Medicine Group. 2024 September 20;5(9):1176-1186. doi: 10.37871/jbres2006.
Exploring the Potential of Xanthatin from Cuban Xanthium strumarium: Isolation, Characterization, and Biological Evaluation
Berenguer A1, Osoria L1, González Suarez N2, Manso A1, Salomón S1, Padrón Yaquis AS1, Annabi B2 and Piloto-Ferrer J1*
2Molecular Oncology Laboratory, Department of Chemistry, University of Quebec in Montreal, Montreal, Quebec, Canada
- Antiproliferative activity
- Cervical cancer
- Characterization
- Colorectal cancer
- Isolation
- Xanthatin
Abstract
Xanthatin possesses anticancer activities through its effect on cancer cell proliferation, apoptosis and metastasis in vitro and in vivo. Here, we described the isolation procedure, characterization and in vitro biological evaluation of the active metabolite xanthatin from Xanthium strumarium grown in Cuba. From the aerial parts of X. strumarium, 995 mg of xanthatin was isolated in the form of prismatic crystals with a yield of 0.07%. The crystals obtained were characterized by HPLC-UV and spectroscopic techniques including UV, IR, and one- and two-dimensional NMR. The signals observed in the spectra obtained confirmed the structure of xanthatin, especially the two-dimensional NMR spectra. The absence of the coupling signal between the H-8 and H-7 protons in the NOESY spectrum allowed us to identify that the fusion of the 7-membered ring and the lactone ring occurs with trans stereochemistry. The isolated xanthatin undergoes a phase transition at 114.30 ± 0.03°C, a value in good agreement with the melting point reported for commercial xanthatin. Biological evaluation showed that xanthatin inhibited the proliferation of HT-29 colorectal cancer cells and HeLa cervical cancer cells, with the greatest inhibitory potential against colorectal cancer. The molecule can also inhibit cell cycle progression by inducing cell cycle arrest at the G2/M phase in HeLa and HT-29 cells, with the greatest effect in HT-29. In conclusion, this study demonstrates promising therapeutic effects of xanthatin from X. strumarium grown in Cuba against colorectal and cervical cancer cell lines, and provides new insights into its potential and effective antitumor properties.
Abbreviations
ATCC: American Type Culture Collection; IX: Isolated Xanthatin; CC: Column Chromatography; DMEM: Dulbecco's Modified Eagle's Medium; DMSO: Dimethylsulfoxide; HPLC: UV-High Performance Liquid Chromatography coupled to UV detector; IC50: Half-maximal Inhibitory Concentration; IR: Infrared Spectrum; MeCN: Acetonitrile; MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide]; NMR: Nuclear Magnetic Resonance; NSCLC: Non-Small Cell Lung Cancer Cells; NOESY: Nuclear Overhauser Effect Spectroscopy; PXT: Paclitaxel; UV: Ultraviolet; XCtrol: Commercial xanthatin; XFC: Xanthium Strumarium Chloroform Extract; TMS: Tetra Methyl Silane
Introduction
One of the priorities of the Cuban National Health System is to carry out research that can lead to new drugs for the treatment of cancer, the leading cause of death worldwide [1,2], or that can improve the quality of life of cancer patients. Accordingly, the growing interest in the biological activities of natural products, specifically those derived from plants, has emerged in light of their structurally diverse components and their skeletons susceptible to synthetic transformations [1]. The search for new phytochemicals for cancer therapy is a worthwhile endeavor. The identification of anticancer plant compounds is typically initiated by collecting a diverse range of samples from various geographical regions or by drawing upon traditional knowledge and practices. In this sense, in a screening of the antitumor activity of plant extracts derived from native Cuban flora, one of the most promising species with antimitotic activity was Xanthium strumarium (Family: Asteraceae) [2] Xanthatin was later shown to be the metabolite responsible for the antimitotic activity [3,4]. X. strumarium has a global distribution and is abundant throughout Eurasia and the Americas. It has historically been used in traditional medicine in Oriental countries. It has also been widely used as analgesics, as antibacterial and anti-inflammatory agents, and have been used for chronic bronchitis, chronic and allergic rhinitis, and to relieve constipation, diarrhea, and vomiting [5].
X. strumarium is known for the production of sesquiterpene lactones termed xanthanolides, which are responsible for most of the biological activities of Xanthium species [6-8]. Structurally, xanthanolides are bicyclic sesquiterpene lactones in which a α-methylene-γ-butyrolactone ring is fused to a carbocycle of 7 carbon atoms. According to the cis- or trans-configuration of the ring fusion, two groups can be distinguished, xanthanolides and 8-epixanthanolides, with xanthatin and 8-epixanthatin being the representatives per excellence of these groups. In addition, most of these compounds also include a side chain of four carbon atoms that can be saturated or unsaturated at the C2 and C3 positions [9]. Xanthatin is one of the main bioactive components of Xanthium extracts, which has been shown to inhibit tumor growth and cell proliferation through proposed mechanisms including inhibition of the mitotic spindle, causing G2/M arrest and disrupting anaphase entry [10,11]. Accordingly, numerous studies demonstrated the effectiveness of xanthatin in suppressing cell proliferation and inducing apoptosis in a wide variety of cancer cell lines including: human gastric carcinoma (MKN-45) [12], non-small cell lung cancer cells (NSCLC, A549 and H1299) [13], triple-negative-derived breast cancer (MDA-MB-231) [14], and colon carcinoma (CT26WT) [11]. Likewise, its antimitotic potential is reported from the interruption of metaphase to anaphase transit leading to apoptotic cell death [4,15].
Previous studies have demonstrated that the xanthatin-enriched fraction derived from the chloroform extract of the aerial parts of X. strumarium is the primary contributor to the antitumour activity observed in these extracts [2-4,16, 17]. However, xanthatin obtained from X. strumarium growing in Cuba had never been isolated and characterized and given the above evidence and the documented invasive nature of this species, which allows it to spread rapidly over large vegetative areas [18], X. strumarium represents a promising candidate for the isolation and evaluation of xanthatin as an antitumour agent. Thus, the main objective of this work was to isolate and characterize xanthatin from the aerial parts of Cuban X. strumarium. In addition, efficacy to inhibit cancer cell proliferation was assessed and compared to a commercially available standard.
Materials and Methods
General experimental procedures
A Spectronic Genesys spectrophotometer (Thermo Scientific, United States Country) was used to obtain Ultraviolet (UV) spectra. A Bruker Tensor-27 Fourier transform infrared spectrometer (Bruker, Germany) was used for scanning IR spectrum with KBr pellets. 1D and 2D Nuclear Magnetic Resonance (NMR) spectra were obtained on Varian VXR-Unity-400 MHz NMR spectrometer (Bruker, Switzerland). Chemical shifts (δ) are expressed in ppm and coupling constants (J) in Hz. The δ values refer to Tetramethylsilane (TMS) as an internal reference. The melting point was determined in an automatic Melting Point M-565, Büchi. HPLC–UV analysis was performed on a KNAUER Azura system with a C18 GL Sciences Inc. column (5 μm, 4 mm × 250 mm, 1 mL/min). The samples were eluted using a solvent system with a gradient (80% H2O/20% MeCN to 40% H2O/60% MeCN) over 40 min. Column Chromatography (CC) was performed on silica gel 60 (70-230 mesh, Qingdao Marine Chemical, Ltd.). PF254 silica gel plates on aluminum support with fluorescent indicator were used, and a 6:4 (v/v) n-hexane/ethyl acetate mixture was used as mobile phase. The plates were developed using a UV lamp at 254 and 366 nm and then sprayed with a 1:70:10 (m/v/v) solution of vanillin/ethanol/sulphuric acid and heated until staining [19]. Cell counting in the different cell phases was carried out on a Becton Dickinson FACSCalibur cytometer. The microplates were read using a SPECTROstar® Omega microplate reader. All reagents and solvents used were of pure analytical quality.
Plant material
The plant Xanthium strumarium was cultivated at the Experimental Station of Medicinal Plants "Dr. Juan Tomás Roig" in Artemisa, Cuba. A specimen of this plant is conserved in the herbarium of the institution with herbarium number ROIG 4594. The leaves and stems, collected in May 2022, were washed with 0.5% sodium hypochlorite, dried in an oven at a temperature of 60°C and grinded in a MANESTY mill with a 1 mm sieve.
Extraction and isolation
Xanthatin extraction and isolation was carried out as described by Romero M, et al. [7] and Ramírez-Erosa I, et al. [19] with modifications. 1.5 kg of dried and ground plant material from the aerial parts of X. strumarium was subjected to preliminary defatting with n-hexane and then extracted with 96% ethanol, in both cases by dynamic ultrasound-assisted maceration and heating to 50°C. A 1:10 ratio of plant material to solvent was used during both extractions. Subsequently, the ethanolic extract redissolved in a 70% hydroalcoholic solution was partitioned by liquid-liquid extraction with chloroform to obtain a xanthatin-enriched extract. The chloroform extract was further fractionated by liquid chromatography on a 60 Å (70-230 mesh) silica gel column with a polarity gradient of n-hexane/ethyl acetate mixtures (4:1 to 3:2). The xanthatin eluted with the solvent mixture in a 3:2 ratio. From the fractions eluted from the chromatographic column it was possible to isolate 995 mg of crude xanthatin. The latter was recrystallized in acetone and 775 mg of prismatic crystals with a faint yellow colour were obtained.
Cervical and colorectal cancer cell cultures
Human cervical carcinoma (HeLa) and human HT-29 colorectal adenocarcinoma cells were obtained from the American Type Culture Collection (ATCC). Cells were grown as monolayers with Dulbecco's modified Eagle's medium (DMEM) containing 10% fetal bovine serum (FBS), 100 U/mL penicillin, and 100 mg/mL streptomycin. All the cells were cultured at 37°C under a humidified 95%-5% (v/v) mixture of air and CO2. All reagents for cell culture were purchased from Sigma®.
Cell viability assay
The MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] cytotoxicity assay was performed according to the protocol described in the literature [20], with some modifications. HT-29 and HeLa cells were seeded in 96-well plates at 1 x 104 cells/mL. After being cultured overnight, cells were exposed to increasing concentrations of isolated and commercial (APExBIO®,) xanthatin, ranging from 1 to 20 µM (200 μL/well, final volume). At the end of this period, the cells were incubated with MTT (5 mg/mL) for 4 hours. IC50 (half-maximal inhibitory concentration) values of the isolated xanthatin were determined from a dose–response curve obtained by using different concentrations (1, 5, 10 and 20 μM). The plates were read in a microplate spectrophotometer at 550 nm. Analyses were made in triplicate for each condition.
Cell cycle analysis
Cell cycle analysis was carried out in accordance with the described by Francisco-Fernandez M, et al. [17]. To determine the concentration of xanthatin that causes cell cycle arrest in the HT-29 and HeLa cell lines, the cells were cultured in 6-well plates (1.6 × 105 cells/mL) and (2 × 105 cells/mL) respectively, until reaching 90% confluence. Cells were then treated for 24 hours with either 0.1% DMSO (controls) or various concentrations of 1-5-10-20 μM of commercial xanthatin or of the isolated xanthatin. Cells were incubated for 24 hours with the treatments in medium containing 10% FBS. After treatment, cells were harvested by mild trypsin digestion, washed with ice-cold PBS, and fixed in ice-cold 70% ethanol in PBS overnight at -20°C. The cells were then centrifuged at 10,000 rpm for 10 minutes and the supernatant was carefully removed. The staining solution containing propidium iodide (PI, 40 μg/mL; Calbiochem, San Diego, CA) and DNase-free RNase (100 μg/mL), was added for at least 30 min at 37°C in the dark before analysis. The proportion of the cell population in each phase of the cell cycle was determined as a function of the DNA content using a flow cytometer. For each measurement, at least 10,000 cells were counted.
Statistical analysis
Statistical analysis of data was performed using ANOVA followed by Dunnet's test. Statistical significance was used to compare the effect of IX with that of vehicle-treated cells. All statistical analyses and graphs were performed using GraphPad Prism software version 5.0b (San Diego, CA). Where indicated, data are presented as mean ± SD of three independent experiments (significance: *p < 0.05, **p < 0.01, ***p < 0.001 versus negative control).
Results and Discussion
Isolation and characterization of xanthatin
Fractionation of the chloroform extract allowed the isolation of 995 mg of xanthatin, representing 0.07% of the mass of the starting plant material. This yield is higher than that previously reported by Zhi X, et al. [21] (0.02%) for the isolation of this compound from 10 kg of X. strumarium, using three macerations at room temperature and a plant mass/solvent ratio of 1:2.5 (kg/L) as the extraction method.
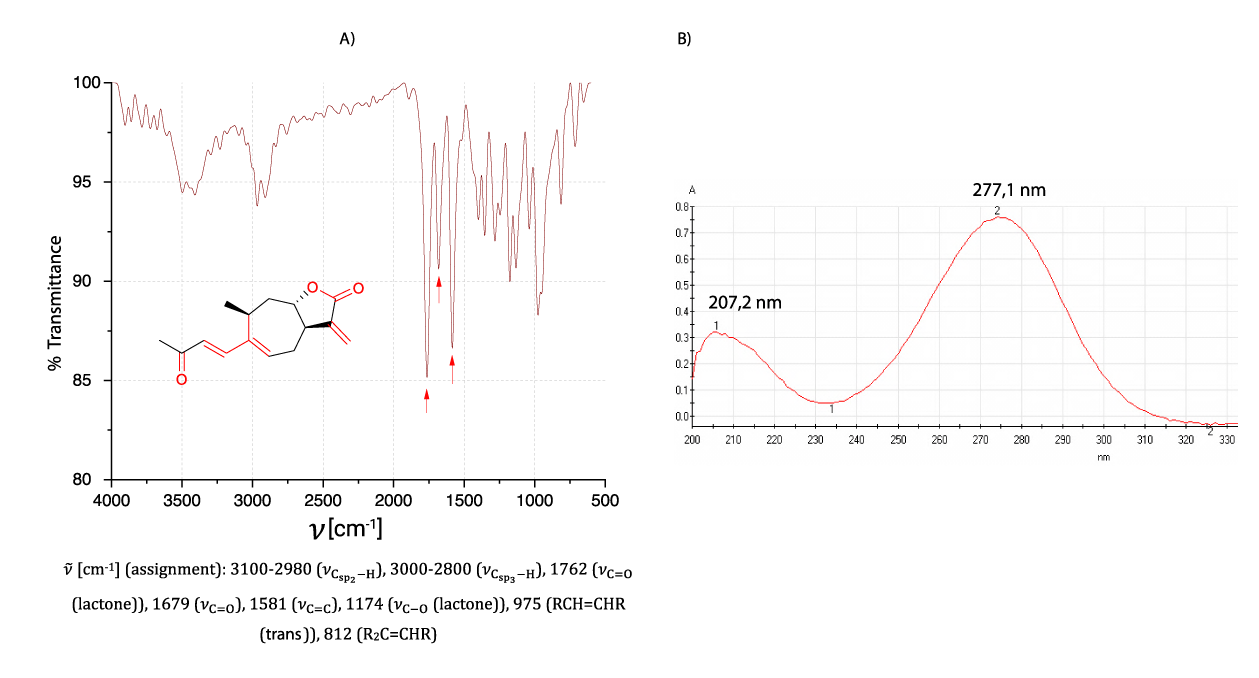
Melting point determination showed that the crystals undergo a phase transition at 114.30 ± 0.03 °C, a value in good agreement with the melting point reported for commercial xanthatin 114.5-115°C [7]. On the other hand, the infrared spectrum (Figure 1A) shows intense signals typical of carbonyl valence vibrations (1762, 1581 cm-1) and double bonds (1679 cm-1), while from the UV spectrum (Figure 1B) recorded for the obtained crystals, absorption maxima were observed at 207.2 and 277.1 nm [7,19]. Both spectra showed characteristic features of the functional groups present in the structure of xanthatin, whose structure was confirmed and assembled by one- and two-dimensional magnetic resonance spectra.
The signals observed in the one-dimensional NMR spectra are shown in table 1, and showed the characteristic signals of the lactone in question, in agreement with those previously reported for this compound [7,19].
| Table 1: 1H-NMR and 13C-NMR assignments for xanthatin in DMSO-d6. | |||
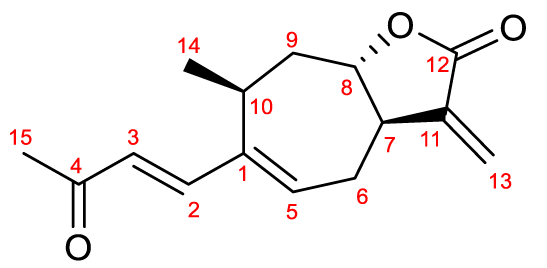 |
Position | δC (ppm) | δH (ppm). multiplicity. (J.Hz) |
| 1 | 143.9 | - | |
| 2 | 148.7 | 7.15. 1H. d (16.1) | |
| 3 | 124.2 | 6.20. 1H. d (16.1) | |
| 4 | 198.0 | - | |
| 5 | 139.5 | 6.42. 1H. dd (9.2; 3.4) | |
| 6 | 26.6 | 6α. 2.85. 1H. ddd (16.7; 9.1; 2.5) | |
| 6β. 2.24. 1H. m | |||
| 7 | 46.6 | 2.59. 1H. m | |
| 8 | 80.9 | 4.42. 1H. ddd. (12.3; 9.8; 2.6) | |
| 9 | 35.9 | 9β 2.24. 1H. m | |
| 6α 1.79. 1H. td. (12.5; 3.9) | |||
| 10 | 28.2 | 3.07. 1H. m | |
| 11 | 139.5 | - | |
| 12 | 169.4 | - | |
| 13 | 118.6 | 13a 5.67. 1H. d (3.1) | |
| 13b 6.03. 1H. d (3.1) | |||
| 14 | 18.5 | 1.09. 3H. d (7.3) | |
| 15 | 27.4 | 2.24. 3H. s | |
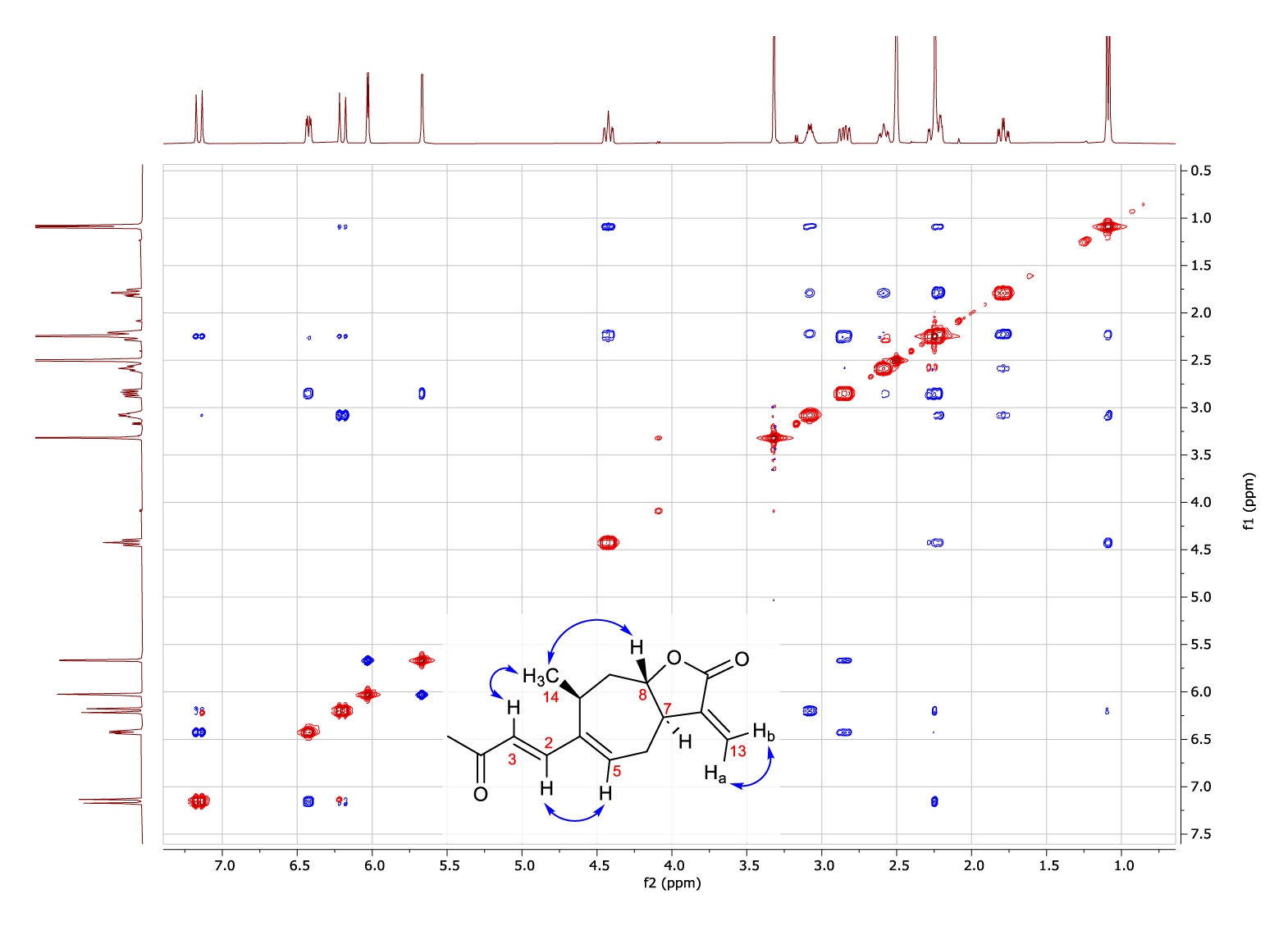
The stereochemistry of the structure was determined from the information of the NEOSY spectrum (Figure 2) whose signal pattern revealed a configuration (E) of the double bond of the side chain, namely the H-3/H-14 (6.22; 1.09 ppm) and H-2/H-5 (7.17; 6.42 ppm) crossover peaks. Based on the isolation of xanthanolides from various species within the Xanthium genus, it has been determined that the methyl group at position 14 consistently exhibits an (S) configuration [9]. Furthermore, the presence of the H-8/H-14 signal due to the dipolar coupling between the protons of the methyl group -CH3 (14) and the H-8 proton indicates that both groups are in close proximity and exhibit the same spatial arrangement. Conversely, the absence of the coupling signal between the H-8 and H-7 protons indicates that the fusion of both rings occurs with trans stereochemistry.
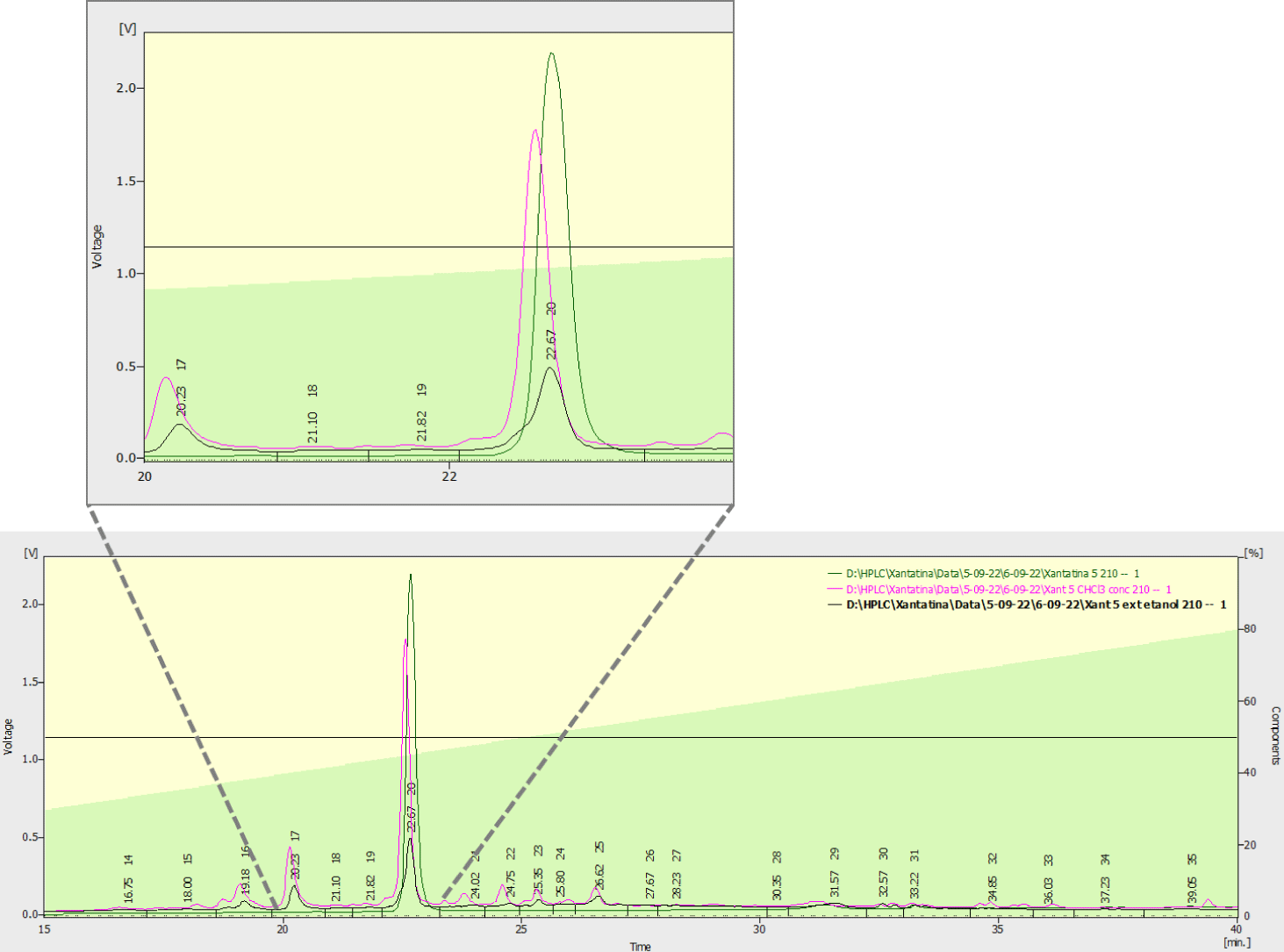
Based on the spectral information obtained, it was possible to confirm that the isolated compound is in fact xanthatin. In addition, chromatographic analysis by HPLC-UV of ethanolic and chloroform extracts and isolated xanthatin (Figure 3), allowed confirming that this lactone is the main metabolite of extracts obtained from plant material of Cuban species of X. strumarium.
Impact of xanthatin on HT-29 and HeLa cancer cells proliferation
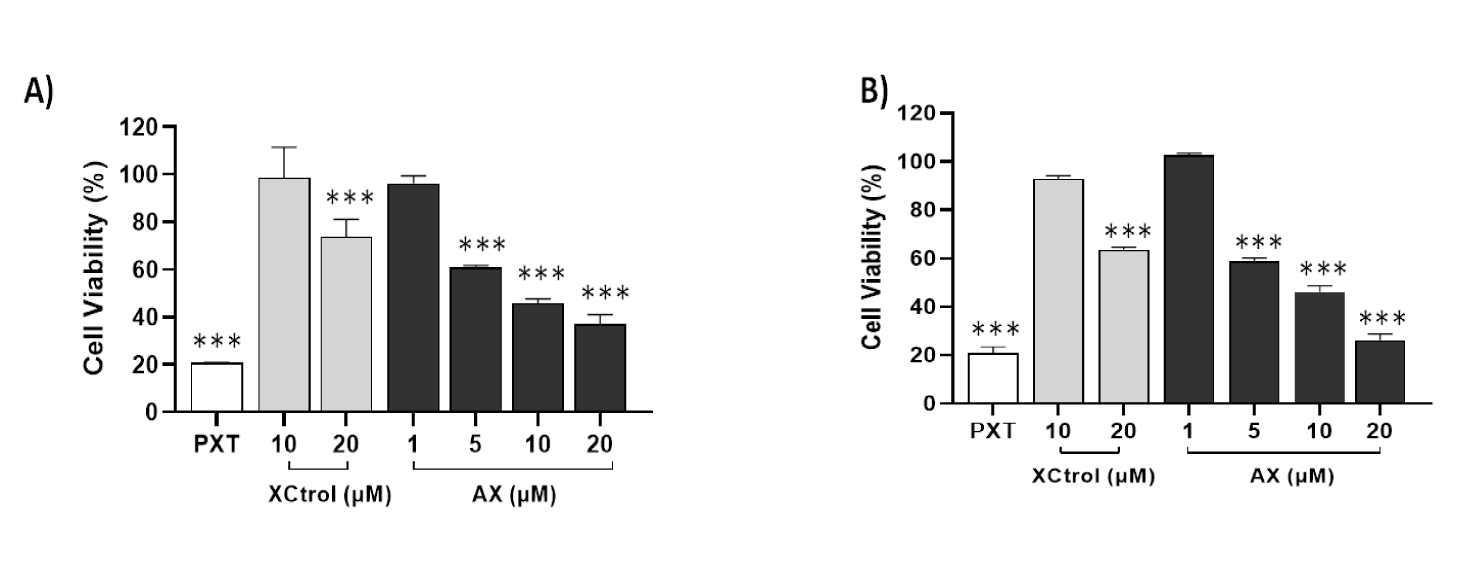
The antiproliferative capacity of xanthatin was first assessed on established human cancer cell models derived from cervical adenocarcinoma (HeLa) and colorectal adenocarcinoma (HT-29). Xanthatin was able to dose-dependently inhibit cell proliferation in both cellular models (Figure 4). When compared to commercial xanthin, the isolated molecule was found to be more active in both tumor cells at the same 10 and 20 µM concentrations (Figure 4A,B), while the highest inhibitory potential was against colorectal cancer cells at 5 µM (Figure 4B). The IC50 values of isolated xanthatin in HeLa and HT-29 cell lines were 8.7 µM and 7.7 µM respectively. This result suggests that xanthatin has the ability to inhibit cancer cell proliferation, with the strongest effect against colorectal cancer cells. In a previous study published by Li L, et al. [22] the effect of xanthatin was evaluated on the energy metabolism of human colorectal cancer cells HT-29 and HCT-116 and their normal progression. The results showed that xanthatin significantly inhibited the migration and invasion of both cancer cell lines, providing important information on its anticancer property. Xie Y, et al. [23] evaluated the inhibition potential of xanthatin on the proliferation of NSCLC and proved that the activity was related to breaking the intracellular redox balance, suggesting that xanthatin is a promising antitumor candidate for the treatment of non-small cell lung cancer.
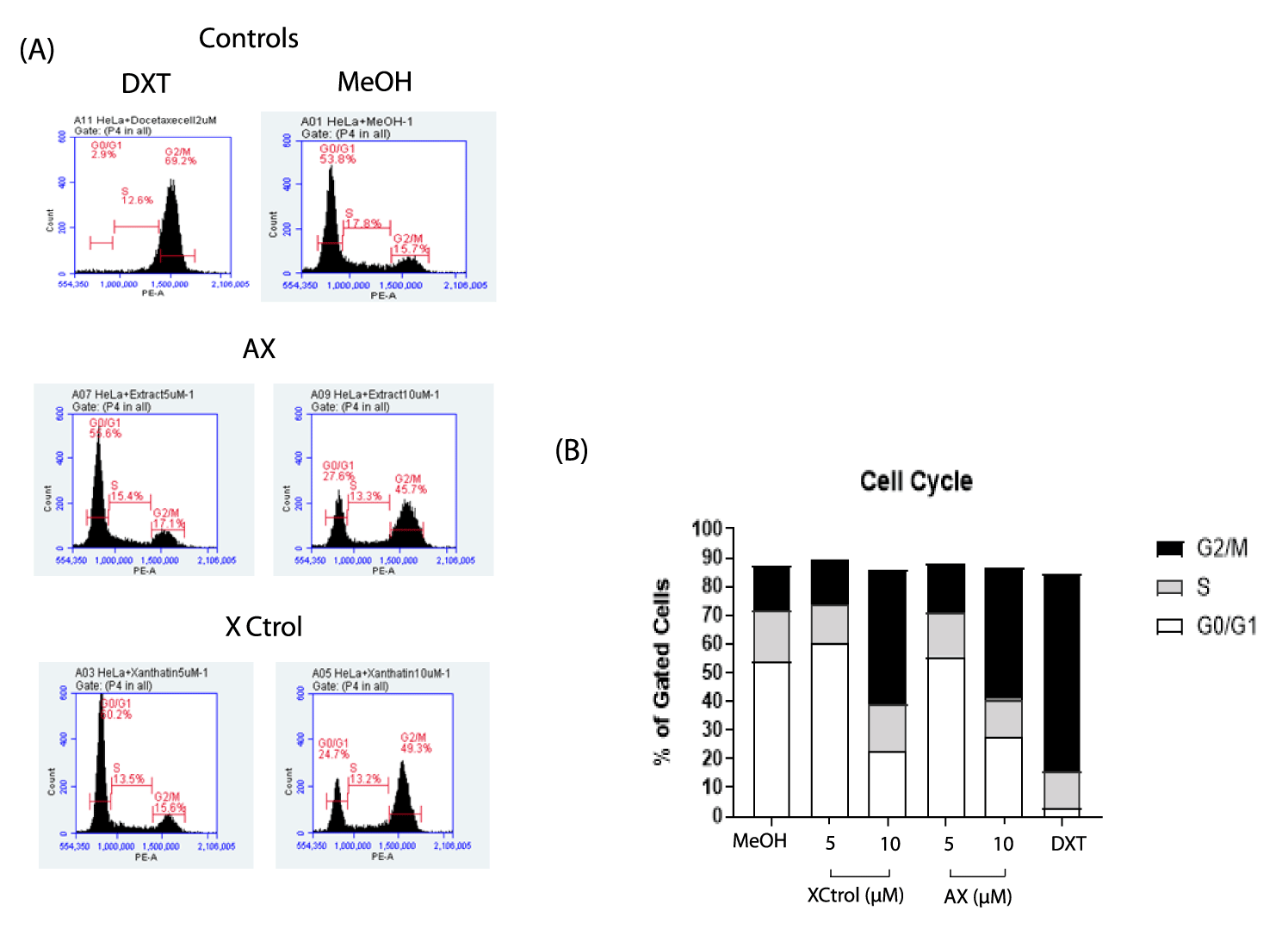
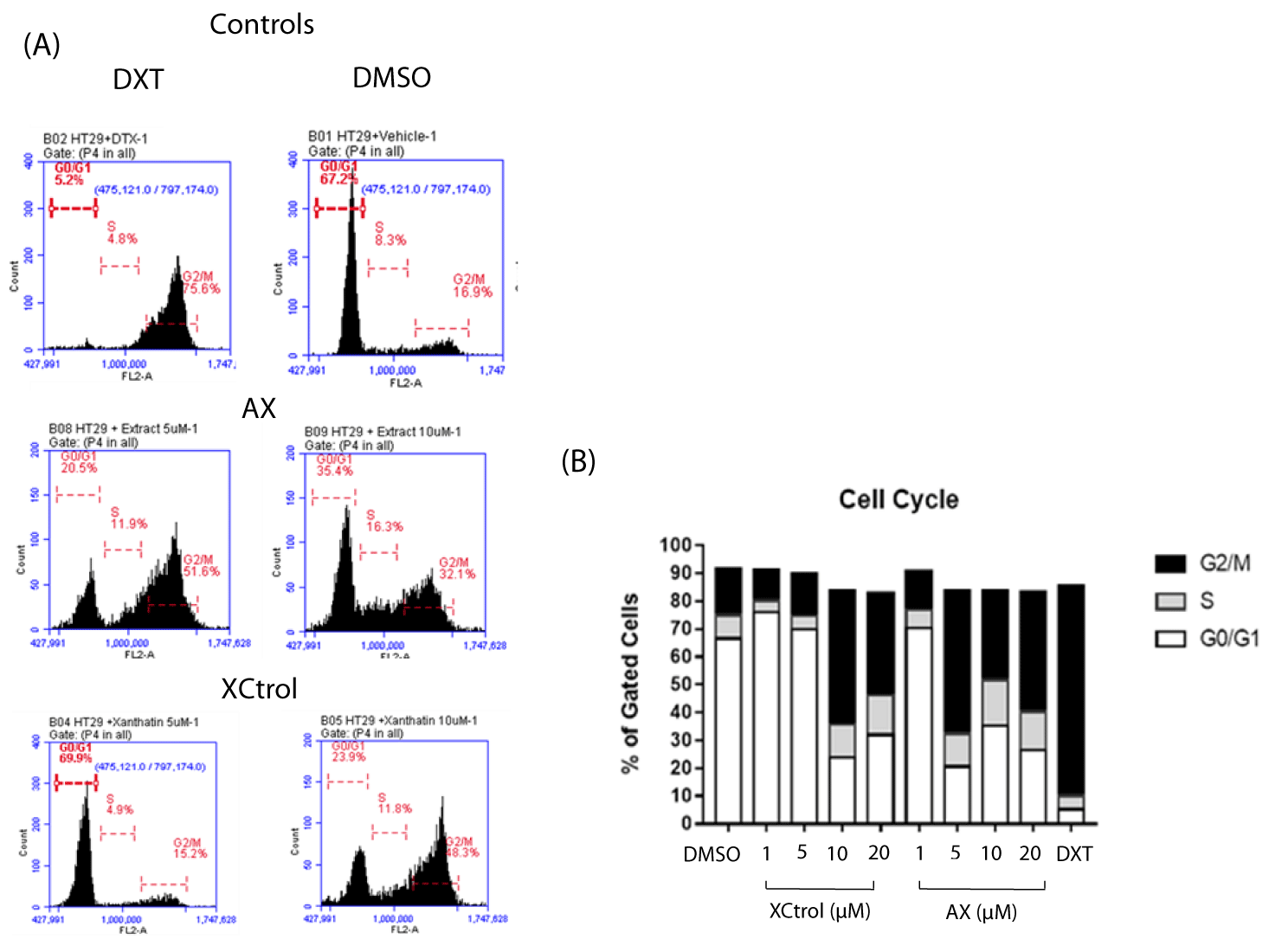
Xanthatin alters HT-29 and HeLa cell cycle division
To evaluate the influence of xanthatin on cell cycle progression in HeLa and HT-29 cancer cells flow cytometry analysis was performed. Two concentrations were tested in the case of HeLa cells (5-10 μM) and four concentrations (1-5-10-20 μM) were tested in the HT-29 cell line. Methanol was used as negative control for HeLa cells and DMSO was used in the case of HT-29 cells; for both cases docetaxel (2 μM) was used as positive control. Commercial xanthatin was included to compare the activity regarding to isolated xanthatin and to ensure that the isolated molecule has the same effectiveness as the former.
The percentage distribution of cell cycle phases for methanol in HeLa cells in G0/G1 phase, S phase, and G2/M phase was 53.8%, 17.8% and 15.7%, respectively. In the case of Docetaxel, the distribution of G0/G1 phase, S phase, and G2/M phases was 2.9%, 12.6% and 69.2%, respectively. In the case of isolated xanthatin, the results indicate that the molecule also induces cell cycle arrest in the G2/M phase at a concentration of 10 μM, when compared to the negative control; the percentage of cells accumulated for this concentration was 27.5%, 13.3% and 45.7% in the G0/G1, S and G2/M phases The effect on cell cycle progression for those cells treated with commercial xanthatin was significant at 10 μM, where the percentage of cell population accumulated in each phase was 24.7%, 13.2% and 49.3% (G0/G1, S phase, and G2/M, respectively). Cell cycle arrest was observed in the G2/M phase compared to the negative control (Figure 5A,B).
In the case of the HT-29 cell line, the percentage distribution of cell cycle phases for DMSO in G0/G1 phase, S phase and G2/M phase was 67.2%, 8.3% and 16.9%, respectively. For Docetaxel the distribution of G0/G1 phase, S phase and G2/M phase was 5%, 4.8% and 75.6%, respectively. The effect on cell cycle progression for those cells treated with commercial xanthatin was significant at 10 μM, where the percentage of the cell population accumulated in each phase was 23.9%, 11.8% and 48.3% (G0/G1, S phase and G2/M, respectively). Cell cycle arrest was observed in the G2/M phase compared to the negative control. In the case of isolated xanthatin, the results indicate that the extract induces cell cycle arrest in the G2/M phase at a concentration of 5 μM, compared to the negative control; the percentage of cells accumulated for this concentration was 20.5%, 11.9% and 51.6% in the G0/G1, S and G2/M phases (Figure 6A,B).
Similar results were obtained by Piloto-Ferrer J, et al. [3] who evaluated the antiproliferative capacity of xanthatin isolated from the total extract of X. strumarium in the murine colon cancer line CT26WT as well as the possibility of arresting tumor growth and proliferation of metastatic sites (lung) in vivo. For this purpose, they evaluated three concentrations (1 µM, 5 µM and 10 µM) of three plant extracts (total extract, ethanolic extract and xanthatin fraction). The results showed that all three extracts arrested cell cycle proliferation in the G2/M phase in CT26WT cells by interfering with the mitotic apparatus, in a dose- and time-dependent manner. Similarly, Francisco-Fernández M, et al. [17] aimed to evaluate the capacity of X. strumarium chloroform extract (XFC) to target and overcome the chemoresistance phenotype in ovarian cancer cells SKOV-3 and ES-2. These studies proved that among the cell lines tested, which included colon, breast and chemoresistant ovarian cancer cells, the best proliferation inhibitory effect for XFC was against the ovarian cancer cells, ranging from 30 to 35 μg/mL. Given its antiproliferative effects, the authors next addressed if XFC could alter cell cycle division by assessing the different phases (sub-G1, G0/G1, S, and G2/M) of the cycle. They proved that for both cell models the cells remained trapped in the G2/M phase after treatment with XFC, suggesting that the extract could significantly alter the molecular events involve in cell division processes controlling therefore cancer cell proliferation.
Conclusion
Our study presents an optimized method for the isolation and characterization of the metabolite xanthatin from the aerial parts of the species X. strumarium growing in Cuba. The molecule showed antiproliferative activity against two tumor cell lines. It was more active against the HT-29 colorectal cancer cells, causing G2/M phase arrest. Our study provides knowledge about xanthatin isolated from the Cuban X. strumarium. This could lead to the use of this natural molecule in the treatment of colorectal cancer. As well as the design of formulations with technological improvements, from a pharmaceutical point of view, to achieve better and more efficient treatment schemes for colorectal cancer.
Funding
This work was supported by the Quebec-Cuba Cooperation Program and by the Cuban Ministry of Science, Technology and Environment CITMA (Program 10 Basic and Natural Science, Cuba (PN223LH010-079).
Compliance with Ethical Standards
The authors declare that they have no conflict of interest. This article does not contain any studies with human or animal subjects performed by any of the authors.
References
- Rajabi S, Maresca M, Yumashev AV, Choopani R, Hajimehdipoor H. The Most Competent Plant-Derived Natural Products for Targeting Apoptosis in Cancer Therapy. Biomolecules. 2021 Apr 3;11(4):534. doi: 10.3390/biom11040534. PMID: 33916780; PMCID: PMC8066452.
- Piloto-Ferrer J, Stoiber T, Parra AV, Hurtado VY, Ferrada CR, Sanabria MLG, Sánchez-Lamar A. Search of new antimitotics compounds from the Cuban flora. Boletin Latinoamericano y del Caribe de Plantas Medicinales y Aromaticas. 2011;10(1):75-82.
- Piloto-Ferrer J, Sánchez-Lamar Á, Francisco M, González ML, Merino N, Aparicio G, Pérez C, Rodeiro I, Lopes MTP. Xanthium strumarium´s xanthatins induces mitotic arrest and apoptosis in CT26WT colon carcinoma cells. Phytomedicine. 2019 Apr;57:236-244. doi: 10.1016/j.phymed.2018.12.019. Epub 2018 Dec 17. PMID: 30797985.
- Sánchez-Lamar A, Piloto-Ferrer J, Fiore M, Stano P, Cozzi R, Tofani D, Cundari E, Francisco M, Romero A, González ML, Degrassi F. Xanthium strumarium extract inhibits mammalian cell proliferation through mitotic spindle disruption mediated by xanthatin. J Ethnopharmacol. 2016 Dec 24;194:781-788. doi: 10.1016/j.jep.2016.11.006. Epub 2016 Nov 11. PMID: 27840259.
- Kamboj A, Saluja AK. Phytopharmacological review of Xanthium strumarium L.(Cocklebur). International Journal of Green Pharmacy. 2010:4(3).
- Fan W, Fan L, Peng C, Zhang Q, Wang L, Li L, Wang J, Zhang D, Peng W, Wu C. Traditional Uses, Botany, Phytochemistry, Pharmacology, Pharmacokinetics and Toxicology of Xanthium strumarium L.: A Review. Molecules. 2019 Jan 19;24(2):359. doi: 10.3390/molecules24020359. PMID: 30669496; PMCID: PMC6359306.
- Romero M, Zanuy M, Rosell E, Cascante M, Piulats J, Font-Bardia M, Balzarini J, De Clerq E, Pujol MD. Optimization of xanthatin extraction from Xanthium spinosum L. and its cytotoxic, anti-angiogenesis and antiviral properties. Eur J Med Chem. 2015 Jan 27;90:491-6. doi: 10.1016/j.ejmech.2014.11.060. Epub 2014 Dec 1. PMID: 25481815; PMCID: PMC7115430.
- Ruiz-Reyes E, Suarez M. Lactonas sesquiterpénicas. Diversidad estructural y sus actividades biológicas. Revista CENIC Ciencias Biológicas. 2015;46(1):9-24.
- Zaki M, Loubidi M, Bilgiç T, Birim D, Akssira M, Dagcı T, Berteina-Raboin S, Saso L, Khouili M, Armagan G. Design, Synthesis, and Biological Evaluation of Novel Tomentosin Derivatives in NMDA-Induced Excitotoxicity. Pharmaceuticals (Basel). 2022 Mar 30;15(4):421. doi: 10.3390/ph15040421. PMID: 35455419; PMCID: PMC9027110..
- Bosco A, Golsteyn RM. Emerging Anti-Mitotic Activities and Other Bioactivities of Sesquiterpene Compounds upon Human Cells. Molecules. 2017 Mar 13;22(3):459. doi: 10.3390/molecules22030459. PMID: 28335397; PMCID: PMC6155193.
- Geng YD, Zhang L, Wang GY, Feng XJ, Chen ZL, Jiang L, Shen AZ. Xanthatin mediates G2/M cell cycle arrest, autophagy and apoptosis via ROS/XIAP signaling in human colon cancer cells. Nat Prod Res. 2020 Sep;34(18):2616-2620. doi: 10.1080/14786419.2018.1544976. Epub 2018 Dec 27. PMID: 30587055.
- Zhou Y, Zhu X, Lin S, Zhu C, Wu L, Chen R, Chen Z, Li W. A Novel Nanoparticle Preparation to Enhance the Gastric Adhesion and Bioavailability of Xanthatin. Int J Nanomedicine. 2020 Jul 14;15:5073-5082. doi: 10.2147/IJN.S252049. PMID: 32764937; PMCID: PMC7368565.
- Tao L, Cao Y, Wei Z, Jia Q, Yu S, Zhong J, Wang A, Woodgett JR, Lu Y. Xanthatin triggers Chk1-mediated DNA damage response and destabilizes Cdc25C via lysosomal degradation in lung cancer cells. Toxicol Appl Pharmacol. 2017 Dec 15;337:85-94. doi: 10.1016/j.taap.2017.10.015. Epub 2017 Oct 23. PMID: 29074359.
- Takeda S, Nishimura H, Koyachi K, Matsumoto K, Yoshida K, Okamoto Y, Amamoto T, Shindo M, Aramaki H. (-)-Xanthatin induces the prolonged expression of c-Fos through an N-acetyl-L-cysteine (NAC)-sensitive mechanism in human breast cancer MDA-MB-231 cells. J Toxicol Sci. 2013;38(4):547-57. doi: 10.2131/jts.38.547. PMID: 23824011.
- Piloto-Ferrer J, Sanchez-Lamar A. Blancos mitóticos de drogas naturales y nuevas estrategias para la terapia anti-cáncer. Revista Cubana de Ciencias Biológicas. 2015:4(2).
- Fernandez MF, Piloto-Ferrer J, Annabi B. Targeting of cancer cell drug resistance phenotype and metastasis with xanthium strumarium L. In: Trends in Pharmaceutical Research and Development. Book Publisher International; 2020:59-78. doi: 10.9734/bpi/tprd/v3.
- Francisco Fernandez M, Charfi C, Piloto-Ferrer J, Lidia González M, Lamy S, Annabi B. Targeting Ovarian Cancer Cell Cytotoxic Drug Resistance Phenotype with Xanthium strumarium L. Extract. Evid Based Complement Alternat Med. 2019 Nov 15;2019:6073019. doi: 10.1155/2019/6073019. PMID: 31827554; PMCID: PMC6885198.
- Ullah R, Nasrullah Khan, Nina Hewitt, Kishwar Ali, David Aaron Jones, Muhammad Ezaz Hasan Khan. Invasive species as rivals: Invasive potential and distribution pattern of xanthium strumarium sustainability. 2022;14(12):7141. doi: 10.3390/su14127141.
- Ramírez-Erosa I, Huang Y, Hickie RA, Sutherland RG, Barl B. Xanthatin and xanthinosin from the burs of Xanthium strumarium L. as potential anticancer agents. Can J Physiol Pharmacol. 2007 Nov;85(11):1160-72. doi: 10.1139/Y07-104. PMID: 18066118.
- Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983 Dec 16;65(1-2):55-63. doi: 10.1016/0022-1759(83)90303-4. PMID: 6606682.
- Zhi XY, Song LL, Liang J, Wei SQ, Li Y, Zhang Y, Hao XJ, Cao H, Yang C. Synthesis and in vitro antifungal activity of new Michael-type amino derivatives of xanthatin, a natural sesquiterpene lactone from Xanthium strumarium L. Bioorg Med Chem Lett. 2022 Jan 1;55:128481. doi: 10.1016/j.bmcl.2021.128481. Epub 2021 Nov 28. PMID: 34852242.
- Li L, Liu P, Xie Y, Liu Y, Chen Z, Geng Y, Zhang L. Xanthatin inhibits human colon cancer cells progression via mTOR signaling mediated energy metabolism alteration. Drug Dev Res. 2022 Feb;83(1):119-130. doi: 10.1002/ddr.21850. Epub 2021 Jun 28. PMID: 34180556.
- Xie Y, Zhu X, Liu P, Liu Y, Geng Y, Zhang L. Xanthatin inhibits non-small cell lung cancer proliferation by breaking the redox balance. Drug Dev Res. 2022 Aug;83(5):1176-1189. doi: 10.1002/ddr.21941. Epub 2022 Apr 24. PMID: 35466412.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.