General Science Group. 2024 September 20;5(9):1187-1199. doi: 10.37871/jbres2007.
Exploring the Potential of Xanthatin from Cuban Xanthium strumarium: Isolation, Characterization, and Biological Evaluation
Sergey P Verevkin1,2*, Kseniya V Zherikova3, Sergey V Vostrikov4 and Irina I Kaygorodova4
2Department of Physical Chemistry, Kazan Federal University, 420008 Kazan, Russian Federation, Russia
3Nikolaev Institute of Inorganic Chemistry of Siberian Branch of Russian Academy of Sciences, 630090 Novosibirsk, Russian Federation, Russia
4Engineering and Technology Department, Samara State Technical University, 443100 Samara, Russia
- Drugs
- Enthalpies of phase transitions
- Vapour pressure
- Structure-property correlation
Abstract
Thermodynamics is crucial for optimizing drug synthesis, developing pharmaceutical formulations and ensuring their stability, effectiveness and safety. The thermochemical properties of active pharmaceutical ingredients with the common biphenyl motif (RS-Flurbiprofen, Diflunisal, Fenbufen and Biprofen) available in the literature were collected, combined with our own complementary experimental results and evaluated. The vapour pressures temperature dependence of RS-Flurbiprofen was measured using the Knudsen effusion method, and the enthalpy of sublimation was obtained from this measurement. The enthalpy of fusion of RS-Flurbiprofen was measured using DSC. A reliable diagnostic method has been outlined to evaluate the quality of the available experimental thermodynamic data of drugs. The data previously available in the literature for RS-flurbiprofen, diflunisal and fenbufen were analysed and diagnosed as "thick". For RS-flurbiprofen, diflunisal, fenbufen and biprofen, the consistent sets of thermodynamic data were evaluated and recommended for the calculations of the pharmaceutic processes. The "paper-and-pencil" concept presented in this paper can be extended to the diagnosis of "sick" or "healthy" experimental thermodynamic data for pharmaceuticals with a structure other than profens.
List of symbols
: Standard molar enthalpy of vaporisation; : Standard molar enthalpy of sublimation; : Standard molar enthalpy of fision; Tfus: Melting temperature; Ta: Ambient temperature; pi: Absolute vapor pressure; pref: Reference vapour pressure; : Standard molar heat capacity at constant pressure; : Difference of the standard molar heat capacities at constant pressure for the gaseous and crystal phase respectively; : Difference of the stamdard molar heat capacities at constant pressure for the gaseous and liquid phase respectively; Jx: Kovats index.
Introduction
Information on the thermodynamics of the phase transition in pharmacy is important for the correct characterisation of Active Pharmaceutical Ingredients (API) in pre-formulation and formulation studies. It is necessary that the sublimation, vaporisation and fusion energetics of the individual components are reliably quantified for a given formulation [1]. Knowing the solubility of a compound is important for predicting its dissolution, the medium to be used for dissolution and the solvents to be used for purification during production. The approximation and prediction of solubility data is also based on thermodynamic data (enthalpies of fusion, enthalpies of vaporisation or sublimation [2]). Moreover, enthalpies of phase transitions and vapour pressures are essential for the characterisation of polymorphic transformations of APIs [3]. Polymorphs of a drug have different molecular packing, which leads to different stabilities. Admittedly, the crystalline phase with the lowest vapour pressure is, as a rule, the most stable phase under the selected conditions. Vapour pressures and phase transition data are therefore essential for a topology of the pressure-temperature phase diagrams of APIs [4]. The reliable thermodynamic properties of APIs are essential both for the development of theoretical models and for practical applications.
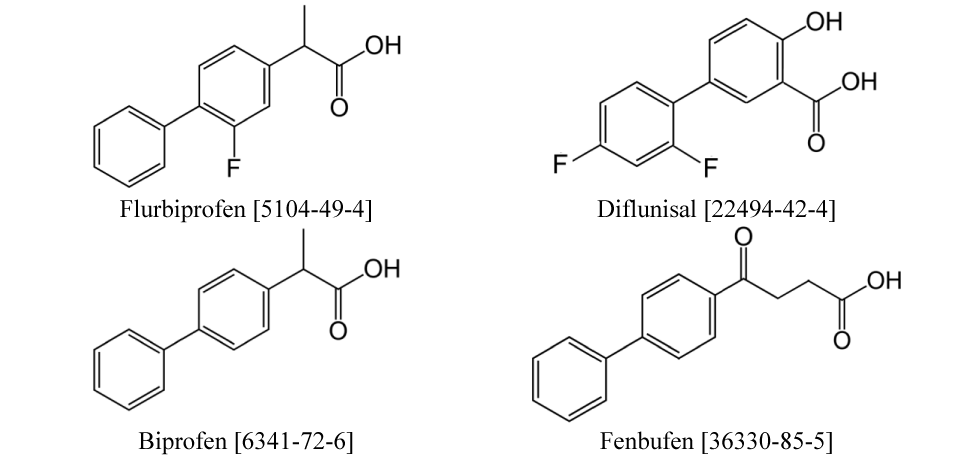
This work is focused on the thermodynamic properties of a class of non-steroidal drugs: Fenbufen, Diflunisal, Biprofen and Flurbiprofen, which are used as anti-inflammatory medicines. These drugs are generally used to relieve pain, reduce inflammation and reduce fever. It is important to note that these APIs can have side effects and care should be taken when using them to ensure appropriate dosing to minimise potential risks. The selected APIs belong to the series of biphenyl derivatives (Figure 1).
In focus of this study is the energetics of liquid-gas, solid-gas, and solid-liquid phase transitions of biphenyl derivatives (or “profens”), as given in figure 1. There are only a few works on thermodynamics of the sublimation and vaporisation of these compounds [5,6,9]. The available results are listed in table 1 and are very misleading. Indeed, the sublimation enthalpy of racemic RS-flurbiprofen (298 K) = 110.2 ± 1.2 kJ⋅mol-1 [5] is significantly lower than its enthalpy of vaporization (298 K) = 127.2 ± 5.5 kJ⋅mol-1 obtained from the CGC method [6]. However, this contradicts common sense (as the enthalpy of sublimation is usually higher than the enthalpy of vaporisation). Which result is not correct? There are numerous methods for predicting the vaporisation enthalpies of organic compounds, e.g., the Advanced Chemistry Development Software (ACD) [7], which is widely used by chemists as it is integrated into the Scifinder online service. However, the enthalpies of vaporisation calculated using this method (Table 1, column 4) make the situation even worse. For example, the vaporisation enthalpy of RS-flurbiprofen (298 K) = 65.8 kJ⋅mol-1 [7] is about twice as small as the enthalpy of vaporization (298 K) = 127.2 ± 5.5 kJ⋅mol-1 from CGC method [6]. To clarify this confusion, the enthalpies of vaporisation of RS-flurbiprofen, fenbufen and diflunisal were additionally estimated using the group-additivity method [8]. However, these new results (Table 1, column 5) were systematically about 10 kJ⋅mol-1 higher than the results from the ACD Software [7], but still far from the experimental value (298 K) = 127.2±5.5 kJ⋅mol-1 [6]. Such an unacceptable discrepancy between the available thermodynamic data of profens requires a diagnostics of the currently available experimental data. The experimental studies of the drugs shown in figure 1 are not trivial, as they belong to the class of extremely low-volatility molecules. Actually, the experimental thermodynamic data for such compounds are either not available or are available as a single experimental result (Table 1). In our experience, these single experimental enthalpies (for each drug) can easily be subject to a systematic error. As has been shown, the empirical methods cannot provide adequate validation of vaporisation and sublimation thermodynamics. To gain at least some insight into the possible reasons for the disarray in the thermodynamics of profens, we decided to carry out complementary sublimation enthalpy measurements on RS-flurbiprofen using the Knudsen effusion method, which is the most suitable conventional method for working with very low volatility compounds [10]. The aim of this work was to reconcile the available phase-change thermodynamics of profens given in figure 1. To do this, a diagnostic method must be developed to validate the thermodynamic data and to evaluate reliable data for safe, effective and reproducible drug delivery.
The idea of this study is to derive the standard molar sublimation enthalpy, (298 K), of profens in independent way using the basic thermodynamic equation.
where (298 K) is the standard molar enthalpy of fusion and (298 K) is the standard molar enthalpy of vaporisation (note that all values in Eq. (1) must be adjusted to the reference temperature T = 298 K).
The fusion enthalpies of pharmaceuticals are nowadays conveniently measured using Differential Scanning Calorimetry (DSC). It should be noted that the DSC method does not always provide reliable results due to insufficient purity of the samples or due to possible decomposition of the samples at elevated temperatures near the melting point. To evaluate the consistency of the fusion enthalpies of profesn (Figure 1), we tested the Walden rule of thermochemistry [11,12].
The measurements of the enthalpies of vaporisation, (T), as the second contribution in Eq. (1), are even more difficult than the sublimation enthalpies, as heating the sample above the typically high melting points (e.g. up to 485 K for diflunisal) could lead to possible decarboxylation. Nevertheless, the contribution (298 K), is very useful and informative for understanding the structure-property relationships. Admittedly, the sublimation enthalpy is not additive. The reason for this is that the non-additive contribution of the fusion enthalpy is contained in the sublimation enthalpy. In contrast, the vaporisation enthalpy obeys the additive rules and is suitable for prediction by different types of Group-Additivity (GA) methods. A modified group additivity method was further developed in this work to obtain a reliable level of vaporisation enthalpies (298 K) for biphenyl derivatives related to drug structures.
This method [13] is based on the selection of a "centrepiece" molecule that comes as close as possible to the main structural motif of the drug structures shown in Figure 1. The experimental sublimation enthalpy, (298 K), and the enthalpy of fusion, (298 K), for RS-Flurbiprofen were measured in this work to validate the "centrepiece" method according to Eq. (1). The diagnostic method based on the "centrepiece" method and Eq. (1) was further propagated to reconcile the available phase-transitions data for Fenbufen, Diflunisal and Biprofen (Figure 1).
Experimental Methods and Materials
The sample of RS-Flurbiprofen (CAS 5104-49-4) from Supelco (certified reference material, 99.9 %) was used in this work without additional purification.
The fusion enthalpy and melting temperature for RS-(±)-flurbiprofen were measured by DSC. The experimental details can be found in the Electronic Supporting Information (ESI).
The Knudsen-Effusion (KE) method [10] was used to measure the vapour pressures (p) for RS-Flurbiprofen. This method relies on the principles of gas flow through a small orifice under low pressure conditions. In the KE device, a sample is placed in a container with an opening and vacuumed. At a constant temperature (T), individual gas molecules escape through the orifice, and the rate of effusion is measured. By measuring the mass loss from the container and applying the Knudsen equation, which relates the effusion rate to the thermodynamic properties of the substance, the sublimation enthalpy is derived from the p - T dependence. The essential details can be found in the ESI.
Results and Discussion
Thermal behaviour and fusion enthalpies
Numerous studies on the thermal behavior and enthalpies of fusion of RS-flurbiprofen, fenbufen, diflunisal and biprofen have been collected in the literature (Table 2).
A total of nine references were collected for RS-flurbiprofen in table 2. Our new result (387.1 K) = 28.6 ± 0.4 kJ⋅mol-1 is consistent with the available values within the combined experimental uncertainties (Table 2). The average value (387.5 K) = 27.8 ± 0.6 kJ⋅mol-1 (Table 2) can be recommended for further thermochemical calculations. For diflunisal, all four fusion enthalpies found in the literature agree very well, resulting in the average value (484.6 K) = 35.6 ± 0.6 kJ⋅mol-1 (Table 2). For fenbufen, with the exception of one outlier (46.2 ± 2.1 kJ⋅mol-1 [26]), the remaining three consistent fusion enthalpy values were averaged (484.6 K) = 35.6 ± 0.6 kJ⋅mol-1 (Table 2) and can be recommended for further thermochemical calculation. No data on fusion enthalpy were found in the literature for biprofen. In our previous work, however, we showed that the required (Tfus)-values can be assessed according to Walden's rule [11,12], expressed by the following equation:
| Table 2: Summary of the melting temperatures, Tfus, and fusion enthalpies, , of profens (in kJ×mol-1)a. | |||||
| Compounds | Tfus/ | Ref. | WC b | c | |
| CAS | K | at Tfus | J·K-1·mol-1 | at 298 K | |
| RS-flurbiprofen | 386.0 ± 0.2 | 26.4 ± 1.6 | Lacoulonche F, et al. [14] | ||
| 5104-49-4 | 387.7 ± 1.0 | 27.9 ± 0.5 | Henck J, et al. [15] | ||
| 386.7 ± 0.4 | 26.4 ± 2.0 | Perlovich GL, et al. [16] | |||
| 387.9 ± 1.0 | 28.0 ± 1.0 | Vippagunta SR, et al. [17] | |||
| 388.2 ± 2.0 | 29.1 ± 4.0 | Grzesiak AL, et al. [18] | |||
| 387.0 ± 0.8 | 27.8 ± 4.6 | Gashi R, et al. [19] | |||
| 388.0 ± 1.0 | 27.4 ± 2.0 | Baird JA, et al. [20] | |||
| 387.1 ± 0.2 | 28.2 ± 0.6 | Umnahanant P, et al. [6] | |||
| 388.8 ± 0.5 | 26.3 ± 0.5 | Tian B, et al. [21] | |||
| 387.1 ± 0.4 | 28.6 ± 0.4 | this work | |||
| 387.5 ± 0.5d | 27.8 ± 0.6 d | 71.7 | 21.7 ± 1.9 | ||
| S-flurbiprofen | 380.6 ± 0.8 | 23.3 ± 0.6 | Umnahanant P, et al. [6] | 61.2 | 17.8 ± 1.8 |
| 51543-39-6 | |||||
| Diflunisal | 483.0 ± 1.0 | 35.0 ± 1.0 | Martínez-Ohárriz MC, et al. [22] | ||
| 22494-42-4 | 486.0 ± 1.0 | 35.9 ± 1.0 | Perlovich Gl, et al. [23] | ||
| 484.8 ± 0.5 | 36.0 ± 1.0 | Surov AO, et al. [24] | |||
| 485.0 ± 0.5 | 35.6 ± 2.0 | Évora AOL, et al. [25] | |||
| 484.6 ± 0.3d | 35.6 ± 0.6 d | 73.5 | 21.7 ± 4.2 | ||
| Fenbufen | 459.3 ± 1.0 | (46.2 ± 2.1) | Wassvik CM, et al.[26] | ||
| 36330-85-5 | 462.9 ± 1.0 | 41.1 ± 1.0 | Kurkov SV, et al. [5] | ||
| 458.2 ± 1.0 | 42.4 ± 3.9 | Gashi Z, et al. [19] | |||
| 457.9 ± 1.0 | 41.5 ± 1.0 | Cong Y, et al. [27] | |||
| 459.7 ± 0.6d | 41.3 ± 0.7 d | 89.8 | 29.3 ± 3.7 | ||
| Biprofen | 431.0 ± 1.0 | 33.7 ± 1.0 e | 24.9 ± 2.8 | ||
| 6341-72-6 | |||||
| average WC | 78.3 ± 5.8 d | ||||
| aUncertainties in this table are presented as expanded uncertainties (0.95 level of confidence with k = 2). bThe Walden Constant (WC) was estimated for each profen using Eq. (2) from results given in columns 3 and 2 of this table. More details are given in ESI. cThe experimental fusion enthalpies measured at Tfus were adjusted to the T = 298 K as explained in ESI. Uncertainties of (298 K) were calculated with 30 % of the total adjustment [28]. dThe weighted average (the experimental uncertainties were taken as the weighing factor) of (Tfus) was estimated (the value given in brackets was excluded) for each profen. Values given in bold were recommended for thermochemical calculations. eEstimated by multiplying WC = (78.3 ± 5.8 J·K-1·mol-1) by Tfus (see text). | |||||
where WC is the Walden´s Constant (Table 2, column 5) and the required fusion parameters (Tfus) and fusion temperature for each profen are collected in table 2. As can be seen from table 2, the WC-values for optically inactive RS-flurbiprofen, diflunisal, and fenbufen were in reasonable agreement and the average value WC = 78.3 ± 5.8 J·K-1·mol-1 was calculated. The latter value can now be multiplied by the Tfus of biprofen to obtain the estimate (431.0 K) = 33.7 ± 1.0 kJ⋅mol-1 (Table 2) required in this work for this compound. Furthermore, the WC value obtained from the profens studied in this work is expected to be valid for evaluating the enthalpies of fusion for other members of the profens family.
The enthalpies of fusion involved in the calculations according to Eq. (1) refer to T = 298 K. The fusion enthalpies of profens referenced to Tfus were adjusted to the reference temperature T = 298 K with help of equation [29]:
where = (g) – (cr) is the difference between the heat capacities of the gaseous (g) and the crystal phase (cr), respectively; and = (g) – (liq) is the difference between the heat capacities of the gaseous (g) and the liquid phase (liq), respectively. The –value and -value used in Eq. (3) are given in table S1. With this adjustment, the fusion enthalpies, (298 K) of profens were estimated (Table 2, column 6) and will be used for estimations according to Eq. (1).
Vapour pressures and sublimation enthalpy of RS-flurbiprofen
The p - T dependence for RS-flurbiprofen measured by the KE method in this work were fitted by the equation [30]:
where R = 8.314462 J.K-1.mol-1 is the ideal gas constant, the reference pressure a and b are adjustable parameters, the arbitrary temperature T0 applied in Eq. (4) was chosen to be T0 = 298 K. The heat capacity differences, , were taken from table S1. The experimental vapour pressures and adjustable parameters of Eq. (4) for RS-flurbiprofen are listed in table S2.
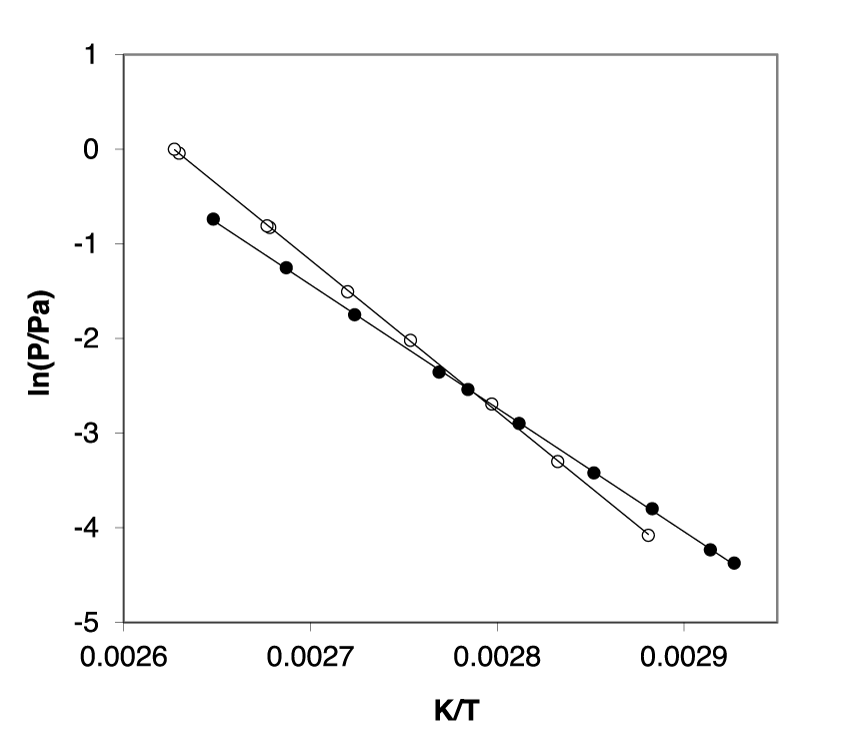
Our new vapour pressures for RS-flurbiprofen, measured using the KE method, are compared in Figure 2 with the values previously measured using transpiration [5].
As shown in figure 2, the trend of temperature dependence of sublimation vapour pressure for flurbiprofen reported by Kurkov and Perlovich [5] is inconsistent with the trend of vapour pressures measured for this compound in our laboratory using the KE method. Furthermore, the differences are rather confusing since, in our experience, at lower temperatures some degree of under-saturation by the transpiration method is expected due to the relatively high flow rate required to obtain a measurable amount of the transported material. However, this is not the case because the vapour pressures measured by transpiration at low temperatures are significantly higher compared to those of the KE method. In contrast, there are no problems with saturation of the gas flow in the transpiration experiment at high temperatures, but the vapour pressures are significantly lower with the transpiration method than with the KE method (Figure 2). Which trend is correct could become clear in the following section.
Experimental standard molar enthalpies of sublimation from vapour pressure measurements
The sublimation enthalpies of RS-flurbiprofen at temperatures T were obtained from the p - T dependence, approximated by Eq. (4) using the equation:
where b is one of the adjustable parameters of Eq. (4). The sublimation enthalpy (298 K) = 136.1 ± 1.2 kJ⋅mol-1 (Table S2) of the RS-flurbiprofen was calculated with help of Eqs. (4) and (5) with the -values from table S1. Eqs. (4) and (5) were also used to approximate the vapour pressures of RS-flurbiprofen, diflunisal, and fenbufen available in the literature and derived the sublimation enthalpies, (298 K) in the same way as our result with the heat capacity differences, , from table S1. The uniformly treated results of the sublimation enthalpies, (298 K), of RS-flurbiprofen, diflunisal, and fenbufen are summarized in table 3 for comparison.
| Table 3: Summary of the sublimation enthalpies, , of RS-flurbiprofen, diflunisal, and fenbufen (in kJ×mol-1). | |||||
| Compounds | Methoda | T-range/ K | Tav | 298 Kb | Ref. |
| RS-flurbiprofen | T | 341.7-377.7 | 108.4 ± 1.1 | 111.0 ± 1.2 | [9] |
| KE | 347.1-380.6 | 133.1 ± 1.0 | 136.1 ± 1.2 | this work | |
| diflunisal | T | 349.2-410.2 | 119.3 ± 1.2 | 122.7 ± 1.4 | [9] |
| fenbufen | T | 378.7-420.7 | 152.0 ± 1.7 | 156.9 ± 2.0 | [5] |
| aMethods: T = transpiration method; KE = Knudsen-Effusion method. bUncertainties of the sublimation enthalpies U() are the expanded uncertainties (0.95 level of confidence). Value given in bold was recommended for further thermochemical calculations. | |||||
As shown in table 3, the sublimation enthalpies of RS-flurbiprofen measured by the KE and transpiration methods differ by 25 kJ⋅mol-1, and we need other independent methods to confirm one or the other value, as shown in the following section.
Vaporisation enthalpies by structure-property correlations
“Centerpiece” approach: Let us apply the basic thermodynamic Eq. (1) to analyse the vaporisation enthalpies of RS-flurbiprofen based on the two experimental results on the enthalpies of sublimation. If we use the transpiration result (298 K) = 111.0±1.2 kJ⋅mol-1 (Table 3) and the enthalpy of fusion (298 K) = 21.7 ± 1.9 kJ⋅mol-1 (Table 2), the enthalpy of vaporisation is:
(298 K) = (298 K) - (298 K) = 111.0 - 21.7 = 89.3 ± 2.3 kJ⋅mol-1 (6).
If we use the KE result (298 K) = 136.1 ± 1.2 kJ⋅mol-1 (Table 3) and the enthalpy of fusion (298 K) = 21.7 ± 1.9 kJ⋅mol-1 (Table 2), the enthalpy of vaporisation is:
(298 K) = 136.1 - 21.7 = 114.4 ± 2.3 kJ⋅mol-1 (7).
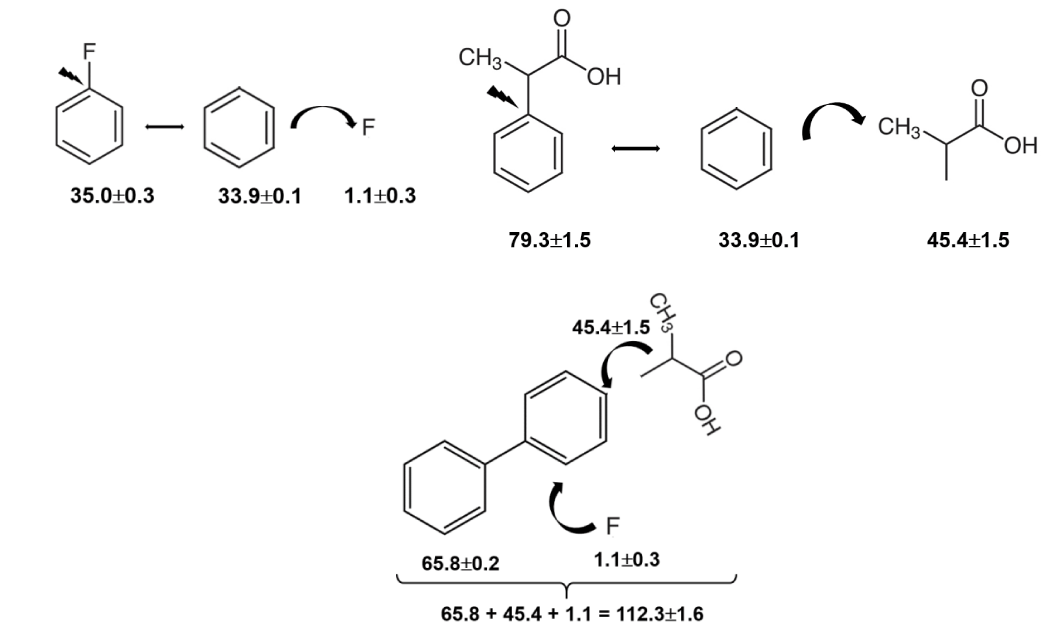
Which result is the more realistic? The structure-property relations are able to help in this unclear situation. The advantage of these relationships is that they are based on a network of reliable thermodynamic data already available in the literature for similarly shaped molecules, so if the new data fit into the existing network, they can be considered reliable. Fig. 1 shows that the common structural motif of all molecules is the biphenyl moiety. The enthalpy of vaporisation of biphenyl is well-known (298 K) = 65.8 ± 0.2 kJ⋅mol-1 (Table S3). For RS-flurbiprofen, two specific substituents (–CH(CH3)COOH and fluorine) are attached to the biphenyl-moiety. The enthalpic contributions for these two substituents can be estimated from the enthalpies of vaporisation of benzene, fluorobenzene, and 2-phenylpropionic acid (Table S3). The estimation algorithm is shown in figure 3.
To build the framework of RS-flurbiprofen, the contribution H→F = 1.1 ± 0.3 kJ·mol-1 and the contribution (H→CH(CH3)COOH) = 45.4 ± 1.5 kJ·mol-1 (Figure 3) is added to the enthalpy of vaporisation of biphenyl (298 K) = 65.8 ± 0.2 kJ⋅mol-1 (table S3). The calculated resulting value (298 K) = 112.3 ± 1.6 kJ·mol-1 can be regarded as a reliable estimate for the enthalpy of vaporisation of RS-flurbiprofen, as no significant interactions of substituents on the biphenyl rings are to be expected. This estimate agrees very well with the result derived according to Eq. (7) from our KE-measurements and not with the result derived according to Eq. (6) from the transpiration method. Such agreement supports the enthalpy of sublimation (298 K) = 136.1 ± 1.2 kJ⋅mol-1 in table 3 and allows us to indicate the transpiration result of Perlovich GL, et al. [9] as questionable.
The estimation algorithm illustrated in figure 3 shows the general idea behind the "centerpiece" (CP) approach (see ESI for details). This method was developed specifically for estimating the energetics of relatively large molecules [13]. The conventional GA methods are generally based on the splitting of the reliable experimental enthalpies of molecules into the smallest possible groups with well-defined numerical contributions. In contrast, the general idea of the CP method is to search for a potentially large "centerpiece" molecule (with the reliable experimental enthalpy) that mimics the structure of the common motif of the desired molecules as closely as possible. The required substituents are then attached to the "centerpiece" molecule, creating the framework of the desired molecule. For the molecules shown in figure 1, the biphenyl serves as the perfect "centerpiece" for predicting the enthalpies of vaporisation of profens. The results for biprofen and fenbufen are shown in figure S2. The estimation algorithm for diflunisal is more complex, as the additional interaction of OH and COOH in the benzene ring must also be included in the calculation (Figure S(3,4)).
The resulting (298 K)-values for RS-flurbiprofen, biprofen, fenbufen obtained using the “centerpiece” method are compared with results of other structure-property methods in Section 3.5.
Correlation gas-chromatography: With the two consistent vaporisation enthalpies of RS-flurbiprofen (298 K) = 114.4 ± 2.3 kJ⋅mol-1 (derived according to Eq. 7) and (298 K) = 112.3 ± 1.6 kJ⋅mol-1 (derived according to the CP approach), it is interesting to understand why the significantly higher enthalpy of vaporisation (298 K) = 127.5 ± 5.5 kJ⋅mol-1 was obtained from the CGC method [6] (Table 1). In fact, CGC is a robust method for the evaluation of enthalpies of vaporisation based on the selection of suitable network of standards (molecules with the reliable vaporisation enthalpies) [31]. The standards and the target substances are injected simultaneously and their retention times are recorded. From the temperature dependences of the corrected retention times the enthalpy of transfer, (Tm), of the analyte from the column material to the gas phase are derived (with Tm as average temperature of the GC-experiment). The transfer enthalpies, (Tm), of the target molecules are correlated with the known enthalpies of vaporisation, (298 K), of the standards. The required enthalpies of vaporisation of the target molecules are derived from this correlation. The selection of the network of
| Table 1: Compilation of the experimental standard molar enthalpies of sublimation, , of profens available in the literature and the standard molar enthalpies of vaporisation, , calculated by empirical methods (at T = 298 K in kJ×mol-1). | ||||
| Compounds | a | b | c | d |
| RS-flurbiprofen | 110.2 ± 1.2 | 127.5 ± 5.5 | 65.8 | 77.1 |
| Diflunisal | 120.1 ± 1.4 | 67.1 | 85.9 | |
| Fenbufen | 155.0 ± 1.6 | 77.2 | 86.6 | |
| aOriginal experimental results, measured using the transpiration method [5]. bOriginal experimental result from the Correlation Gas-Chromatography (CGC) [6]. cThe results estimated with help Advanced Chemistry Development [7]. dThe results estimated using the group additivity [8]. | ||||
As suitable standards for flurbiprofens a series of short-chained alkyl-benzoic acids, fluoro-benzoic acids, as well as two long-chained alkyl-benzoic acids (4-hexylbenzoic acid and 4-octylbenzoic acid) were selected in Ref. [6]. The required for CGC correlations vaporisation enthalpies of 4-alkyl-benzoic acids were calculated as the difference of the available enthalpies of sublimation and enthalpies of fusion. Unfortunately, these compounds either have solid to solid phase transitions prior melting or form liquid crystals. The enthalpies of fusion of these compounds, which are required to process the data for the CGC, were calculated as the sum of all phase transitions from solid to solid and from solid to liquid [6]. The proper adjustment of such fusion enthalpies to T = 298 K is usually hampered by complications. This uncertainty in the fusion enthalpies is probably the reason for the inconsistency between the CGC result [6] and our evaluated result for RS-flurbiprofen. To recover the experimental (Tm)-values from Ref. [6] we removed all data for the long-chained alkyl-benzoic acids (4-hexylbenzoic acid and 4-octylbenzoic acid) from the correlation of (298 K) with (Tm) and re-evaluated the (298 K) = 114.3 ± 1.5 kJ⋅mol-1 for RS-flurbiprofen from the remaining data set (Table 4, column 4).
It has turned out, that the re-evaluated (298 K) = 114.3 ± 1.5 kJ⋅mol-1 for RS-flurbiprofen is in very good agreement with the result obtained in Section 3.4.1.
Kovats indices: In gas chromatography, the Kovats indices, Jx, are determined experimentally by comparing the retention times of sample compounds to those of a series of standard n-alkanes. Kovats indices are a measure of the relative retention times of compounds in a gas chromatograph that can be used to identify and characterise different chemical compounds. The correlation between experimental enthalpies of vaporisation, (298 K), and Kovats indices is a concept based on the similarity of the liquid-gas transitions of the pure liquid compound and the liquid coating of the chromatography column.
The (298 K)-values and the Kovats indices generally correlate linearly in a series of structurally parent molecules [32]. Table 5 summarises the Kovats indices and enthalpies of vaporization of profens and some related compounds used for correlation in this work.
The (298 K)-values summarised in table 5 show a good correlation with the corresponding Jx-values:
(8).
The high correlation coefficient of Eq. (8) is evidence of the consistency of the data used for the correlation. The "empirical" enthalpies of vaporisation obtained from Eq. (7) and the GC results derived in tables 4, 5 are summarised in table 6 for comparison.
| Table 4: The correlation of (298 K) with (Tm) for benzoic acid derivatives and flurbiprofens (in kJ×mol-1). | ||||||
| (Tm)a | (298 K)expb | (298 K)CGCc | ∆d | (298 K)e | (298 K)CGCf | |
| 4-fluorobenzoic acid | 38.7 | 78.9 ± 1.4 | 77.3 | 1.6 | ||
| 2-fluorobenzoic acid | 40.0 | 79.3 ± 1.1 | 79.0 | 0.3 | ||
| 2-methylbenzoic acid | 41.2 | 79.9 ± 0.8 | 80.6 | -0.7 | ||
| 4-methylbenzoic acid | 42.6 | 81.8 ± 0.8 | 82.4 | -0.6 | ||
| 4-ethylbenzoic acid | 44.8 | 84.2 ± 1.5 | 85.3 | -1.1 | ||
| biphenyl-4-carboxylic acid | 61.5 | 107.6 ± 5.3 | 107.3 | 0.3 | ||
| RS- flurbiprofen | 66.8 | 114.4 ± 2.3g | 114.3 | 0.1 | 21.7 ± 1.9 | 136.0 ± 2.4 |
| (R)-flurbiprofen | 67.0 | 114.5 | 17.8 ± 1.8 | 132.3 ± 2.3 | ||
| aThe transfer enthalpies, (Tm) from Ref. [6]. bThe literature data for standards from table S3. cEstimated according to the correlation (298 K)CGC = 1.317× (Tm) +26.3 with R2 = 0.9964. Expanded uncertainty (0.95 level of confidence, k = 2) of ±1.5 kJ.mol-1. dThe difference between experimental (column 3) and calculated values (column 4). eFrom table 1, last column. fEstimated as sum of columns 4 and 6. gCalculated according to Eq. (7). | ||||||
| Table 5: Correlation of enthalpies of vaporisation, (298 K), of profens and some related compounds with their Kovats indices (in kJ×mol-1). | ||||||
| Compound | Jx a | (298 K)exp b | (298 K)calcc | ∆d | (298 K)e | (298 K)DB-1f |
| Phenacetin | 1656 | 97.2 ± 2.5 [Table S4] | 95.3 | 1.9 | ||
| Diflunisal | 2031 | 111.1 ± 2.5 [Figure S4] | 110.8 | 0.3 | 21.7 ± 4.2 | 132.5 ± 4.7 |
| RS-ibuprofen | 1600 | 91.4 ± 0.9 [Table S3] | 92.9 | -1.5 | ||
| RS-naproxen | 2053 | 110.7 ± 2.8 [Table S3] | 111.7 | -1.0 | ||
| salicylamide | 1409 | 87.3 ± 1.3 [Table S3] | 79.0 | -1.6 | ||
| salicylic acid | 1263 | 77.4 ± 2.3 [Table S4] | 85.5 | 1.8 | ||
| aThe Kovats indices, Jx, on the low-polar DB-1 column from Ref. [33]. bExperimental data from tables S3 and S4. cEstimated using equation (298 K) = 26.7 + 0.0414×Jx with R² = 0.9853. Expanded uncertainty (0.95 level of confidence, k = 2) of ±2.0 kJ.mol-1. dThe difference between experimental (column 3) and calculated values (column 4). eFrom table 2, last column. fEstimated as sum of columns 4 and 6. | ||||||
| Table 6: The vaporisation enthalpies, (298 K), of profens as derived by different methods (in kJ×mol-1)a. | |||||
| Eq.(7) b | CP c | CGC d | DB-1 e | (average)f | |
| RS-flurbiprofen | 114.4±2.3 | 112.3 ± 1.6 | 114.3 ± 1.5 | 113.6 ± 1.0 | |
| R-flurbiprofen | 112.3 ± 1.6 | 114.5 ± 1.5 | 114.6 ± 1.5 | ||
| Fenbufen | 136.0 ± 1.0 | 136.0 ± 1.0 | |||
| Diflunisal | 111.1 ± 2.5 | 110.8 ± 2.0 | 110.9 ± 1.6 | ||
| Biprofen | 111.2 ± 1.5 | 111.2 ± 1.5 | |||
| aUncertainties are presented as expanded uncertainties (0.95 level of confidence with k = 2). bThe vaporisation enthalpy, , estimated according to Eq. (7). cThe vaporisation enthalpy, (CP), estimated using the “centerpiece” (CP) method. dThe vaporisation enthalpy, (CGC), estimated with CGC (Table 4, column 4). eThe vaporisation enthalpy, (DB-1), estimated from th e correlation of (298 K) and the Kovats indices (Table 5, column 4). fWeighted average value. Values given in bold are recommended for further thermochemical calculations. | |||||
As it is obvious from this table, the "empirical" vaporization enthalpies for diflunisal and flurbiprofen determined by various methods were found to be in good agreement. In order to obtain more reliability, the average values for diflunisal and flurbiprofen were computed for thermodynamic calculations.
Diagnostic check of the solid-gas phase transitions of profens
Table 7 presents a comparison of the enthalpies of sublimation of fenbufen, diflunisal, biprofen and flurbiprofen, which were determined using different methods.
It is evident that the enthalpy of sublimation for RS-flurbiprofen measured using the KE method agrees within the given uncertainties with the empirical results obtained using the CGC method and also with the estimate based on the CP approach. Therefore, the weighted mean value, (298 K)average = 135.5 ± 0.9 kJ·mol-1 (Table 7) was evaluated from all three entries in table 7. This result helps to neglect the enthalpy of sublimation, (298 K) = 110.2 ± 1.0 kJ·mol-1, originally reported by Kurkov and Perlovich [5], which is dramatically lower by ≈ 25 kJmol-1 than the consistent results of this study.
| Table 7: The enthalpies of sublimation, (298 K) of profens as derived by different methods (in kJ×mol-1)a. | |||||
| Exp b | CP c | CGC d | DB-1 e | (average)f | |
| RS-flurbiprofen | 136.1 ± 1.2 | 134.0 ± 1.7 | 136.0 ± 2.4 | 135.5 ± 0.9 | |
| R-flurbiprofen | 130.1 ± 1.8 | 132.3 ± 2.3 | 130.9 ± 1.4 | ||
| Diflunisal | (122.7 ± 1.4) | 132.8± 4.9 | - | 132.5 ± 4.7 | 132.6 ± 3.4 |
| Fenbufen | (156.9 ± 2.0) | 165.3 ± 3.8 | - | - | 165.3 ± 3.8 |
| Biprofen | - | 137.1 ± 3.2 | - | - | 137.1 ± 3.2 |
| aUncertainties are presented as expanded uncertainties (0.95 level of confidence with k = 2). bThe sublimation enthalpies, , directly measured with the transpiration method or with KE method (Table 3). cThe sublimation enthalpies, (CP), estimated as the sum of vaporisation enthalpy estimated using the “centerpiece” approach (Table 6, column 3) and (298 K), evaluated in table 2. dThe sublimation enthalpies, (CGC), estimated as the sum of vaporisation enthalpy derived from correlation gas-chromatography method (Table 6, column 4) and (298 K), evaluated in table 2. eThe sublimation enthalpies, (DB-1), estimated as the sum of vaporisation enthalpy derived according to Eq. (8) (Table 6, column 5) and (298 K), evaluated in table 2. fWeighted average values. Values in parentheses were excluded from averaging. Values given in bold are recommended for further thermochemical calculations. | |||||
For diflunisal, the empirical enthalpies of sublimation obtained using the GC method and the CP approach agree even better than the attributed uncertainties, but differ by 13 kJ·mol-1 from the previous experimental result reported by Kurkov and Perlovich [5]. Therefore, the weighted mean value, (298 K)average = 132.6 ± 3.4 kJ·mol-1 (Table 7), was evaluated from both empirical results only and recommended for thermochemical calculations instead of the experimental one.
For fenbufen, the empirical enthalpy of sublimation determined using CP approach differs by 10 kJ⋅mol-1 from the previous experimental result of Kurkov and Perlovich [5]. Having established the validity of the CP approach for reliable prediction of profens vaporisation thermodynamics, the value, (298 K) = 165.3 ± 3.8 kJ·mol-1 (Table 7), was recommended for thermochemical calculations instead of the available experimental value given in table 3.
Finally, the diagnostic check of the phase transitions energetics of active pharmaceutical ingredients carried out in this work has revealed the significant inconsistencies in the available experimental results. The differences from 10 to 35 kJ·mol-1 are truly dramatic, as they significantly exceed the acceptable level of uncertainty in thermochemistry. Furthermore, such “sick” experimental data leads to the erroneous development of thermodynamic models commonly used to predict lattice energies and solubilities of drugs. This study clearly demonstrated that we should be concerned about the quality of thermodynamic data on APIs. The diagnostics of energetics of phase transitions in pharmaceutics developed in this work could be considered as a useful quick "health check" of new and old experimental data. Indeed, in a first step, the “centerpiece” approach provides a practical "pencil-and-paper" tool to assess the expected level of the liquid-gas phase transition enthalpy. As a rule, the experimental solid-liquid phase transition enthalpies are readily available in the literature as they are essential for the purity attestation of the synthetic drugs. If not, the Walden Rule can be applied to obtain a reasonable estimate for the second step. Summing the enthalpy results from steps one and two gives the “theoretical” sublimation enthalpy of the API. Comparing the theoretical and available (298 K)-values answers the question of “sick” or “healthy” for the compound of interest. Furthermore, the approach outlined in this paper can be used for the reliable “ad hoc” prediction of vaporisation, fusion, and sublimation enthalpies of active pharmaceutical ingredients.
Conclusion
The consistent thermodynamic data are crucial for selecting the most suitable solvent for purification of active pharmaceutical ingredients during drug development. The thermochemical properties of RS-Flurbiprofen, Diflunisal, Fenbufen and Biprofen available in the literature were collected, combined with our own complementary experimental results and evaluated. The vapour pressures temperature dependence of RS-Flurbiprofen was measured using the Knudsen effusion method, and the enthalpy of sublimation was obtained from this measurement. The enthalpy of fusion of RS-Flurbiprofen was measured using DSC. In this work, a reliable diagnostic approach was outlined to evaluate the quality of the available experimental thermodynamic data of drugs. The result of this work made it possible to analyse and reconcile the available vapour pressures and the energetics of the solid to gas, solid to liquid, and liquid to gas phase transitions of active pharmaceutical ingredients bearing the common biphenyl motif. The data previously available in the literature for RS-flurbiprofen, diflunisal and fenbufen were analysed using the complementary experimental measurements and diagnosed as “thick”. For RS-flurbiprofen, diflunisal, fenbufen and biprofen, the consistent set of thermodynamic data was evaluated and recommended for the lattice energies and solubility calculations. The concept proposed in this work can be extended to the diagnosis of "sick" or "healthy" experimental thermodynamic data for pharmaceuticals with a structure other than profens.
Acknowledgment
SPV gratefully acknowledges the financial support of the German Research Foundation in the frame of SPP 1807 “Control of London Dispersion Interactions in Molecular Chemistry”, grant VE 265-9/2. The work was supported by the Ministry of Science and Higher Education of the Russian Federation (theme No. FSSE-2024-0021) as part of the state task of the Samara State Technical University (creation of new youth laboratories). The work is carried out in accordance with the Strategic Academic Leadership Program "Priority 2030" of the Kazan Federal University of the Government of the Russian Federation. We acknowledge the contribution of Stanislav O. Kondratev in the Knudsen-effusion study. KVZ is grateful to the Ministry of Science and Higher Education of the Russian Federation for the financial support (project No.121031700314-5).
All research data used for the preparation of the manuscript are available in the main text and in the Electronic Supplementary Information.
References
- Malak W, Haslam AJ, Jackson G, Galindo A. Phase behaviour and pH-solubility profile prediction of aqueous buffered solutions of ibuprofen and ketoprofen, Fluid Phase Equil. 2022;560:113504. doi: 10.1016/j.fluid.2022.113504.
- Dohrn S, Luebbert C, Lehmkemper K, Kyeremateng SO, Degenhardt M, Sadowski G. Solvent mixtures in pharmaceutical development: Maximizing the API solubility and avoiding phase separation, Fluid Phase Equil. 548 (2021) 113200. doi: 10.1016/j.fluid.2021.113200.
- Li K, Gbabode G, Barrio M, Tamarit JL, Vergé-Depré M, Robert B, Rietveld IB. The phase relationship between the pyrazinamide polymorphs α and γ. Int J Pharm. 2020 Apr 30;580:119230. doi: 10.1016/j.ijpharm.2020.119230. Epub 2020 Mar 19. PMID: 32199962.
- Bauer M, Lacoulonche F, Céolin R, Barrio M, Khichane I, Robert B, Tamarit JL, Rietveld IB. On the dimorphism of prednisolone: The topological pressure-temperature phase diagram involving forms I and II. Int J Pharm. 2022 Aug 25;624:122047. doi: 10.1016/j.ijpharm.2022.122047. Epub 2022 Jul 25. PMID: 35902055.
- Kurkov SV, Perlovich GL. Thermodynamic studies of Fenbufen, Diflunisal, and Flurbiprofen: sublimation, solution and solvation of biphenyl substituted drugs. Int J Pharm. 2008 Jun 5;357(1-2):100-7. doi: 10.1016/j.ijpharm.2008.01.059. Epub 2008 Feb 9. PMID: 18343061.
- Umnahanant P, Hasty D, Chickos J. An examination of the thermodynamics of fusion, vaporization, and sublimation of (R,S)- and (R)-flurbiprofen by correlation gas chromatography. J Pharm Sci. 2012 Jun;101(6):2045-54. doi: 10.1002/jps.23094. Epub 2012 Mar 12. PMID: 22411450.
- Advanced Chemistry Development (ACD/Labs) Software V11.02 (© 1994-2024 ACD/Labs).
- Joback KG, Reid RC. Estimation of pure-component properties from group-contributions. Chem Eng Commun. 1987;57:233-243. doi: 10.1080/00986448708960487.
- Perlovich GL, Kurkov SV, Bauer-Brandl A. Thermodynamics of solutions II. Flurbiprofen and diflunisal as models for studying solvation of drug substances. Eur J Pharm Sci. 2003;19:423-432. doi: 10.1016/S0928-0987(03)00145-3.
- Verevkin SP, Zaitsau DH, Schick C, Heym F. Development of direct and indirect methods for the determination of vaporization enthalpies of extremely low-volatile compounds. Handbook of Therm Anal & Calorim. 2018;6:1-46. doi: 10.1016/B978-0-444-64062-8.00015-2.
- Walden P. Über die schmelzwärme, spezifische kohäsion und molekulargrösse bei der schmelztemperatur. Z Elektrochem Angew Phys Chem. 1908;14:713-728. doi: 10.1002/bbpc.19080144302.
- Abdelaziz A, Zaitsau DH, Kuratieva N, Verevkin SP, Schick C. Melting of nucleobases. Getting the cutting edge of “Walden’s Rule”. Phys Chem Chem Phys. 2019;21:12787-12797. doi: 10.1039/c9cp00716d.
- Zherikova KV, Verevkin SP. Error or exemption to the rule? Development of a diagnostic check for thermochemistry of metal- Organic compounds. RSC Adv. 2020;10:38158-38173. doi: 10.1039/D0RA06880B.
- Lacoulonche F, Chauvet A, Masse J. An investigation of flurbiprofen polymorphism by thermoanalytical and spectroscopic methods and a study of its interactions with poly-(ethylene glycol) 6000 by differential scanning calorimetry and modelling. Int J Pharm. 1997;153:167-179. doi: 10.1016/S0378-5173(97)00102-6.
- Henck JO, Kuhnert-Brandstätter M. Demonstration of the terms enantiotropy and monotropy in polymorphism research exemplified by flurbiprofen. J Pharm Sci. 1999 Jan;88(1):103-8. doi: 10.1021/js9801945. PMID: 9874709.
- Perlovich GL, Hansen LK, Bauer-Brandl A. Solvates with anomalous low melting points: preparation, structural and thermochemical aspects. J Therm Anal Calorim. 2003;73:715-725.
- Vippagunta SR, Wang Z, Hornung S, Krill SL. Factors affecting the formation of eutectic solid dispersions and their dissolution behavior. J Pharm Sci. 2007 Feb;96(2):294-304. doi: 10.1002/jps.20754. PMID: 17051588.
- Grzesiak AL, Matzger AJ. New form discovery for the analgesics flurbiprofen and sulindac facilitated by polymer-induced heteronucleation. J Pharm Sci. 2007 Nov;96(11):2978-86. doi: 10.1002/jps.20954. PMID: 17567888; PMCID: PMC2581769.
- Gashi Z, Censi R, Malaj L, Gobetto R, Mozzicafreddo M, Angeletti M, Masic A, Di Martino P. Differences in the interaction between aryl propionic acid derivatives and poly(vinylpyrrolidone) K30: A multi-methodological approach. J Pharm Sci. 2009 Nov;98(11):4216-28. doi: 10.1002/jps.21734. PMID: 19645003.
- Baird JA, Van Eerdenbrugh B, Taylor LS. A classification system to assess the crystallization tendency of organic molecules from undercooled melts. J Pharm Sci. 2010 Sep;99(9):3787-806. doi: 10.1002/jps.22197. PMID: 20623696.
- Tian B, Feng Y, Li X, Yang J, Ding Z, Huang X, Yin Q, Xie C, Hao H. Solution thermodynamic properties of flurbiprofen in twelve solvents from 283.15 to 323.15 K. J Mol Liq. 2019;296:111744. doi: 10.1016/j.molliq.2019.111744.
- MMartínez-Ohárriz MC, Martín C, Goñi MM, Rodríguez-Espinosa C, Tros de Ilarduya-Apaolaza MC, Sánchez M. Polymorphism of diflunisal: isolation and solid-state characteristics of a new crystal form. J Pharm Sci. 1994 Feb;83(2):174-7. doi: 10.1002/jps.2600830212. PMID: 8169784.
- Perlovich GL, Hansen LK, Bauer-Brandl A. Interrelation between thermochemical and structural data of polymorphs exemplified by diflunisal. J Pharm Sci. 2002 Apr;91(4):1036-45. doi: 10.1002/jps.10043. PMID: 11948542.
- Surov AO, Voronin AP, Manin AN, Manin NG, Kuzmina LG, Churakov AV, Perlovich GL. Pharmaceutical cocrystals of diflunisal and diclofenac with theophylline. Mol Pharm. 2014 Oct 6;11(10):3707-15. doi: 10.1021/mp5004652. Epub 2014 Sep 11. PMID: 25184906.
- Évora AOL, Castro RAE, Maria TMR, Silva MR, ter Horst JH, Canotilho J, Eusébio MES. Co-Crystals of diflunisal and isomeric pyridinecarboxamides - A thermodynamics and crystal engineering contribution. Cryst Eng Comm. 2016;18:4749-4759. doi: 10.1039/C6CE00380J.
- Wassvik CM, Holmén AG, Bergström CA, Zamora I, Artursson P. Contribution of solid-state properties to the aqueous solubility of drugs. Eur J Pharm Sci. 2006 Nov;29(3-4):294-305. doi: 10.1016/j.ejps.2006.05.013. Epub 2006 Jun 7. PMID: 16949802.
- Cong Y, Du C, Xing K, Bian Y, Li X, Wang M. Investigation on co-solvency, solvent effect, hansen solubility parameter and preferential solvation of fenbufen dissolution and models correlation. J Mol Liq. 2022;348:118415. doi: 10.1016/j.molliq.2021.118415.
- Gobble C, Chickos J, Verevkin SP. Vapor pressures and vaporization enthalpies of a series of dialkyl phthalates by correlation gas chromatography. J Chem Eng Data. 2014;59:1353-1365. doi: 10.1021/je500110d.
- Acree WE, Chickos JS. Phase transition enthalpy measurements of organic and organometallic compounds. Sublimation, vaporization and fusion enthalpies from 1880 to 2015. Part 1. C1 - C10. J Phys Chem Ref Data. 2016;45:33101. doi: 10.1063/1.4948363.
- Kulikov D, Verevkin SP, Heintz A. Determination of vapor pressures and vaporization enthalpies of the aliphatic branched c5 and c6 alcohols. J Chem Eng Data. 2001;46:1593-1600. doi: 10.1021/je010187p.
- Chickos JS, Hosseini S, Hesse DG. Determination of vaporization enthalpies of simple organic molecules by correlations of change in gas chromatographic net retention times. Thermochim Acta. 1995;249:41-62. doi: 10.1016/0040-6031(95)90670-3.
- Verevkin SP. Vapour pressures and enthalpies of vaporization of a series of the linear n-alkyl-benzenes. J Chem Thermodyn. 2006;38:1111-1123. doi: 10.1016/j.jct.2005.11.009.
- Sharp ME. A rapid screening procedure for acidic and neutral drugs in blood by high resolution gas chromatography. J Anal Toxicol. 1987 Jan-Feb;11(1):8-11. doi: 10.1093/jat/11.1.8. PMID: 3821079.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.