were adjusted to McFarland 0.5 density and smear cultivation was performed by adding 1% to appropriate agar media. Discs were impregnated with 50 μl silver nanoparticles and placed on agar plates and left to incubate for 24 hours at 37°C. At the end of incubation, the zones around the discs were measured and evaluated. Tetracycline (30 μg) and fluconazole (25 μg) antibiotic discs were used as control.
Cytotoxic activity
Human fetal lung fibroblast (MRC-5) cells were cultured in DMEM supplemented with fetal bovine serum (10%), antibiotic (penicillin-streptomycin solution 1%), and glutamine. The cell cultures were incubated at 37°C in a 5% CO2 atmosphere. MRC-5 cells were pre-incubated in a 96-well plate at a density of 2 x 105 cells per well for 24 hours. After incubation, the medium was removed, and DMEM-containing nanoparticles were introduced into the wells for testing the activity at 24 and 48 hrs. Cytotoxic activity was determined by the MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) method. The medium in the wells was removed at the end of the 24th and 48th incubations. MTT solution at 1 mg/ml prepared in DMEM was then added to the wells and kept at 37°C for 2 hours. The production of formazan crystals was analyzed by dissolving the cells with 100 μg DMSO. Subsequently, absorbance was measured at 570 nm, and cell viability was calculated [29].
Results and Discussion
Characterization of AgNPs
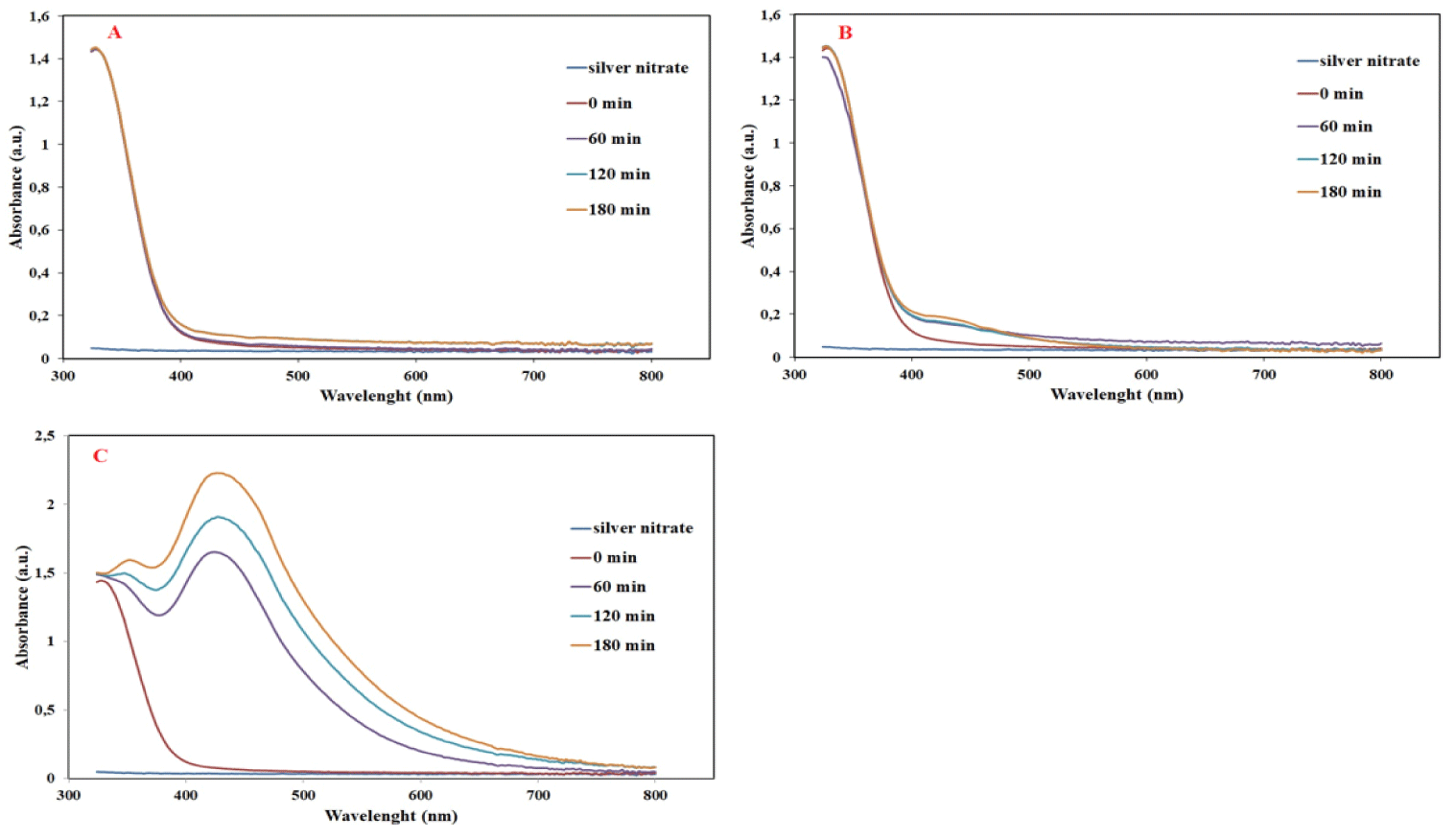
UV–Vis spectral analysis: Figure 2 shows the color change of a chemical reaction involving the reduction of Ag+ ions in the reaction mixture, which manifests itself with a visible color change from light yellow to dark brown.
We used UV-Vis analysis, a widely used technique to follow Ag+ ion reduction reactions and confirm the formation of Silver nanoparticles (AgNPs). The UV–Vis spectrum of AgNPs exhibits a significant band spanning the visible region from 300 nm to 800 nm, with a particularly sharp surface plasmon peak at 425 nm. This distinctive peak is indicative of the presence of silver nanoparticles, which strengthens the validity of the reduction process. The experimental design extends to exploring the effect of temperature and reaction time on the Ag+ reduction reaction. UV-Vis analysis was performed at three different temperature values (30°C, 60°C, 90°C) at various time intervals (60, 120, and 180 minutes) to obtain a comprehensive data set. The graphical representation of these results in Figure 3 reveals that the maximum absorbance at 425 nm was obtained if the reaction was carried out for 180 min at 90°C (Figure 3C).
This meticulous investigation of the temperature and time dependence of the reaction provides critical information for optimizing the synthesis of silver nanoparticles. Specific conditions that provide the highest absorbance at 425 nm are crucial to increase the reproducibility of the Ag+ reduction process and thus contribute to the advancement of nanoparticle synthesis techniques.
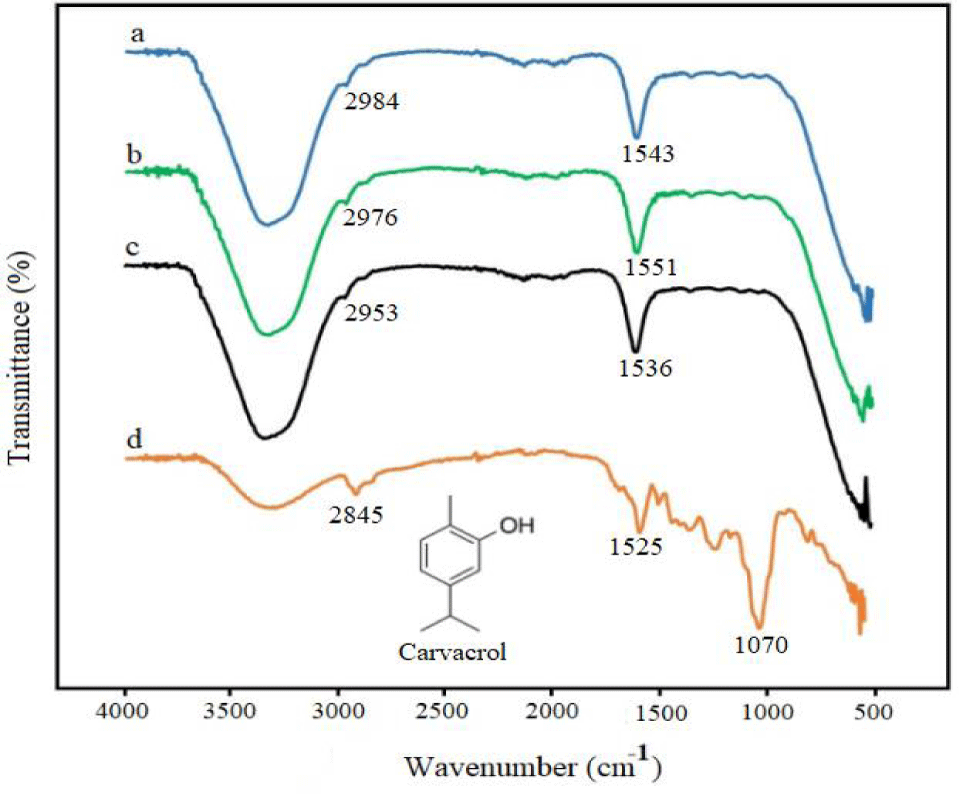
FTIR: The FTIR spectra of silver nanoparticles synthesized from O. onites extract at different temperatures are presented in figure 4. A comparison of the FTIR spectra of the extract and silver nanoparticles indicates the formation of silver nanoparticles. The main component of the oil has been determined to be 70.6% carvacrol in the literature [34]. In the FTIR spectrum of silver nanoparticles, a broad band in the range of 3000-3500 cm-1 is attributed to OH stretching, observed consistently in all spectra [35]. For the pure O. onites extract, a C-H asymmetric stretching band is visible at 2845 cm-1. Peaks corresponding to C–H asymmetric stretching are observed at 2984, 2976, and 2953 cm-1 for the a, b, and c spectra, respectively.
Furthermore, the stretching vibration of the C = C functional group appears at 1525 cm-1 in the spectrum of O. onites extract. This peak is observed at 1543 cm-1, 1551 cm-1, and 1536 cm-1 for the a, b, c, and d spectra, respectively. The peak observed at 1070 cm-1 in the spectrum of O. onites extract (d) can be attributed to the C–O stretching of alcohol. The differences between the extract and nanoparticle spectra are attributed to the oxidation of some molecules in the extract due to the reduction of silver ions.
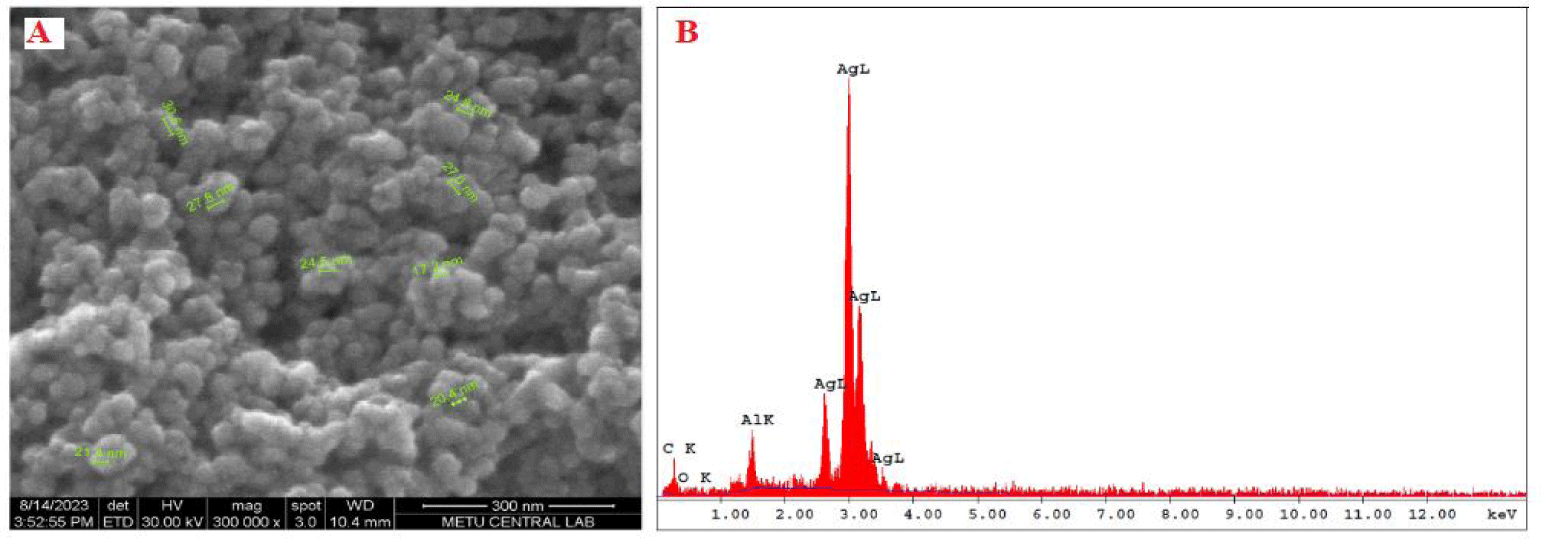
SEM and EDX analysis: The morphological form of AgNPS was elucidated using SEM. The distribution of AgNPS over a large surface area was revealed through SEM images. SEM images were obtained for three different temperatures (30ºC, 60ºC, and 90ºC) values (Figure 5). The SEM images of O. onites-mediated AgNPS demonstrated the nanoparticles synthesized at all three temperature values were small and spherical and not much aggregated.
The EDX confirmed the formation of silver nanoparticles (AgNPs) (Figure 6). A prominent, sharp signal at 3 keV distinctly indicates that silver (Ag) constitutes the primary element. Additional signals within the 0–1.0 keV range correspond to the characteristic absorption of carbon and oxygen. SEM images at 300000x magnification (Figure 6(A)) depicting O. onites-mediated silver nanoparticles revealed the presence of very small and uniformly spherical nanoparticles. Figure 6(B) exhibited a strong signal in the silver region, further confirming the successful formation of AgNPs.
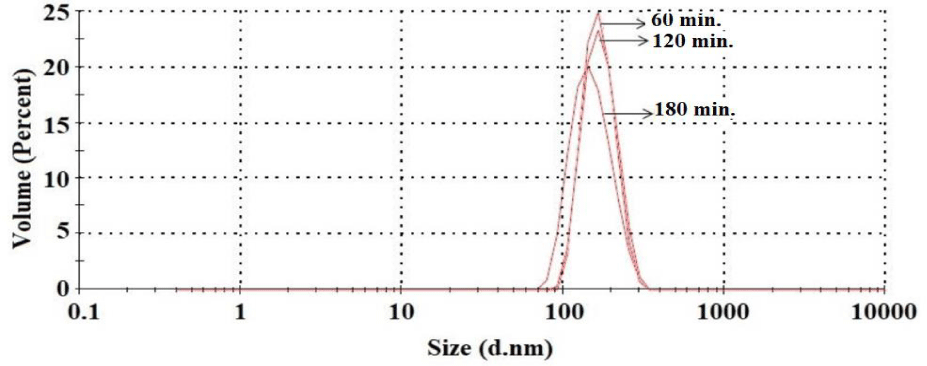
Particle size distribution curves for AgNPs prepared at different reaction times at 90ºC are presented in figure 7. When the particle distribution of AgNPs prepared at three different reaction times is examined, it is observed that AgNPs with very similar particle sizes are formed. At 90ºC, the average particle sizes for reaction times of 60, 120, and 180 minutes were determined as 171 nm, 169 nm, and 152 nm, respectively.
Antioxidant activity
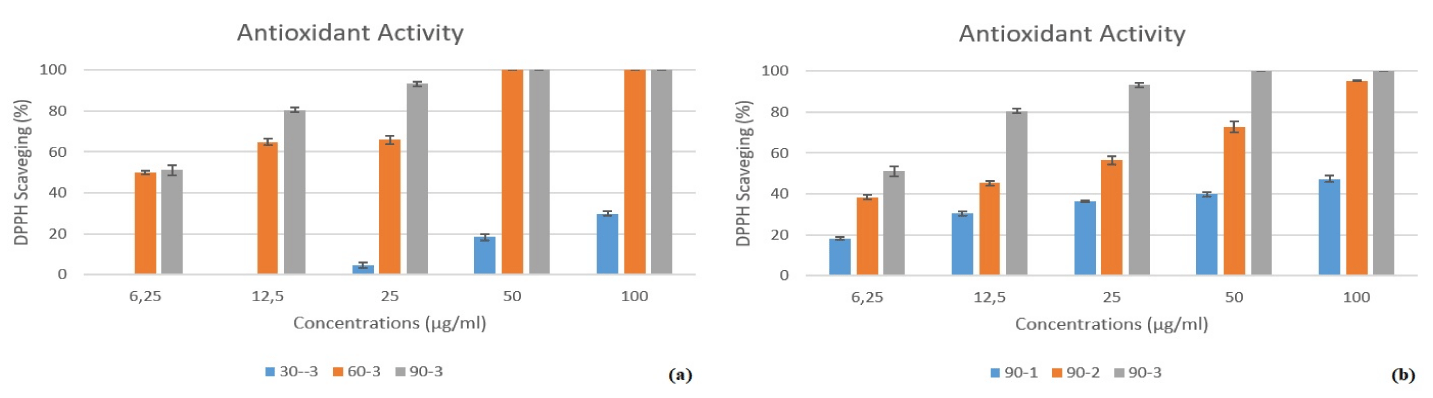
The antioxidant activity of O. onites-mediated AgNPs was evaluated using DPPH (1,1-diphenyl-2-picrylhydrazyl) free radical scavenging assay (Figure 8). The synthesized AgNPs had free radical scavenging activity. The cleaning ability increased in a temperature-dependent manner. In our study, antioxidant experiments were conducted on extraction solutions from three-hour (60, 120, and 180 minutes) processes at all three temperatures (30ºC, 60ºC, and 90ºC) values. It is evident that samples at 60ºC and 90ºC exhibit DPPH removal efficiency across all concentration levels for 180 min. whereas samples at 6.25 and 12.5 mg/ml concentrations for 30 ºC do not demonstrate DPPH removal efficiency (Figure 8A). Figure 8A illustrates that samples at 90ºC exhibit consistently high DPPH removal efficiency across all concentration values. For this reason, antioxidant measurements were performed for AgNP nanoparticle synthesis at 90ºC for 60, 120, and 180 minutes (Figure 8B). It was observed that the nanoparticles synthesized for 3 hours displayed the highest DPPH removal efficiency.
Origanum species are known to be strong antioxidants due to the terpenes, phenols, phenolic acids, flavonoids, carvacrol, thymol etc compounds they contain [36]. In recent studies, it has been reported that silver nanoparticles obtained from Origanum species are also strong antioxidants and can be used in medical, cosmetic, and food industries due to their natural and environmentally friendly nature. Genç, et al [37] reported that AgNPs (60ºC, 2 h) obtained from O. onites collected from Tokat/Turkey had high DPPH removal (IC50,18.42 ± 0.34 µg/mL) and therefore could be used as an antioxidant agent in the food industry. In another study, DPPH removal of AgNPs obtained by biosynthesis with O. majorana extract at 70ºC for 2 hours was investigated. As a result of the study, it was reported that the obtained AgNPs had a high DPPH removal activity (IC50, 12.25 µg/mL) and could be used in food and pharmaceutical applications
Antimicrobial activity
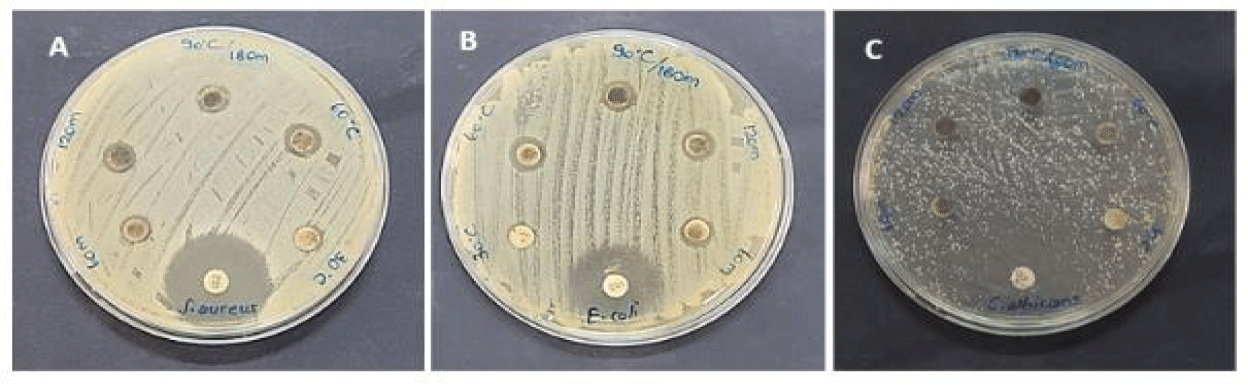
AgNPs obtained by green synthesis were found to have antimicrobial activity on all microorganisms studied (Figure 9, table 1). Antibacterial activity was found to be higher than antifungal activity. The antimicrobial activity of AgNPs on E. coli (Figure 9B) and S. aureus (Figure 9A) was found to be almost similar. When the 180 minutes antimicrobial activities of 30, 60, and 90ºC temperatures were compared, a low activity was observed at 30ºC, while high antimicrobial activity was found at 60ºC (E. coli and S. aureus: 10 mm, C. albicans: 6.5 mm) and 90ºC (E. coli: 10 mm, S. aureus: 9.5 mm and C. albicans: 7 mm). The antimicrobial activity of nanoparticles synthesized at three different times of 90ºC temperature (60, 120, and 180 hours) was found to cause inhibition on microorganisms at all three times. The highest activity was observed at 2 and 3 hours (Table 1). Diseases and deaths caused by pathogenic microorganisms are a serious problem worldwide [38]. E. coli is a Gram-negative (Gr-) coliform bacteria. It can cause various potentially fatal infectious diseases, including colitis, acute renal failure [39], and diarrhea [38]. It has been reported that more than 2 million people die each year from E. coli-related diseases [39]. S. aureus is an important opportunistic pathogen normally found on human skin and nasal mucosa. It is known to be responsible for nosocomial infections [40]. It can also cause pneumonia, endocarditis, meningitis, and infection in wounds [41]. C. albicans is a fungus that survives on the upper respiratory tract, gastrointestinal and genital mucosal surfaces, and skin in humans [42]. C. albicans is responsible for intestinal intolerance, vaginal fungi, and wound and hospital infections. It has also been reported that it can cross the body barrier enter the bloodstream and affect the central nervous system, liver, spleen, heart, and kidneys [43,44]. Antimicrobial resistance has become a global health problem [45,46]. WHO (2019) report states that at least 700 thousand people die from drug-resistant diseases every year [46]. Plants stand out as alternative antimicrobial sources. Studies with O. onites indicated that the extract [24] and essential oil [47,48] of this species have high antimicrobial activity on resistant pathogens. It is known that silver nanoparticles show very strong antimicrobial activity on resistant microorganisms [49]. Within the scope of the study, silver nanoparticles were obtained at different temperatures and times with O. onites, which have known antimicrobial properties, and their antimicrobial activities were compared. The best activity was detected at 180 minutes of 60 and 90ºC temperatures. There is no literature on the antimicrobial activity of AgNp from O. onites. However, studies with O. vulgare are available. Sankar R, et al. [50] reported that they obtained a 10 mm zone of inhibition against E. coli with silver nanoparticles synthesized with O. vulgare, similar to our study. Benedec D, et al. [51] reported that the gold nanoparticles synthesized with O. vulgare resulted in a high zone on S. aureus and C. albicans, but did not cause inhibition in E. coli Hambardzumyan S, et al. [52] reported that silver nanoparticles from O. vulgare were effective on S. aureus and E. coli.
| Table 1: Antimicrobial activity (mm) for AgNPs synthesized at 180 minutes at 30ºC, 60ºC, 90ºC and at 90ºC for 60, 120, and 180 minutes. | |||
| E. coli | S. aureus | C. albicans | |
| 30ºC 180 min | 6.7 ± 0.58 | 8.7 ± 0.58 | 5.0 ± 0 |
| 60ºC 180 min | 9.7 ± 0.58 | 10.7 ± 0.58 | 6.3 ± 0.58 |
| 90ºC 180 min | 9.7 ± 0.58 | 9.7 ± 0.58 | 6.7 ± 0.58 |
| 90ºC 120 min | 7.3 ± 0.58 | 9.7 ± 0.58 | 6.3 ± 0.58 |
| 90ºC 60 min | 7.7 ± 0.58 | 8.3 ± 0.58 | 5.7 ± 0.58 |
| Tetracycline* | 21.7 ± 0.58 | 24.00 ± 1.0 | |
| Fluconazole** | - | - | 18.3 ± 1.15 |
| *Tetracycline: antibacterial disc, **fluconazole: antifungal disc | |||
Cytotoxic activity
It was observed that AgNps obtained with O. onites were not toxic to healthy cells and even increased healthy cell growth. At 60, 120, and 180 minutes for 90ºC, the viability was 95% and 98% at the 60th minute, but the viability increased at the 120th and 180th minutes. When the 180-minute cytotoxicity of 30ºC, 60ºC, and 90ºC temperatures was compared, it was observed that the number of living cells increased depending on time. The highest viability was detected at temperatures of 60ºC and 9 ºC (Figure 10). Silver has been used in biomedical fields for many years because it is a powerful antioxidant and antimicrobial agent. However, the safety of its use is still controversial. For this reason, it is important that the NPs to be obtained show high biological activity and do not harm healthy cells [29, 53,54]. It is known that the factors used in the synthesis of NPs (temperature, time, method, etc.) affect the size and shape of the nanoparticle to be obtained and this can directly affect the properties of the NPs [51]. In our study, it was observed that different temperatures and times changed the activity of AgNPs and none of the AgNPs obtained did not harm healthy cells. It has been shown that silver nanoparticles obtained from O. onites leave show cytotoxic activity on cancer cells, human pancreatic adenocarcinoma cell lines (Capan-1), and human colon adenocarcinoma cell line (Caco-2), while they do not harm healthy cells, mouse normal fibroblast cell lines (L929). In fact, at some doses, it has been shown to increase cell growth, similar to our study [29]. In another study, it was stated that silver nanoparticles obtained by plant-derived green synthesis caused cytotoxicity in MCF-7 (breast cancer cell lines) and Hela (cervical cancer cell line) cancer cells, but did not harm healthy cells NIH-3 T3 (mouse embryonic fibroblast) [55]. In another study conducted with MCF-7 and healthy L-929 cell lines, it was stated that green synthesis AgNps selectively inhibited cancer cells but did not harm healthy cells. It is suggested that cancer cells have an abnormal metabolism and high proliferation rate, making them vulnerable, and AgNPs show superior cytotoxicity against cancer cells due to the high uptake of nanoparticles by these cells rather than healthy cells [56].
Cell viability can be attributed to various factors related to synthesis conditions and resulting nanoparticle properties. Nanoparticles synthesized at different temperatures and times may differ in size, shape, surface charge, and the presence of surface-bound phytochemicals, all of which can significantly affect their interactions with cells.
Size and surface area: Figure 7 shows the size distributions of nanoparticles synthesized at 90ºC for 60, 120 and 180 minutes. It is seen that as the preparation time of nanoparticles increases, they have a smaller surface area. This shows that smaller nanoparticles that are synthesized in a longer time generally have a larger surface area compared to their volume. Therefore, nanoparticles with larger surface area cause greater interaction with cell membranes, which could potentially explain higher cell viability. Nanoparticles synthesized at 90°C for 60 minutes did not reach the optimal size that supports this enhanced interaction, and it is thought that this may cause a decrease in cell viability.
Surface chemistry and phytochemical covering: Synthesis time can also affect the density and composition of phytochemicals coating the nanoparticle surface. These phytochemicals from O. onites extract can interact with receptors on the cell surface or enter cells, promoting cellular proliferation. Nanoparticles synthesized for 60 min at 90°C may have a less dense or different phytochemical coating, leading to a reduced proliferative effect compared to those synthesized for a longer time.
Production of reactive oxygen species: Production of Reactive Oxygen Species (ROS) by nanoparticles may vary depending on synthesis conditions. Nanoparticles that produce ROS at levels that promote cell signaling and proliferation rather than causing oxidative stress may explain the unusually high cell viability. It is possible that nanoparticles synthesized for 60 min at 90ºC generate lower levels of ROS or different types of ROS, which may not trigger the same proliferative response as those synthesized for longer periods of time.
Performance evaluation of AgNPs obtained with origanum onites extract
The performance evaluation of the Green Synthesis of AgNPs presented in this study is commendable, reflecting a meticulous and comprehensive approach to both preparation and
Characterization. The utilization of O. onites plant extract, collected from Denizli, Turkey, adds a unique and environmentally friendly dimension to the synthesis process.
The study systematically investigates the impact of various parameters, such as temperature and reaction time, on the synthesis of AgNPs. The exploration of different temperature values (30ºC, 60ºC, and 90ºC) and reaction times (60, 120, and 180 minutes) provides valuable insights into the controllable factors influencing nanoparticle formation. This attention to detail enhances the reproducibility of the synthesis method and contributes to a more nuanced understanding of the process.
Characterization techniques including UV–Vis Spectrophotometry, Scanning Electron Microscopy, Energy Dispersive X-ray analysis, Particle size distribution, and Fourier-transform infrared spectroscopy contribute to a thorough understanding of the properties of the synthesized AgNPs. The examination of surface plasmon resonance, morphological features, elemental composition, and chemical bonds provides a robust foundation for evaluating the nanoparticles' potential applications.
The study goes beyond mere characterization and delves into the functional aspects of the AgNPs. The evaluation of antioxidant activity using the DPPH radical scavenging method showcases a temperature-dependent enhancement in scavenging capacity. This not only highlights the multifunctional nature of the AgNPs but also emphasizes the importance of synthesis conditions in tailoring their desired characteristics.
The exploration of antimicrobial properties against both bacteria (S. aureus, E. coli) and fungus (C. albicans) reveals significant inhibitory effects, with a noteworthy temperature-dependent variation. The correlation between synthesis temperature and duration with antimicrobial efficacy further underscores the careful consideration of these parameters in optimizing the performance of the AgNPs. The cytotoxicity evaluation on human fetal lung fibroblast (MRC- 5) cells is a crucial aspect of the study, demonstrating the biocompatibility of the AgNPs over a 48-hour incubation period. This finding is particularly promising for potential biomedical applications.
The significance of this manuscript is underscored by the fact that these microorganisms are known to cause serious health issues due to their drug-resistant properties, making the study of AgNPs particularly relevant. We have thoroughly examined the antibacterial properties of AgNPs synthesized with O. onites extract and provided a detailed discussion of the results. The bactericidal efficacy of these nanoparticles has been highlighted. Through this focused analysis, we aim to contribute to a more robust and targeted evaluation of the potential of O. onites-mediated AgNPs in combating bacterial infections, especially those caused by drug-resistant strains.
The study provides a thorough and well-executed investigation into the Green Synthesis of AgNPs using O. onites plant extract. The detailed exploration of synthesis conditions, comprehensive characterization, and functional assessments contribute to the understanding of the AgNPs' properties and potential applications. The temperature-dependent variations observed underscore the importance of synthesis conditions in tailoring the nanoparticles for specific purposes.
Comparison with existing green synthesis methods: Green synthesis of silver nanoparticles (AgNPs) has been explored using various plant extracts due to their eco-friendly and sustainable nature. Among the numerous methods reported in the literature, plants like Medicago sativa, Azadirachta indica, and Origanum vulgare have been widely used as they contain a rich variety of bioactive compounds that can act as reducing and stabilizing agents. In this study, we employed Origanum onites, a plant native to the Eastern Mediterranean region, for the green synthesis of AgNPs, which is relatively underexplored compared to other species.
Enhanced antioxidant and antimicrobial activities: Our results demonstrated that AgNPs synthesized using O. onites exhibit remarkable antioxidant and antimicrobial activities, particularly at synthesis temperatures of 60°C and 90°C for 180 minutes. This contrasts with other studies where different plants were used, showing varying degrees of activity. For example, AgNPs synthesized using Origanum majorana showed significant DPPH scavenging activity, but the results obtained with O. onites were more pronounced, indicating its superior potential as a reducing agent in green synthesis. Moreover, the antimicrobial activity of O. onites-mediated AgNPs was found to be more effective against Staphylococcus aureus, Escherichia coli, and Candida albicans compared to nanoparticles synthesized with other plant extracts. Studies with O. vulgare have shown good antimicrobial properties, but the zone of inhibition observed with O. onites was higher, suggesting that the nanoparticles synthesized from O. onites may have stronger interactions with microbial cell membranes, likely due to the specific bioactive compounds present in this plant.
Biocompatibility: A critical advantage of using O. onites in the green synthesis of AgNPs is the biocompatibility of the resulting nanoparticles. The cytotoxicity studies conducted on human fetal lung fibroblast (MRC-5) cells revealed that the AgNPs synthesized with O. onites were non-toxic and even promoted cell viability at certain concentrations. This is consistent with other studies where plant-mediated AgNPs have shown selective cytotoxicity towards cancer cells while being safe for normal cells. The non-toxic nature of O. onites-mediated AgNPs makes them particularly suitable for biomedical applications, offering an edge over other synthesis methods where cytotoxicity remains a concern.
Environmental impact and sustainability: In terms of environmental impact, the use of O. onites offers a sustainable and renewable resource for nanoparticle synthesis. The process described in this study adheres to green chemistry principles, minimizing the use of harmful chemicals and energy-intensive processes. Compared to conventional chemical synthesis methods, the approach using O. onites is more environmentally friendly and aligns with the global trend towards sustainable nanotechnology.
Conclusion
In conclusion, this study delineates a comprehensive investigation into the preparation, extraction, and applications of O. onites plant extract-derived silver nanoparticles (AgNPs) obtained through a green synthesis approach. The meticulous preparation of the O. onites extracts involved cleaning, drying, and grinding of leaves, followed by extraction in a water-ethanol solution using a Soxhlet Apparatus. The resulting extract was utilized for the green synthesis of AgNPs, with the impact of varying temperatures (30ºC, 60ºC, and 90ºC) on nanoparticle formation investigated.
The synthesized AgNPs were characterized using UV–Vis Spectrophotometry, Scanning Electron Microscopy, and FT-IR spectroscopy, providing valuable insights into their surface plasmon resonance, morphological features, and chemical composition. The antioxidant activity of the AgNPs was evaluated through the DPPH radical scavenging method, demonstrating a temperature-dependent enhancement in scavenging capacity, particularly at 60ºC and 90ºC.
Furthermore, the antimicrobial properties of the AgNPs were explored against Gram-positive (S. aureus), Gram-negative (E. coli) bacteria, and the fungus C. albicans. The AgNPs exhibited significant antibacterial activity, surpassing their antifungal effects, with the highest efficacy observed at 60ºC and 90ºC synthesis temperatures. Notably, the antimicrobial activity was dependent on both temperature and synthesis duration, with longer durations at 90ºC demonstrating the highest inhibitory effects.
The study investigated the cytotoxicity of AgNPs on human fetal lung fibroblast (MRC-5) cells, revealing their biocompatibility over a 48-hour incubation period. Additionally, the study found that AgNPs exhibited non-toxicity on a healthy cell line and human fetal lung fibroblast (MRC-5) cells, demonstrating their biocompatibility over the same 48-hour incubation period. Results highlight the potential of O. onites-mediated AgNPs as multifunctional nanoparticles with antioxidant and antimicrobial activities. The temperature-dependent variations observed in their properties open avenues for tailored applications, emphasizing the importance of synthesis conditions in optimizing the desired characteristics.
Acknowledgment
We gratefully acknowledge Gazi University Photonics Application and Research Center and Middle East Technical University Central Laboratory.
Conflict of interest statement
The authors declare no competing interests.
References
- Choudhry A, Sharma A, Siddiqui SI, Ahamad I, Sajid M, Khan TA, Chaudhry SA. Origanum vulgare manganese ferrite nanocomposite: An advanced multifunctional hybrid material for dye remediation. Environ Res. 2023 Mar 1;220:115193. doi: 10.1016/j.envres.2022.115193. Epub 2022 Dec 29. PMID: 36587717.
- Al-Sheddi ES, Alsohaibani N, Rshoud N, Al-Oqail MM, Al-Massarani SM, Farshori NN, Malik T, Al-Khedhairy AA, Siddiqui MA. Anticancer efficacy of green synthesized silver nanoparticles from Artemisia monosperma against human breast cancer cells. S Afr J Bot. 2023;160:123-131. doi: 10.1016/j.sajb.2023.07.001.
- Yontar AK, Çevik S. Effects of plant extracts and green-synthesized silver nanoparticles on the Polyvinyl Alcohol (PVA) nanocomposite films. Arab J Sci Eng. 2023;48:12043-12060. doi: 10.1007/s13369-023-07643-w.
- Menichetti A, Mavridi-Printezi A, Mordini D, Montalti M. Effect of Size, Shape and Surface Functionalization on the Antibacterial Activity of Silver Nanoparticles. J Funct Biomater. 2023 Apr 26;14(5):244. doi: 10.3390/jfb14050244. PMID: 37233354; PMCID: PMC10219039.
- Nie P, Zhao Y, Xu H. Synthesis, applications, toxicity and toxicity mechanisms of silver nanoparticles: A review. Ecotoxicol Environ Saf. 2023 Mar 15;253:114636. doi: 10.1016/j.ecoenv.2023.114636. Epub 2023 Feb 15. PMID: 36806822.
- Singh S, Maurya IC, Tiwari A, Srivastava P, Bahadur L. Green synthesis of TiO2 nanoparticles using Citrus limon juice extract as a bio-capping agent for enhanced performance of dye-sensitized solar cells. Surf Interfaces. 2022;28:101652. doi: 10.1016/j.surfin.2021.101652.
- Li X, Li B, Liu R, Dong Y, Zhao Y, Wu Y. Development of pH-responsive nanocomposites with remarkably synergistic antibiofilm activities based on ultrasmall silver nanoparticles in combination with aminoglycoside antibiotics. Colloids Surf B Biointerfaces. 2021 Dec;208:112112. doi: 10.1016/j.colsurfb.2021.112112. Epub 2021 Sep 14. PMID: 34600361.
- Ghosh A, De SK, Mondal S, Halder A, Barai M, Guchhait KC, Raul P, Karmakar S, Ghosh C, Patra A, Panda AK, Senapati D, Sur UK. Green synthesis of silver nanoparticles and its applications as sensor, catalyst, and antibacterial agent. Materials Today: Proceedings. 2023. doi: 10.1016/j.matpr.2023.03.159.
- Kaur N, Singh A, Ahmad W. Microwave assisted green synthesis of silver nanoparticles and its application: A review. J Inorg Organomet Polym Mater. 2023;33(3):663-672. doi: 10.1007/s10904-022-02470-2.
- Mahmood RI, Kadhim AA, Ibraheem S, Albukhaty S, Mohammed-Salih HS, Abbas RH, Jabir MS, Mohammed MKA, Nayef UM, AlMalki FA, Sulaiman GM, Al-Karagoly H. Biosynthesis of copper oxide nanoparticles mediated Annona muricata as cytotoxic and apoptosis inducer factor in breast cancer cell lines. Sci Rep. 2022;12:16165. doi: 10.1038/s41598-022-20360-y.
- Susanna D, Balakrishnan RM, Ettiyappan JP. Ultrasonication-assisted green synthesis and characterization of gold nanoparticles from Nothapodytes foetida: An assessment of their antioxidant, antibacterial, anticancer and wound healing potential. J Drug Deliv Sci Technol. 2023;87:104740. doi: 10.1016/j.jddst.2023.104740.
- Gutiérrez-Del-Río I, López-Ibáñez S, Magadán-Corpas P, Fernández-Calleja L, Pérez-Valero Á, Tuñón-Granda M, Miguélez EM, Villar CJ, Lombó F. Terpenoids and Polyphenols as Natural Antioxidant Agents in Food Preservation. Antioxidants (Basel). 2021 Aug 8;10(8):1264. doi: 10.3390/antiox10081264. PMID: 34439512; PMCID: PMC8389302.
- Gardea-Torresdey JL, Gomez E, Peralta-Videa JR, Parsons JG, Troiani H. and Yacaman MJ. Alfalfa sprouts: a natural source for the synthesis of silver nanoparticles. Langmuir. 2003;19(4):1357-1361. doi: 10.1021/la020835i.
- Rajeshkumar S, Bharath LV. Mechanism of plant-mediated synthesis of silver nanoparticles - A review on biomolecules involved, characterisation and antibacterial activity. Chem Biol Interact. 2017 Aug 1;273:219-227. doi: 10.1016/j.cbi.2017.06.019. Epub 2017 Jun 21. PMID: 28647323.
- Vidyasagar Patel RR, Singh SK, Singh M. Green synthesis of silver nanoparticles: methods, biological applications, delivery and toxicity. Mater Adv. 2023;4(8):1831-1849. doi: 10.1039/d2ma01105k.
- Sharma NK, Vishwakarma J, Rai S, Alomar TS, AlMasoud N, Bhattarai A. Green Route Synthesis and Characterization Techniques of Silver Nanoparticles and Their Biological Adeptness. ACS Omega. 2022 Jul 25;7(31):27004-27020. doi: 10.1021/acsomega.2c01400. PMID: 35967040; PMCID: PMC9366950.
- Chakraborty B, Bhat MP, Basavarajappa DS, Rudrappa M, Nayaka S, Kumar RS, Almansour AI, Perumal K. Biosynthesis and characterization of polysaccharide-capped silver nanoparticles from Acalypha indica L. and evaluation of their biological activities. Environ Res. 2023 May 15;225:115614. doi: 10.1016/j.envres.2023.115614. Epub 2023 Mar 6. PMID: 36889569.
- Hassan RM. Kinetics of reduction of Se (IV) by vitamin C with green synthesis of cluster-grapes nanoparticles: A Mechanistic approach on electron-transfer of nanoparticle growth rates. J Mol Struct. 2022;1250:131575. doi: 10.1016/j.molstruc.2021.131575.
- Deshmukh SP, Patil SM, Mullani SB, Delekar SD. Silver nanoparticles as an effective disinfectant: A review. Mater Sci Eng C Mater Biol Appl. 2019;97:954-965. doi: 10.1016/j.msec.2018.12.102.
- Ganesh Kumar A, Pugazhenthi E, Sankarganesh P, Muthusamy C, Rajasekaran M, Lokesh E, Khusro A, Kavya G. Cleome rutidosperma leaf extract mediated biosynthesis of silver nanoparticles and anti-candidal, anti-biofilm, anti-cancer, and molecular docking analysis. Biomass Convers Bioref. 2023;1-13. doi: 10.1007/s13399-023-03806-9.
- De Matteis V, Rizzello L, Ingrosso C, Liatsi-Douvitsa E, De Giorgi ML, De Matteis G, Rinaldi R. Cultivar-Dependent Anticancer and Antibacterial Properties of Silver Nanoparticles Synthesized Using Leaves of Different Olea Europaea Trees. Nanomaterials (Basel). 2019 Oct 30;9(11):1544. doi: 10.3390/nano9111544. PMID: 31671618; PMCID: PMC6915347.
- Remya RR., Julius A, Suman TY, Aranganathan L, Dhas TS, Mohanavel V, Muhibbullah M. Biofabrication of silver nanoparticles and current research of its environmental applications. Journal of Nanomaterials. 2022;1-11. doi: 10.1155/2022/2670429.
- Ietswaart JH. A taxonomic revision of the genus Origanum (Labiatae). Leiden Botanical Series, Netherlands: Leiden University Press. 1980;4(1):1-153.
- Canli K, Bozyel ME, Turu D, Benek A, Simsek O, Altuner EM. Biochemical, Antioxidant Properties and Antimicrobial Activity of Steno-Endemic Origanum onites. Microorganisms. 2023 Aug 2;11(8):1987. doi: 10.3390/microorganisms11081987. PMID: 37630547; PMCID: PMC10457892.
- Sargın SA, Akçicek E, Selvi S. An ethnobotanical study of medicinal plants used by the local people of Alaşehir (Manisa) in Turkey. J Ethnopharmacol. 2013 Dec 12;150(3):860-74. doi: 10.1016/j.jep.2013.09.040. Epub 2013 Oct 11. PMID: 24126062.
- Tepe B, Cakir A, Sihoglu Tepe A. Medicinal Uses, Phytochemistry, and Pharmacology of Origanum onites (L.): A Review. Chem Biodivers. 2016 May;13(5):504-20. doi: 10.1002/cbdv.201500069. PMID: 27062715.
- Kotronia M, Kavetsou E, Loupassaki S, Kikionis S, Vouyiouka S, Detsi A. Encapsulation of Oregano (Origanum onites L.) Essential Oil in β-Cyclodextrin (β-CD): Synthesis and Characterization of the Inclusion Complexes. Bioengineering (Basel). 2017 Sep 9;4(3):74. doi: 10.3390/bioengineering4030074. PMID: 28952553; PMCID: PMC5615320.
- Salayová A, Bedlovičová Z, Daneu N, Baláž M, Lukáčová Bujňáková Z, Balážová Ľ, Tkáčiková Ľ. Green Synthesis of Silver Nanoparticles with Antibacterial Activity Using Various Medicinal Plant Extracts: Morphology and Antibacterial Efficacy. Nanomaterials (Basel). 2021 Apr 14;11(4):1005. doi: 10.3390/nano11041005. PMID: 33919801; PMCID: PMC8070782.
- Gecer EN. Synthesis and characterization of silver nanoparticles using Origanum onites leaves: Cytotoxic, apoptotic, and necrotic effects on Capan-1, L929, and Caco-2 cell lines. Green Process Synth. 2023;12(1):20228126. doi: 10.1515/gps-2022-8126.
- Shaik MR, Khan M, Kuniyil M, Al-Warthan A, Alkhathlan HZ, Siddiqui MRH, Adil SF. Plant-extract-assisted green synthesis of silver nanoparticles using Origanum vulgare L. extract and their microbicidal activities. Sustainability. 2018;10(4):913. doi: 10.3390/su10040913.
- Blois MS. Antioxidant determinations by the use of a stable free radical. Nature. 1958;181:1199-1200. doi: 10.1038/1811199a0.
- Doğan YS, Atasagun S, Ergönül MB. Determination of chemical content of Lemna minor L. by GC-MS and investigation of antioxidant activity. Commun Fac Sci Univ Ank Series C. 2022;31(1):53-64. doi: 10.53447/communc.1122558.
- Kotan R, Cakir A, Ozer H, Kordali S, Cakmakci R, Dadasoglu F, Kazaz C. Antibacterial effects of Origanum onites against phytopathogenic bacteria: Possible use of the extracts from protection of disease caused by some phytopathogenic bacteria. Sci Hortic. 2014;172:210-220. doi: 10.1016/j.scienta.2014.03.016.
- Tasdemir D, Kaiser M, Demirci B, Demirci F, Baser KHC. Antiprotozoal Activity of Turkish Origanum onites Essential Oil and Its Components. Molecules. 2019 Dec 3;24(23):4421. doi: 10.3390/molecules24234421. PMID: 31817023; PMCID: PMC6930659.
- Kondolot Solak E, Er A. pH-sensitive interpenetrating polymer network microspheres of poly(vinyl alcohol) and carboxymethyl cellulose for controlled release of the nonsteroidal anti-inflammatory drug ketorolac tromethamine. Artif Cells Nanomed Biotechnol. 2016 May;44(3):817-24. doi: 10.3109/21691401.2014.982805. Epub 2015 Jan 24. PMID: 25619756.
- Sharifi-Rad M, Berkay Yılmaz Y, Antika G, Salehi B, Tumer TB, Kulandaisamy Venil C, Das G, Patra JK, Karazhan N, Akram M, Iqbal M, Imran M, Sen S, Acharya K, Dey A, Sharifi-Rad J. Phytochemical constituents, biological activities, and health-promoting effects of the genus Origanum. Phytother Res. 2021 Jan;35(1):95-121. doi: 10.1002/ptr.6785. Epub 2020 Aug 12. PMID: 32789910.
- Erenler R, Dag B. Biosynthesis of silver nanoparticles using Origanum majorana L. and evaluation of their antioxidant activity. Inorg Nano-Met. 2021;52(4):485-492. doi: 10.1080/24701556.2021.1952263.
- Gutierrez-Gines MJ, Alizadeh H, Alderton E, Ambrose V, Meister A, Robinson BH, Horswell J. Phytoremediation of microbial contamination in soil by New Zealand native plants. App Soil Ecol. 2021;167:104040. doi: 10.1016/j.apsoil.2021.104040.
- Jang J, Hur HG, Sadowsky MJ, Byappanahalli MN, Yan T, Ishii S. Environmental Escherichia coli: ecology and public health implications-a review. J Appl Microbiol. 2017 Sep;123(3):570-581. doi: 10.1111/jam.13468. Epub 2017 Jul 3. PMID: 28383815.
- Kondolot Solak E, Kaya S, Asman G. Preparation, characterization, and antibacterial properties of biocompatible material for wound healing. J Macromol Sci A. 2021;58:10:709-716. doi: 10.1080/10601325.2021.1929315
- Taylor TA, Unakal CG. Staphylococcus aureus Infection. In: StatPearls. StatPearls Publishing, Treasure Island (FL) PMID: 28722898. 2023.
- Lopes JP, Lionakis MS. Pathogenesis and virulence of Candida albicans. Virulence. 2022 Dec;13(1):89-121. doi: 10.1080/21505594.2021.2019950. PMID: 34964702; PMCID: PMC9728475.
- Gulati M, Nobile CJ. Candida albicans biofilms: development, regulation, and molecular mechanisms. Microbes Infect. 2016 May;18(5):310-21. doi: 10.1016/j.micinf.2016.01.002. Epub 2016 Jan 22. PMID: 26806384; PMCID: PMC4860025.
- Hermanns R, Cremers NAJ, Leeming JP, van der Werf ET. Sweet Relief: Determining the Antimicrobial Activity of Medical Grade Honey Against Vaginal Isolates of Candida albicans. J Fungi (Basel). 2019 Sep 9;5(3):85. doi: 10.3390/jof5030085. PMID: 31505796; PMCID: PMC6787731.
- Nourbakhsh F, Lotfalizadeh M, Badpeyma M, Shakeri A, Soheili V. From plants to antimicrobials: Natural products against bacterial membranes. Phytother Res. 2022 Jan;36(1):33-52. doi: 10.1002/ptr.7275. Epub 2021 Sep 16. PMID: 34532918.
- WHO. Antimicrobial resistance. 2023.
- Altintas A, Tabanca N, Tyihák E, Ott PG, Móricz AM, Mincsovics E, Wedge DE. Characterization of volatile constituents from Origanum onites and their antifungal and antibacterial activity. J AOAC Int. 2013 Nov-Dec;96(6):1200-8. doi: 10.5740/jaoacint.sgealtintas. PMID: 24645494.
- Kaskatepe B, Yildiz SS, Kiymaci ME, Yazgan AN, Cesur S, Erdem SA. Chemical composition and antimicrobial activity of the commercial Origanum onites L. oil against nosocomial carbapenem resistant extended spectrum beta lactamase producer Escherichia coli isolates. Acta Biol Hung. 2017 Dec;68(4):466-476. doi: 10.1556/018.68.2017.4.11. PMID: 29262709.
- Rai MK, Deshmukh SD, Ingle AP, Gade AK. Silver nanoparticles: the powerful nanoweapon against multidrug-resistant bacteria. J Appl Microbiol. 2012 May;112(5):841-52. doi: 10.1111/j.1365-2672.2012.05253.x. Epub 2012 Mar 28. PMID: 22324439.
- Sankar R, Karthik A, Prabu A, Karthik S, Shivashangari KS, Ravikumar V. Origanum vulgare mediated biosynthesis of silver nanoparticles for its antibacterial and anticancer activity. Colloids Surf B Biointerfaces. 2013 Aug 1;108:80-4. doi: 10.1016/j.colsurfb.2013.02.033. Epub 2013 Mar 4. PMID: 23537829.
- Benedec D, Oniga I, Cuibus F, Sevastre B, Stiufiuc G, Duma M, Hanganu D, Iacovita C, Stiufiuc R, Lucaciu CM. Origanum vulgare mediated green synthesis of biocompatible gold nanoparticles simultaneously possessing plasmonic, antioxidant and antimicrobial properties. Int J Nanomedicine. 2018 Feb 20;13:1041-1058. doi: 10.2147/IJN.S149819. PMID: 29503540; PMCID: PMC5824763.
- Hambardzumyan S, Sahakyan N, Petrosyan M, Nasim MJ, Jacob C, Trchounian A. Origanum vulgare L. extract-mediated synthesis of silver nanoparticles, their characterization and antibacterial activities. AMB Express. 2020 Sep 5;10(1):162. doi: 10.1186/s13568-020-01100-9. PMID: 32889670; PMCID: PMC7474311.
- Netchareonsirisuk P, Puthong S, Dubas S, Palaga T, Komolpis K. Effect of capping agents on the cytotoxicity of silver nanoparticles in human normal and cancer skin cell lines. J Nanopart Res. 2016;18:322. doi: 10.1007/s11051-016-3624-6.
- Xu L, Wang YY, Huang J, Chen CY, Wang ZX, Xie H. Silver nanoparticles: Synthesis, medical applications and biosafety. Theranostics. 2020;10(20):8996-9031. doi: 10.7150/thno.45413.
- Ahmed MJ, Murtaza G, Rashid F, Iqbal J. Eco-friendly green synthesis of silver nanoparticles and their potential applications as antioxidant and anticancer agents. Drug Dev Ind Pharm. 2019 Oct;45(10):1682-1694. doi: 10.1080/03639045.2019.1656224. Epub 2019 Sep 2. PMID: 31407925.
- Khorrami S, Zarrabi A, Khaleghi M, Danaei M, Mozafari MR. Selective cytotoxicity of green synthesized silver nanoparticles against the MCF-7 tumor cell line and their enhanced antioxidant and antimicrobial properties. Int J Nanomedicine. 2018 Nov 27;13:8013-8024. doi: 10.2147/IJN.S189295. PMID: 30568442; PMCID: PMC6267361.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.