General Science Group. 2024 September 30;5(9):1229-1237. doi: 10.37871/jbres2010.
Extremely Low Activation Energies of Anti-perovskite Na3OX Solid Electrolytes
Wei Shi1*, Yuki Osaki1, Mariko Murayama1,2, Kenji Hashimoto3 and Xinwei Zhao1,4*
2Faculty of Science and Engineering, Toyo University, Kawagoe-shi, Saitama 350-8585, Japan
3KRI, Inc., Kyoto, 6008813, Japan
4International Institute for Urban Systems Engineering, Southeast University, Nanjing 210096, China
Abstract
Solid Electrolytes (SE) for all solid Sodium-Ion Batteries (SIB) have been attracting much attention as an alternative energy storage system, which have the advantage of being extremely safe because it can be charged quickly and is nonflammable. We have synthesized anti-perovskite type Na3OX (X = Br, and I) electrolytes with high purity. It was confirmed that a large crystal strain was introduced by eutecticization, which reduced the activation energy of Na ion conduction and lead to an improvement of the electric conductivity. In our previous work, an ionic conductivity of σ = 1.55x10-7 S/cm for Na3OBr0.6I0.4, and the activation energy was also reduced from 1.14eV to 0.64eV. These values, however, are still poor for a practical application as SE. In this paper, we focused on the Na3OBr1-xIx (x = 0 to 0.4) series and increased the synthesizing time from 3x6 to 3 x 9 hours. An extremely low activation energy of 0.25eV and high ionic conductivity of σ = 2.6x10-3 S/cm at 90oC and have been observed.
Introduction
There have been considerable attentions on secondary ion batteries due to the industry developments on computers, smartphones, and Electric Vehicles (EV). Lithium-Ion Batteries (LIB) are now the main product using in wide areas. However, Li elements reserves on the earth is limited and is facing to gradual reduction or depletion with the widespread use of lithium battery technologies and increasing demand for LIBs on EV related industry. Thus, the cost of Li resource has turned more and more expensive in recent years and tending to keeping increasing in the visible future with the worldwide energy crisis. It is urgent to find an alternative energy source to compensate for lacking of Li resource storage. As the cost of LIB increase as well as energy resource risk and environmental problems rise, sodium-ion batteries (SIB) have been attracting much attention as an alternative energy storage system in recent years. The price advantage of sodium (Na) is also attractive with the widespread use of electric vehicles. Comparing with lithium (Li), sodium (Na) has its price advantage in high volume demand for high-density and large-capacity batteries with the widespread use of EV vehicles. Therefore, Na, as the same group element of Li, considered as an alternative choice instead of Li because it has the similar chemical characters with Li while easy to be obtained at relatively lower cost.
Especially, Solid electrolytes (SE) for all solid sodium-ion batteries (SIB) have been attracting much attention as an alternative energy storage system, which have the advantage of being extremely safe because it can be charged quickly and is nonflammable. In the research with higher voltage, higher capacity and stable cycle characteristics, synthesizing materials such as electrodes and electrolytes are important topics. Though Na and Li has the similar characters, SIB couldn’t get the same level high conductivity as LIB did in the past experience. Therefore, it is urgent to improve the performance of Na-SE material (application requirement: σ> 10⁻³ in RT), which is relatively low at present.
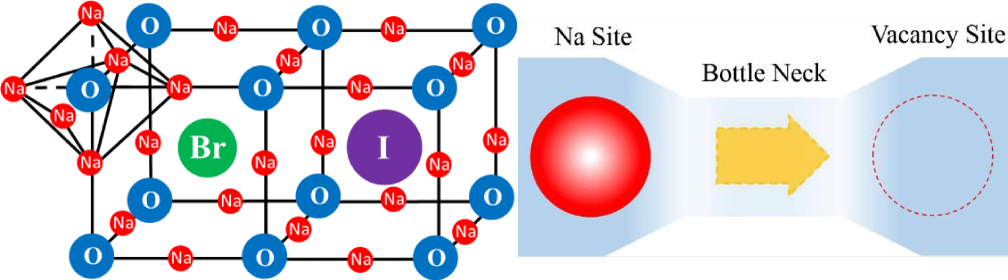
Our lab is researching on synthesis and conductivity improvement of anti-perovskite Na3OX solid electrolytes. Na rich anti-perovskite structure is proved to a promising structure in which Na ionic conductivity can be improved up to a relatively high level compared with others. Figure 1(a) shows the Na-rich anti-perovskite structure, Na3OX (X = Br, I), in this structure, Na+ is easy to break away from Na-O band and moving through spare space of crystal. This movement carry and transfer electrons. The ability of how easy it can move and how many electrons it can transfer represent its ionic conductivity. On the other hand, there are the narrowest corridors in spare spaces, which is called “Bottle Neck”, as shown by figure 1(b). It affects the movement of Na+. The lowest energy supplied to pass bottlenecks called activity energy. In another word, the conductivity could be improved by decreasing the activity energy. The important thing is how to improve its conductivity in order to developing applicable Na-rich Anti-perovskite structure solid electrolyte.
We have synthesized anti-perovskite type Na3OX (X = Br, and I) electrolytes with high purity. As shown in figure 1(a), an increased lattice distortion leading to lower activation energy has been predicted by theoretical calculations. We added different ratio of X (X = Br, I) ions into sodium oxide to make high-purity synthetic Na3OX crystal, then using X-ray diffraction (XRD) measurements to identify their crystal structure and material purity. It was confirmed that all compositions of Na3OBr1-xIx (x = 0 to 0.4) had the anti-perovskite structure with Pm-3m cubic symmetry. It is also demonstrated that the impurity peaks could hardly be confirmed. In our previous work, it was confirmed that a large crystal strain was introduced by eutecticization, which reduced the activation energy of Na ion conduction and lead to an improvement of the electric conductivity. An ionic conductivity of σ = 1.55x10⁻7 S/cm for Na3OBr0.6I0.4, which is about three orders higher than that in literature. The activation energy was also reduced from 1.14eV to 0.64eV. These values, however, are still poor for a practical application as SE. One reason is the residual reacting materials after the synthesizing and the inhomogeneity of the grain size.
Experimental Methodsc
Materials synthesis
We added different ratio of I ions into Na3OBr1-xIx (x = 0 to 0.4) series and increased the synthesizing time from 3 x 6 to 3 x 9 hours. Then using X-ray diffraction (XRD) measurements to identify the performance of its purity. And we use the Williamson-Hall measurement to evaluate its lattice distortion. Impedance measurement is used to analyze their conductive activation energy.
The SE Na3OX (X = Br, I) were synthesized by following reactions.
0.8Na2O+0.2Na2O2+(1-x) NaX2+x NaX2→→Na3OX11−𝑥X2+0.1O2↑ (1)
Different with previous work, we improved experimental conduction this time. First, NaX (X = Cl, Br, I) was crushed in a glove box filled with N2. Then quickly grind sodium oxide (0.8Na2O+0.2Na2O2) was added with the above halogen mixture. After mixing, it was filled in an alumina crucible and heated for about 9 hours at 330oC. Then, in order to promote the reaction, the obtained sintered body was re-crushed and reheated to obtain a pure reacting material.
Measurement methods
XRD measurement: An XRD system Ultima IV manufactured by Rigaku was used for the measurement. X-ray characteristics of CuKα1 (λ = 1.5406 Å) and CuKα2 (λ = 1.5443 Å) were used, mixed to create CuKα radiation (λ = 1.5418 Å). The tube voltage was set at 40 kV, and the tube current was either 20 mA or 40 mA. Measurements were taken at room temperature in the range of about 2θ = 15 ° to 90° with a step width of 0.01°. Na3OBr was also measured as background and the samples Na3OX (X = Br, Br0.8I0.2, Br0.7I0.3, Br0.6I0.4) were measured.
Impedance measurement: AC impedance measurements were made in cooperation with KRI, Inc. The crushed sample powder was placed in a measurement cell (inner diameter 10 mm) and pressed in an Ar glove box at a hydraulic pressure of about 2.0 tons (about 250 MPa) for 15 minutes to form pellets with a thickness of about 0.72mm (Br0.8I0.2), 0.75mm (Br0.7I0.3) and 0.79mm (Br0.6I0.4), respectively. The AC impedance characteristic was carried out by the sandwiched two-terminal method using a blocking electrode and evaluated with an instrument VersaSTAT4 manufactured by Princeton Applied Research. The measurements were conducted in the frequency range of 1MHz to 0.1Hz. Then, the ionic conductivity evaluation cell was secured with a torque wrench at a force of 8N and placed inside a sealed container. The sealed container was then placed in a constant temperature bath, and the ionic conductivity was measured after one hour at each measurement temperature (25°C, 50°C, 70°C, 90°C, 110°C, 130°C and 150℃) in a frequency range of 1MHz to 0.1Hz. The whole measurements were carried out in the same glove box (dew point below -80°C) to avoid the deoxidization of the samples. The samples Na3OX (X = Br, Br0.8I0.2, Br0.7I0.3, Br0.6I0.4) were measured.
SEM-EDS measurement and analysis. Scanning Electron Microscopy (SEM) and Energy Dispersive X-ray Spectroscopy (EDS) measurements were performed to evaluate the surface state of the sample and the elemental composition and distribution within the sample. SEM Regulus 8230 manufactured by Hitachi and EDS Ultim Max manufactured by Oxford Instrument, supported by Industrial Technology Research Institute at Toyo University, were used for the measurement. SEM utilized accelerated electron beams to scan the surface of a sample to a depth of approximately 10 nm, detecting signals generated from the sample and obtain its SEM image, which showed the morphology and composition of small area on the surface. EDS detected a variety of characteristic X-rays generated by the electron beam's irradiation and measured their energy and occurrence frequency, by which the elements compositions and their concentrations were determined.
Results and Discussion
XRD measurement results
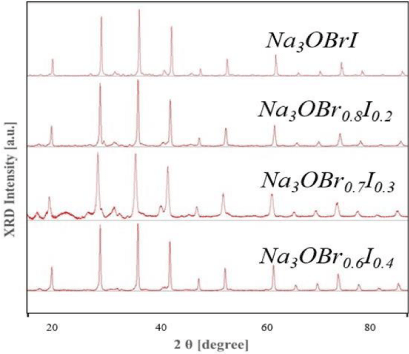
XRD patterns: The XRD patterns for x = 0, 0.2, 0.3, and 0.4 are shown in figure 2. Samples with 4 different ratio compositions were synthesized with a sintering temperature of 330°C and a sintering time of 9 hours, repeated 3 times. As a result, no peaks from precursor materials or by-products other than Na3OBr1-xIx were observed for x = 0.2, 0.4, indicating successful synthesis with high purity. For x = 0.3, tiny waves other than Na3OBr1-xIx were observed, but those waves were more like noises by experimental conditions rather than peaks from by- products. It could still be considered that high purity Na3OBr0.7I0.3 was successful synthesized. All the samples were used in the follow-up measurements and analysis.
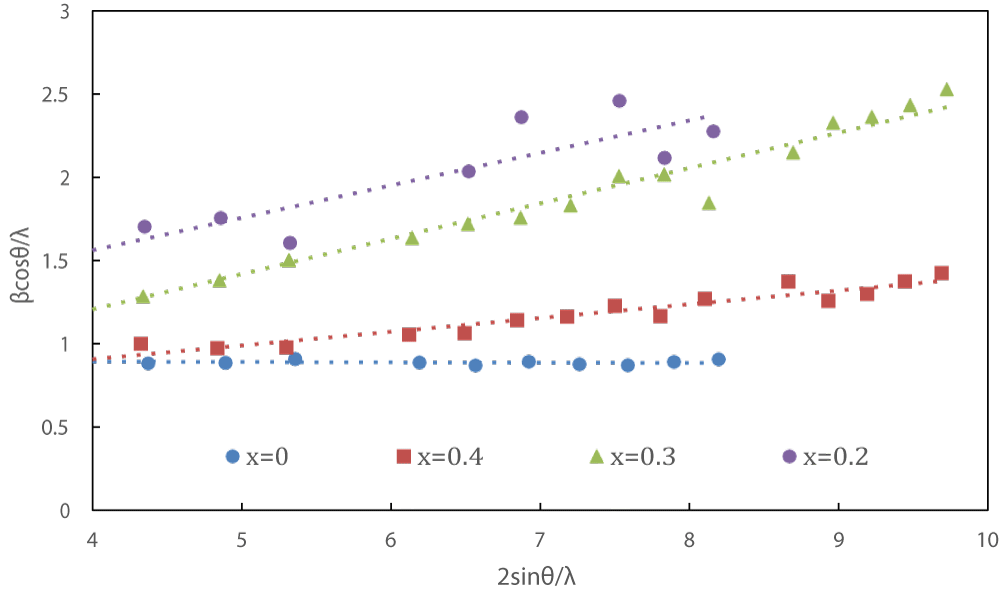
Williamson-Hall Plot: Williamson and Hall proposed a method for obtaining size and lattice strain by considering peak width as a function of 2θ.
Here, D represents the crystal size, β is the full width at half maximum (FWHM), K is the Scherrer constant according to the crystal shape, ε is lattice strain, and λ is the wavelength of X-rays.
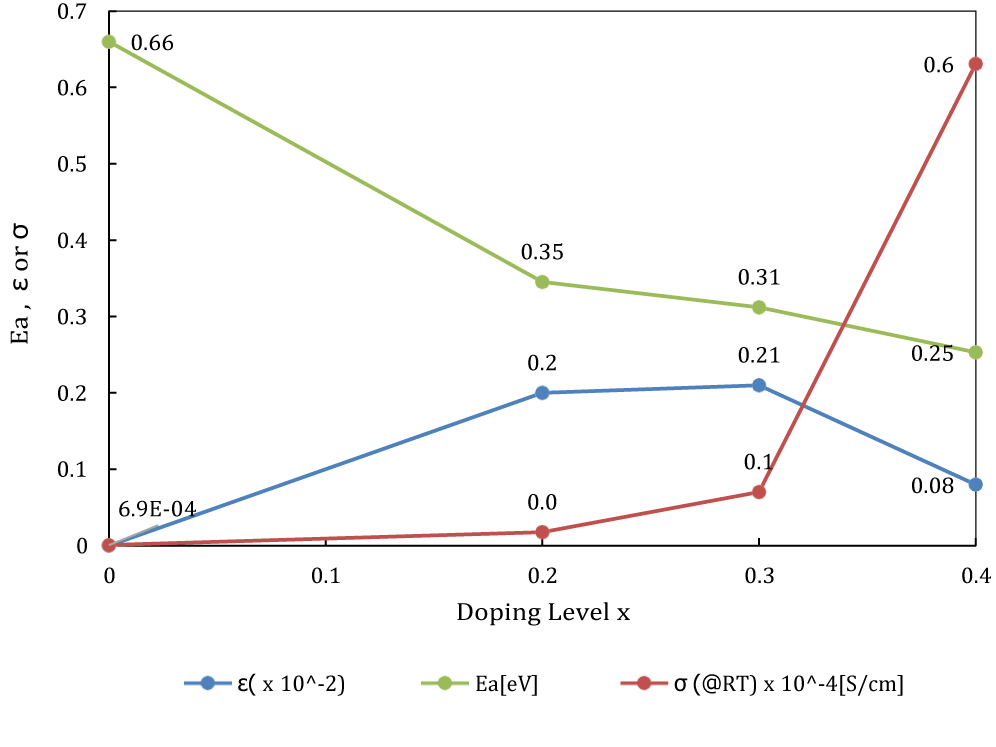
By plotting βcosθ/λ on y axis against 2sinθ/λ on x axis, we get the Williamson Hall plot (Figure 3). Using Williamson Hall plot, the lattice strain ε of each composition was calculated (from the slope). Table 1 shows the lattice strain value of each sample. The lattice strain is around 0.2 at x = 0.2, 0.3, by comparison, lattice strain at x = 0.4 is only 0.08. Noticed in previous work the highest lattice strain appeared at x = 0.2 among the range of x = 0.1~0.4, it may indicate the lattice strain reaches to a peak value at somewhere of range x = 0.2 ~ 0.3 and then drop down to 0.08 till x = 0.4. At least, Na3OBr0.6I0.4 does not obtain the highest lattice strain among all the I doping ratios, it will be discussed further.
| Table 1: Lattice Strain of experimental compositions. | |
| Composition | Lattice Strainε |
| Na3OBr0.8I0.2 | 0.2 |
| Na3OBr0.7I0.3 | 0.21 |
| Na3OBr0.8I0.4 | 0.08 |
Impedance measurement
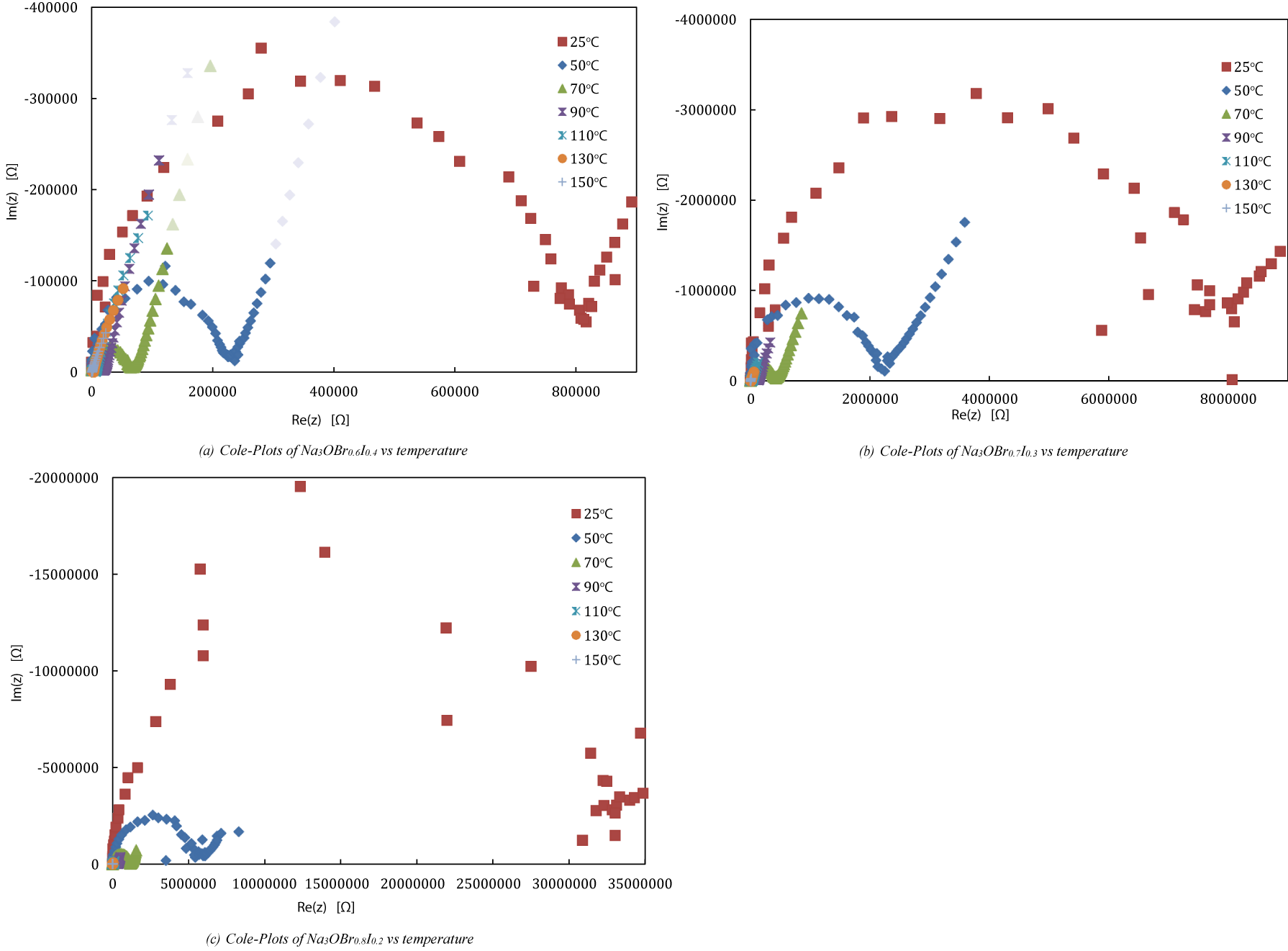
Cole-plots: The impedance measurements have been carried out for Na3OBr0.8I0.2, Na3OBr0.7I0.3, Na3OBr0.6I0.4 samples from room temperature (RT) to 150℃. As shown in figure 4(a), Na3OBr0.6I0.4 sample gives a series of Cole-plots from RT to 150℃, the resistances (corresponding to diameter of the semicircle) decrease with temperature increasing. The highest resistances of Na3OBr0.6I0.4 is obtained at RT. Although Na3OBr0.8I0.2 didn’t give a perfect Cole-Plot at RT, an approximate resistance value at RT can still be estimated based on curve trend, which is around 3.5 x 107Ω (Figure 4(c)). Compared with resistance values shown in figure 4(a), (b) and (c), it is easy to demonstrated that Na3OBr0.6I0.4 have the lowest bulk resistance Rb at RT as well at other temperatures. It indicates a higher ionic conductivity of Na3OBr0.6I0.4 at RT compared with two others. The ionic conductivity (σ) [S/cm] can be determined by the following equation:
Here, Rb represents the bulk resistance of sample [Ω], L represents the thickness of the measured sample [cm], and S is the cross-sectional area of the sample [cm2].
Using the above equation, the ionic conductivity of Na3OBr0.8I0.2, Na3OBr0.7I0.3, Na3OBr0.6I0.4 were calculated and shown in table 2. The ionic conductivity of each composition (x = 0.2, 0.3, 0.4) shows an obvious increasing with temperature rising. The higher conductivity, which is the expectation in this research, will be obtained in higher temperature. But here we would like to fucus on ionic conductivity in room temperature, because actual operating temperature for secondary batteries is often at/around room temperature. Therefore, in this study, the ion conductivity at room temperature for each sample was emphasized and predicated according to experimental data.
The ion conductivity of pure Na3OBr at room temperature (25°C) is 𝜎 = 6.9×10⁻⁸ S/cm. In contrast, for samples doped with I ions, x = 0.4 had an ion conductivity of 𝜎 = 6.3 x 10-5 S/cm, while x=0.3 and x = 0.2 had ion conductivities at room temperature of 𝜎 = 7.0 x 10-6 S/cm and 𝜎 = 1.8×10-6 S/cm, respectively (Table 2). All the Na3OBr1-xIx (x = 0.2, 0.3, and 0.4) samples showed low ionic conductivity nearby RT, comparing with our previous work. The lowest conductivity was obtained at RT from either of 3 samples. These values at room temperature are still relatively high among solid electrolytes with Na ion conductivity.
| Table 2: Ionic conductivity calculated by equation (3). | |||
| σ | Na3OBr0.6I0.4 | Na3OBr0.7I0.3 | Na3OBr0.8I0.2 |
| 25OC | 6.3 x 10-5 | 7.0 x 10-6 | (1.8 x 10-6) |
| 50OC | 2.3 x 10-4 | 2.5 x 10-5 | 1.0 x 10-5 |
| 70OC | 7.9 x 10-4 | 1.3 x 10-4 | 5.2 x 10-5 |
| 90OC | 2.6 x 10-3 | 4.3 x 10-4 | 1.7 x 10-4 |
| 110OC | 7.8 x 10-3 | 1.7 x 10-3 | 1.0 x 10-3 |
| 130OC | 1.6 x 10-2 | 5.2 x 10-3 | 4.3 x 10-3 |
| 150OC | 4.9 x 10-2 | 2.9 x 10-2 | 1.5 x 10-2 |
| *Note: (1.8 x 10-6) data in () is predicted value from plot trend. | |||
Furthermore, comparing the results with different I ratio samples Na3OBr1-xIx (x = 0.2, 0.3, and 0.4) at the same temperature, the highest ionic conductivity was obtained from the Na3OBr0.6I0.4 powder at RT as well as other temperatures, which is considered to give a lower activity energy.
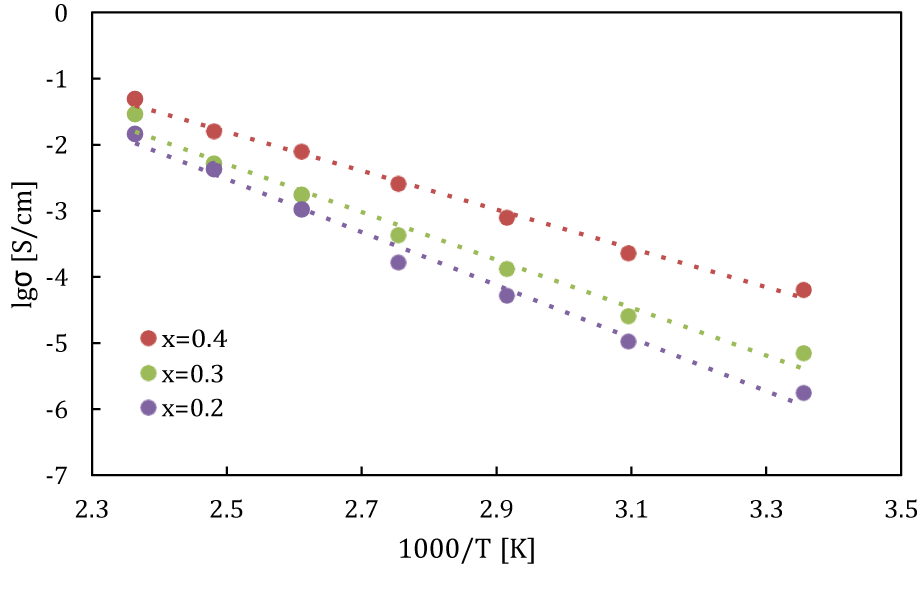
Arrhenius plot: By analyzing above series of ionic conductivity vs temperature using Arrhenius equation, the activation energy used by ion movement can be clarified. Arrhenius equation is an empirical equation and represents the temperature dependence of ionic conductivity 𝜎, described as below:
In equation (4), 𝜎 is ionic conductivity [S/cm], A is the pre-exponential factor, Ea is the activation energy (in the same units as kT), k is Boltzmann constant (k≈1.380649×10−23(J/K)), and T is absolute temperature [K].
When the activation energy for ion conduction is low, it means that ions can easily conduct even at low temperatures. By plotting log10 𝜎 on y axis against 1000/T on x axis, we get an Arrhenius plot. This plot is used to calculate (from the slope) the activation energy Ea of each composition for carrier ion diffusion from the gradient of the data points on the graph. The plot of each composition is shown in figure 5 and activity energy calculated from figure 5 (as well as previous related results for comparation) shows in table 3. The activation energy values Ea = 0.66 eV for pure Na3OBr, while in the case of samples doped with I ions, Ea = 0.25 eV (x = 0.4), 0.31 eV(x = 0.3) and Ea = 0.35 eV (x = 0.2), respectively. The Na3OBr0.6I0.7 powder gives an extremely low activation energy of 0.25eV. It is almost a half value of our previous work and is comparable with that of a lithium SE.
| Table 3: Activity Energy of Na3OBr1-xIx (x=0, 0.2, 0.3, 0.4) solid electrolytes. | ||
| Composition | ΔEa(eV) | Heating Time t(h) |
| Na3OBr | 0.66 | 3 x 9h |
| Na3OBr0.6I0.4 | 0.64 | Previous research, t=3 x 6h |
| Na3OBr0.8I0.2 | 0.35 | 3 x 9h |
| Na3OBr0.7I0.3 | 0.31 | 3 x 9h |
| Na3OBr0.6I0.4 | 0.25 | 3 x 9h |
SEM-EDS Analysis
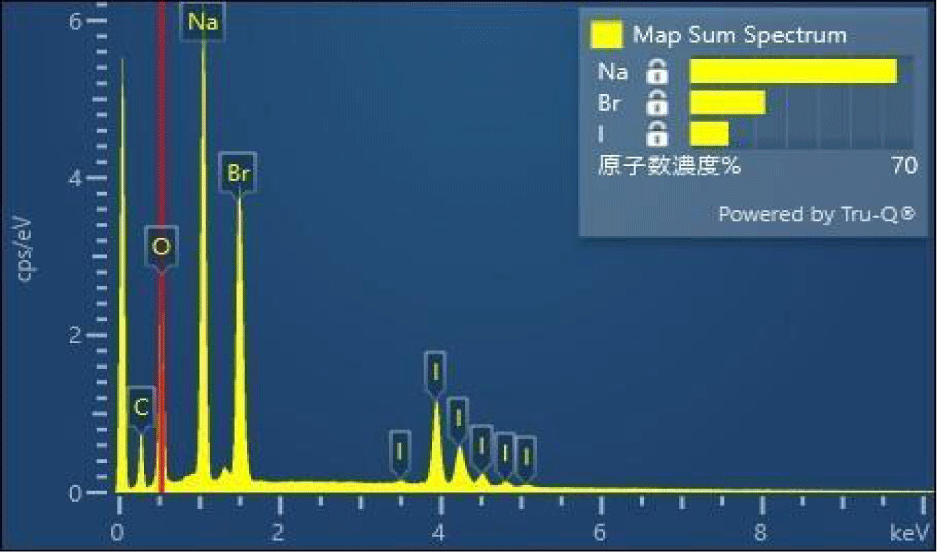
SEM Analysis: Figure 6(a) shows the SEM image of the Na3OBr0.6I0.4 powder, the homogeneous grain size was confirmed. The energy dispersive X-ray spectroscopy (EDS) results were shown in figure 6(b).The distributions of Na, Br, I and O are same and have a similar distribution as the grain shape shown by the SEM picture. This homogeneity should be an important factor on increasing Na ion conductivity in the Na- rich anti-perovskite solid electrolytes.
EDS Analysis: The element ratio of the Na3OBr0.6I0.4 SE is shown in figure 7. The Na-rich SE has been successfully obtained. Remember that we are trying to fabricate the Na-rich anti-perovskite solid electrolytes, which have as more as 74% Na ions than that of a perovskite structure. Indeed, as can be seen from the EDS spectra, the atomic ratio of Na in near to 65%, with ~22% Br and ~13% I ions, respectively. Although there is no standard sample for Na3OBr1-xIx crystal, it is suggested that the Na-rich anti-perovskite Na3OBr1-xIx solid electrolytes have been successfully synthesized. The extremely low conductivity activation energy and high ionic conductivity at RT clearly indicate that Na3OBr1-xIx solid electrolytes are promise candidates in all solid SIB applications.
Discussion
Because the strain introduction optimizing the bottleneck and destabilizing the stronger Na-O bond, a reduction in ion transfer energy and defect formation energy should be confirmed.
It had been demonstrated that the Br-I mixture introduced additional lattice distortion in the eutectic, which led to a large reduction of the activation energy. But the reduction of the activation energy was not completely following with the distortion increase. As shown in figure 8, Na3OBr0.6I0.4 does not obtain the highest lattice strain among all the I doping ratios. But Na3OBr0.6I0.4 powder gives a highest ionic conductivity at RT and lowest activity energy. It might be due to the effect of polarizability of the introduced X ions. When the ligand ions constituting the bottleneck are highly polar in the case of Na ion diffusing, they are flexibly deformed even if the bottleneck radius is narrow, and the Na ions could push away the ligands so that they could diffuse with low energy.
Considering the relationship between activity energy and ionic conductivity, the activity energy from x = 0.3 to 0.4 decreased only 0.06 eV but ionic conductivity increased remarkable, comparing with change from x = 0.2 to 0.3. Even we know that it is not a liner relationship, still it is an interesting topic which factors make the ionic conductivity change in this way.
Finding a compositional balance that maximizes the contribution of both strain and polarity should be an effective way to improve the conductivity further. It is also considered that the impedance measurement result itself changes with the surrounding environment and the pellet preparation method, which will be presented in the conference.
Conclusion
This research succeeded in synthesizing Na3OBr1-xIx (x = 0.1~0.4) with high purity and it was confirmed that a large crystal strain was introduced by eutecticization. An extremely low activation energy of 0.25eV and high ionic conductivity of σ = 2.6x10-3 S/cm at 90℃ and σ = 6.3x10-5 S/cm at RT for Na3OBr0.6I0.4 have been observed. A significant improvement up to several orders was achieved compared with previous work. Especially, the Na3OBr0.6I0.4 powder gives an extremely low activation energy of 0.25eV. It is almost a half value of our previous work and is comparable with that of a lithium SE [1-18].
Acknowledgement
My most profound appreciation goes to Dr. GUAN Sujun for sharing his pearls of wisdom on the research direction, and his kindly comments and help during lab experiment that greatly improved the manuscript. Mr. Ohda his previous research and ideas in his paper always give some inspiration during this work. We would also like to thank the Faculty of Industrial Technology Research Institute at Toyo University and company KRI for providing me with the measurement instruments, which let us complete the experiments successfully.
References
- Kubota K, Komaba A. Review-Practical Issues and Future Perspective for Na-Ion Batteries. J Electrochem Soc. 2015;162:A2538-A2550. doi: 10.1149/2.0151514jes.
- Palomares V, et al. Na-ion batteries, recent advances and present challenges to become low cost energy storage systems. Energy Environ Sci. 2012;5:5884-5901. doi: 10.1039/C2EE02781J.
- Hwang JY, Myung ST, Sun YK. Sodium-ion batteries: present and future. Chem Soc Rev. 2017 Jun 19;46(12):3529-3614. doi: 10.1039/c6cs00776g. PMID: 28349134.
- Slater MS, Kim D, Lee E, Johnson CS. Sodium-Ion Batteries. Adv Funct Mater. 2013;23:947-958. doi: 10.1002/adfm.201200691.
- Yabuuchi N, Kubota K, Dahbi M, Komaba S. Research development on sodium-ion batteries. Chem Rev. 2014 Dec 10;114(23):11636-82. doi: 10.1021/cr500192f. Epub 2014 Nov 12. PMID: 25390643.
- Wang Y, et al. Structural manipulation approaches towards enhanced sodium ionic conductivity in Na-rich antiperovskites. J Power Sources. 2015;293:735. doi: 10.1016/j.jpowsour.2015.06.002.
- Sun Y, Wang Y, Liang X, Xia Y, Peng L, Jia H, Li H, Bai L, Feng J, Jiang H, Xie J. Rotational Cluster Anion Enabling Superionic Conductivity in Sodium-Rich Antiperovskite Na3OBH4. J Am Chem Soc. 2019 Apr 10;141(14):5640-5644. doi: 10.1021/jacs.9b01746. Epub 2019 Mar 29. PMID: 30912936.
- Gao S, Broux T, Fujii S, Tassel C, Yamamoto K, Xiao Y, Oikawa I, Takamura H, Ubukata H, Watanabe Y, Fujii K, Yashima M, Kuwabara A, Uchimoto Y, Kageyama H. Hydride-based antiperovskites with soft anionic sublattices as fast alkali ionic conductors. Nat Commun. 2021 Jan 8;12(1):201. doi: 10.1038/s41467-020-20370-2. PMID: 33420012; PMCID: PMC7794446.
- James AD, Hungru Chen, Saiful Islam M. Composition Screening of Lithium and Sodium- Rich Anti-Perovskites for Fast-Conducting Solid Electrolytes. J Phys Chem C. 2018;122:23978-23984. doi: 10.1021/acs.jpcc.8b08208
- Wei Shi, Masataka Ohta, Hiroaki Asakawa, Yuki Osaki, Mariko Murayama, Xinwei Zhao. Synthesis and Conductivity Characterization of Anti-Perovskite Na3OX Solid Electrolytes for All Solid Na-Ion Batteries. Optics and Photonics Journal. 2023;13:189-198. doi: 10.4236/opj.2023.137017.
- Dawson JA, Famprikis T, Johnston KE. Anti-perovskites for solid-state batteries: recent developments, current challenges and future prospects. J Mater Chem A. 2021;9:18746-18772. doi: 10.1039/D1TA03680G.
- Takashi Ida. Chemical analysis using powder X-ray diffraction method (nitech.ac.jp) (Japanese).
- Izumi Nakai, Fujio Izumi. Practical Powder X-ray Structural Analysis Introduction to Rietveld Method (Japanese).
- Altomare A, et al. EXPO2013: a kit of tools for phasing crystal structures from powder data. J Appl Cryst. 2013;46:1231-1235. doi: 10.1107/S0021889813013113.
- Kobayashi K, Suzuki TS. Free analysis and visualization programs for electrochemical impedance spectroscopy coded in Python. Electrochem. 2021. doi: 10.5796/electrochemistry.21-00010.
- Ernest Ahiavi, James A Dawson, Ulas Kudu, Matthieu Courty, Saiful Islam M, et al. Mechanochemical synthesis and ion transport properties of Na3OX (X = Cl, Br, I and BH4) antiperovskite solid electrolytes. J Power Sources. Elsevier. 2020;471:228489. doi: 10.1016/j.jpowsour.2020.228489.
- Kwangnam Kim, Donald J Siegel. Correlating lattice distortions, ion migration barriers, and stability in solid electrolytes. J Mater Chem A. 2019;7:3216. doi: 10.1039/C8TA10989C.
- Yasutoshi Saito, Toshio Maruyama. JME Materials Science High ionic conductivity in solids (Japanese).
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.