Medicine Group. 2024 September 20;5(9):1164-1175. doi: 10.37871/jbres2005.
Periodontal Disease Shortens the Telomere Length in Human Gingival Crevicular Epithelium Cells and Human Umbilical Endothelial Cells
Kazuo Sonoki1*, Kosuke Muraoka2, Masaki Morishita2, Shuji Awano2 and Keisuke Nakashima3
2Division of Clinical Education Development and Research, Department of Oral Function, Kyushu Dental University, Kitakyushu, Fukuoka, Japan
3Division of Periodontology, Department of Oral Function, Kyushu Dental University, Kitakyushu, Fukuoka, Japan
- Periodontal disease
- Telomere length
- Gingival crevicular epithelium cells
- HUVECs
- Lipopolysaccharide
Abstract
Background: Telomere length, a biomarker of cellular aging, is decreased by inflammation. It is uncertain whether chronic inflammation, such as periodontal disease (PD), shortens telomere lengths. We compared the telomere lengths of gingival crevicular epithelium cells from both the healthy side and the diseased side of patients with PD and investigated whether lipopolysaccharide derived from a periodontal bacterium, Porphyromonas gingivalis (Pg-LPS) shortened the telomere lengths of human umbilical endothelial cells (HUVECs).
Methods: We extracted genomic DNA from gingival crevicular epithelium cells on the diseased and healthy sides collected from 25 patients with PD (mean age 72.4 years, 17 females. 8 males) and measured the telomere lengths by the polymerase chain reaction (PCR) method. After stimulating HUVECs with Pg-LPS at concentrations of 0.05, 0.1, and 1.0 µg/mL, we extracted genomic DNA and measured the telomere lengths.
Results: The telomere lengths on the patients' diseased sides were significantly shorter than those on the healthy sides (392.6 ± 232.8 kb vs. 599.5 ± 467.0 kb, p = 0.004). In the HUVECs, a concentration-dependent but nonsignificant decrease in telomere length was observed in response to Pg-LPS. Expressed as a ratio relative to the telomere lengths of unstimulated HUVECs, the ratio of telomere lengths decreased in a concentration-dependent manner, and the Pg-LPS concentration 1.0 µg/mL significantly decreased the ratio compared to the telomere lengths of unstimulated HUVECs.
Conclusions: Gingival crevicular epithelial cells of individuals with PD are believed to undergo aging due to telomere shortening. The involvement of one of the factors of periodontal virulence, LPS, is suspected as the mechanism.
Background
Telomeres are the heterochromatic repeat regions at the ends of eukaryotic chromosomes, and the length of telomeres is considered a determinant of biological aging. Normal aging is associated with telomere shortening, and the rate of this shortening may be further increased by inflammation and oxidative stress [1]. Periodontal disease is characterized by chronic inflammation caused by periodontal bacteria, and recent evidence suggests that chronic inflammatory diseases such as type 2 diabetes mellitus, atherosclerosis, obesity, rheumatoid arthritis, and periodontitis are associated with shorter leukocyte telomere length (LTL), independent of an individuals' age [2]. In the context of the relationship between LTL and periodontitis, three clinical studies revealed that the LTL was shorter in individuals with chronic periodontitis compared to healthy controls [3-5]. However, two other studies observed that the LTL was comparable between patients with periodontitis and controls [6,7]. The discrepancy might be explained by the differences in study populations, the definition of periodontitis, and the techniques used to measure LTLs [5]. In order to avoid these differences, minimize the other factors influencing telomere length as much as possible, and clarify the telomere shortening that is due to periodontal disease in humans, we conducted the present study to compare the telomere length of gingival crevicular epithelium cells from both the healthy side and the diseased side in patients with periodontal disease. We speculated that in patients with chronic periodontal disease (rather than in acute cases), the telomere length on the diseased side would be shorter than that on the healthy side.
Telomere length shortens at each cell replication due to incomplete lagging strand replication, as a phenomenon that is called the 'end-replication problem. This telomere attrition causes cell senescence when a telomere's length reaches a critically short length [8]. In a pro-inflammatory milieu, the length of telomeres is sensitive to inflammation because more telomeres are lost with a high blood cells replication rate [1,5]. Lipopolysaccharide (LPS) is an endotoxin released mainly from Gram-negative bacteria during inflammation (such as the inflammation that occurs in sepsis and periodontitis). In vitro investigations using lung alveolar epithelial cells simulated with LPS revealed a slight decrease in telomere length that occurred in a concentration-dependent manner [9]. In another study however, LPS induced features of premature senescence of stromal-vascular cells; i.e., it induced the activation of p53, an elevation of SA-β-galactosidase activity, and increased hydrogen peroxide production, but not telomere-length shortening [10]. Differences in the telomere-length shortening induced by LPS thus exist, and these differences are probably due to the cell lines and/or types of LPS and usage of LPS.
We have reported that in Human Umbilical Vascular Endothelial Cells (HUVECs), high glucose enhanced the expression of Monocyte Chemoattractant Protein-1 (MCP-1), which is an atherosclerosis-promoting factor and is induced by a virulence factor of periodontal disease: LPS derived from a periodontal pathogenic bacterium, Porphyromonas gingivalis (Pg-LPS) [11]. We have found no published report of a study of the effects of Pg-LPS on the telomere length of HUVECs, and we thus decided to measure the telomere length of HUVECs after stimulation with Pg-LPS. We hypothesized that Pg-LPS stimulation would shorten the telomere length of HUVECs.
Materials and Methods
Patients
For the measurement of the telomere lengths of gingival crevicular epithelium cells from patients with chronic periodontal disease, we recruited 31 consecutive periodontal patients at the maintenance stage who were attending the outpatient clinic of Kyushu Dental University Hospital during the period from June to August in 2023. Written informed consent was obtained from all patients, in line with the Helsinki Declaration, before inclusion in this study. Ethical approval for each assessment phase was obtained from the the Human Ethics Committee of Kyushu Dental University (No. 22-57). The patients were 20 females and 11 males aged 55-84 years (mean ± SD 71.7 ± 7.4 years). A single dentist (K.M.) examined the periodontal status of each patient including the current number of teeth, the probing pocket depth (PPD), the attachment loss (AL), bleeding on probing (BOP), and the mobility of teeth, just before collecting the patient's gingival crevicular epithelium cells. The diseased and healthy sides were determined from the condition of the periodontal tissue. The diseased side was determined from the presence or absence of more than 4 mm PPD, gingival bleeding and gingival redness. The collection of the gingival crevicular epithelium cells was done by rubbing the gingival sulcus with interdental brushes (DENT.EX®, 4S type, Lion Co., Mie, Japan) gently from the healthy side and from the diseased side in each patient. In this process, a minimum of three interdental brushes per single tooth were used on the diseased or healthy side. The collection was performed while avoiding bleeding during the collection of the gingival crevicular epithelium cells with the interdental brushes, in order to prevent contamination from blood cells. If bleeding occurred, the interdental brush with blood was not used for genomic DNA extraction.
Genomic DNA extraction and the measurement of the telomere length
The genomic DNA was extracted from the gingival crevicular epithelium cells with the use of the Genomic DNA Purification Kit (MagExtractor, NPK-101, Toyobo, Osaka, Japan) from the three interdental brushes detached from their handles and held in a 1.5-mL tube. We measured the telomere lengths in the gingival crevicular epithelium cells by conducting a real-time Polymerase Chain Reaction (PCR) using the Telomere Length qPCR Kit (Absolute Human Telomere Length Quantification qPCR Assay Kit, #8918, ScienCell Research Laboratories, Carlsbad, CA, USA) and a LightCycler 96® PCR system (Roche, Rotkreuz, Switzerland).
In a preliminary experiment, the concentration of genomic DNA obtained from the three interdental brushes was ≥10 ng/µL in 100 µL, which is sufficient for the measurement of telomere lengths using real-time PCR. In brief, 1 µL of an extracted genomic DNA sample or a reference human genomic DNA sample (telomere length 726 ± 70 kb per diploid cell) was added to 9 µL of stock solution consisting of 2 µL of a telomere primer set or a single-copy reference (SCR) primer set and 7 µL of nuclease-free H2O in each well of LightCycler 8-Tube Strips. Then, 10 µL of 2×GoldNStart TaqGreen qPCR master mix (dye: SYBR™ Green I) was added for the total reaction liquid volume to become 20 µL. Several LightCycler eight-tube strips were mounted symmetrically on the block cycler unit of the LightCycler 96 instrument, and the real-time PCR was started. The reaction conditions were as follows: initial denaturation at 95°C for 10 min, three-step amplification (35 cycle): first step (denaturation) at 95°C for 20 sec, second step (annealing) at 52°C for 20 sec, and third step (extension) at 72°C for 45 sec, melting at 95°C for 10 sec, 60°C for 60 sec, and 95°C for 1 sec, and cooling at 40°C for 180 sec. The telomere length was calculated using the quantification cycle (Cq) obtained from the LightCycler 96. The ΔCq (Telomere) is the difference in the quantification cycle's number of telomeres between the target and the reference genomic DNA samples. The ΔCq (SCR) is the difference in the quantification cycle's number of SCRs between the target and the reference genomic DNA samples. The ΔΔCq is the difference between the ΔCq (Telomere) and the ΔCq (SCR). The relative telomere length of the target sample to the reference sample is expressed as 2−ΔΔCq. The total telomere length of the target sample per diploid cell is expressed as the reference sample telomere length (726 ± 70 kb) × 2−ΔΔCq.
Cell culture
HUVECs were harvested from umbilical cord veins and cultured in M199 medium supplemented with 10% Fetal Calf Serum (FCS; Invitrogen, Carlsbad, CA), 20 µg/mL endothelial cell growth supplement (Sigma-Aldrich, St. Louis, MO), 100 µg/mL heparin (Sigma-Aldrich), and 5 μL/mL penicillin-streptomycin liquid (Sigma-Aldrich) in 10-cm gelatin-coated dishes (cat.# 4020-020, Iwaki, Tokyo). Cells were used within six passages.
The HUVECs were then seeded onto 35-mm gelatin-coated dishes, and we divided the HUVECs into two groups: the unstimulated group with no administration of Pg-LPS (cat.# tlrl-pglps, InvivoGen, San Diego, CA) (one dish; the Base group) and the stimulated group with Pg-LPS administration at three different concentrations: 0.05, 0.1, and 1.0 µg/mL (three dishes: the L0.05, L0.1, and L1.0 groups). The HUVECs were cultured until they reached confluence, with a change of 2 mL of culture medium and Pg-LPS administration every 2 days. The HUVECs typically reached confluence in 3 days. We subsequently conducted genomic DNA extraction and measured the total telomere length using real-time PCRs with the HUVECs as was done with the patients' gingival crevicular epithelium cells. The total telomere length of each group was normalized with the value of the Base group set to 1.00.
Statistical Analysis
All data are presented as the mean ± SD. Matched pairs from two groups were compared by the Wilcoxon test. We used Pearson's correlation coefficient to determine the correlations between pairs of binary variables. The comparison of multiple groups was performed by the Kruskal-Wallis test. If a significant difference was found in the Kruskal-Wallis test (p < 0.05), a post hoc Mann-Whitney U-test with a significance level of 0.0083 (adjusted using the Bonferroni correction) was performed for pairwise comparisons between the two groups. Results were considered significant when the p-value was <0.05 (SPSS 17.0, SAS, Cary, NC).
Results
The patients' characteristics
Of the 32 recruited periodontal patients at the maintenance stage, we obtained a sufficient amount of genomic DNA from both sides of the crevicular epithelium in 25 patients: 17 females, 8 males, 55-84 years old (72.4 ± 7.8 yrs). The average treatment period from the first visit to the date of the crevicular epithelium collection was 119.4 ± 77.2 months. In an oral examination, the average current number of teeth was 23.4 ± 4.5 teeth. The sampling locations for the patients' periodontal-disease sides were upper-right (n = 8), upper-left (n = 15), lower-left (n = 0), and lower-right (n = 2). The sampling locations for the patients' healthy sides were upper-right (n = 13), upper-left (n = 10), lower-left (n = 2), and lower-right (n = 0). The maximum PPD was 2.5 ± 0.6 mm on the healthy sides and 3.6 ± 0.9 mm on the diseased sides (p = 0.000). The maximum AL was 3.1 ± 1.1 mm on the healthy sides and 4.6 ± 2.1 mm on the diseased side (p = 0.001). BOP was observed in 4 sites on the healthy side and 19 sites on the diseased side. The mobility of teeth in periodontal disease was up to level 2 at six sites on the diseased side, and up to level 2 at three sites on the healthy side.
The telomere lengths of the gingival crevicular epithelium cells of periodontal patients
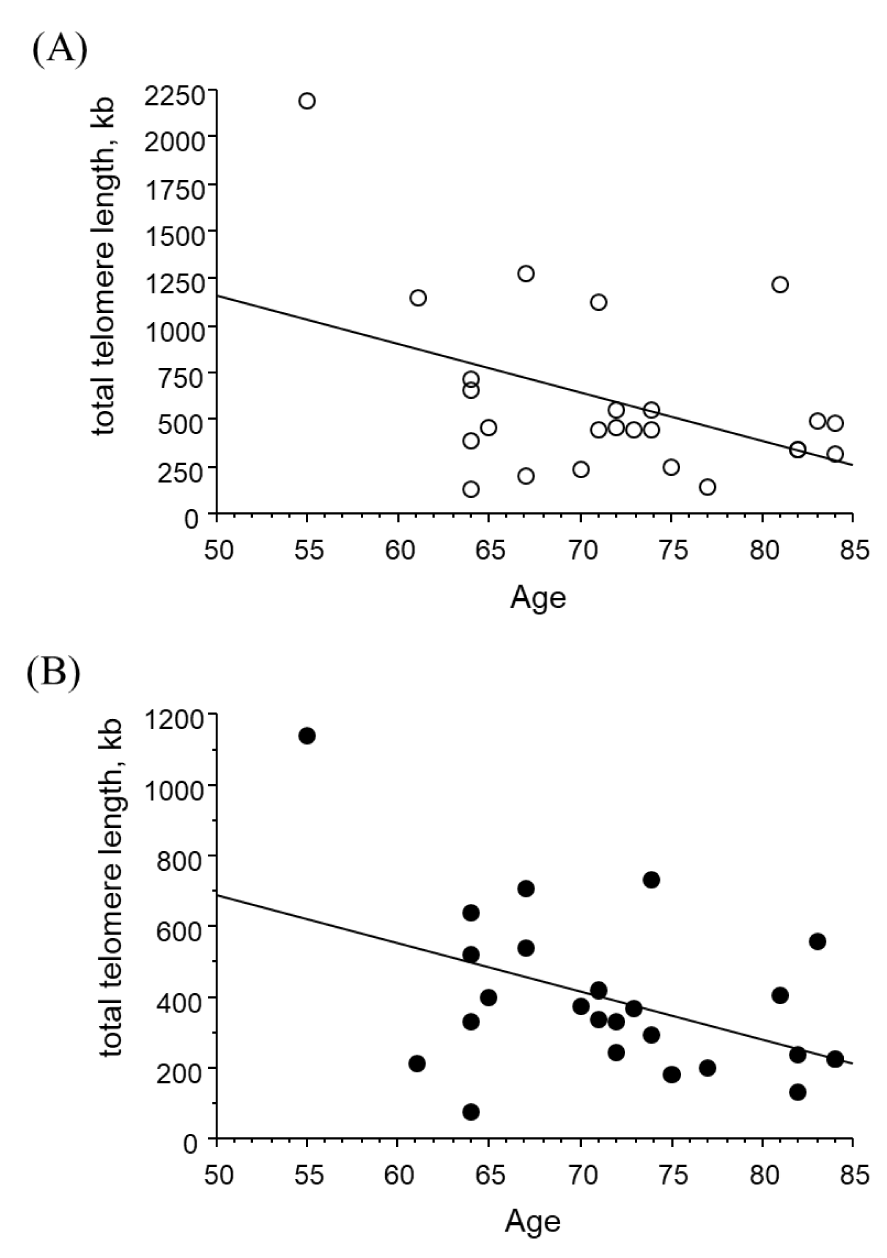
The total telomere length of gingival crevicular epithelial cells on the patients' healthy sides was 599.5 ± 467.0 kb and significantly shorter on the diseased sides at 392.6 ± 232.8 kb (p = 0.004). Figure 1 illustrates the relationship between the patients' ages and the total telomere length on each side. The total telomere length from each side showed a significant negative correlation with age (healthy side: correlation coefficient = −0.460, p = 0.021, diseased side: correlation coefficient = −0.437, p = 0.029). Our investigation of the correlation between the total telomere length from each side and the treatment period from the initial visit to the collection of gingival crevicular epithelium cells revealed no significant relationship. In addition, there was no significant correlation between the telomere length of the gingival epithelial cells and the maximum PPD or maximum AL on either the healthy or disease sides.
The telomere length of the HUVECs stimulated by Pg-LPS
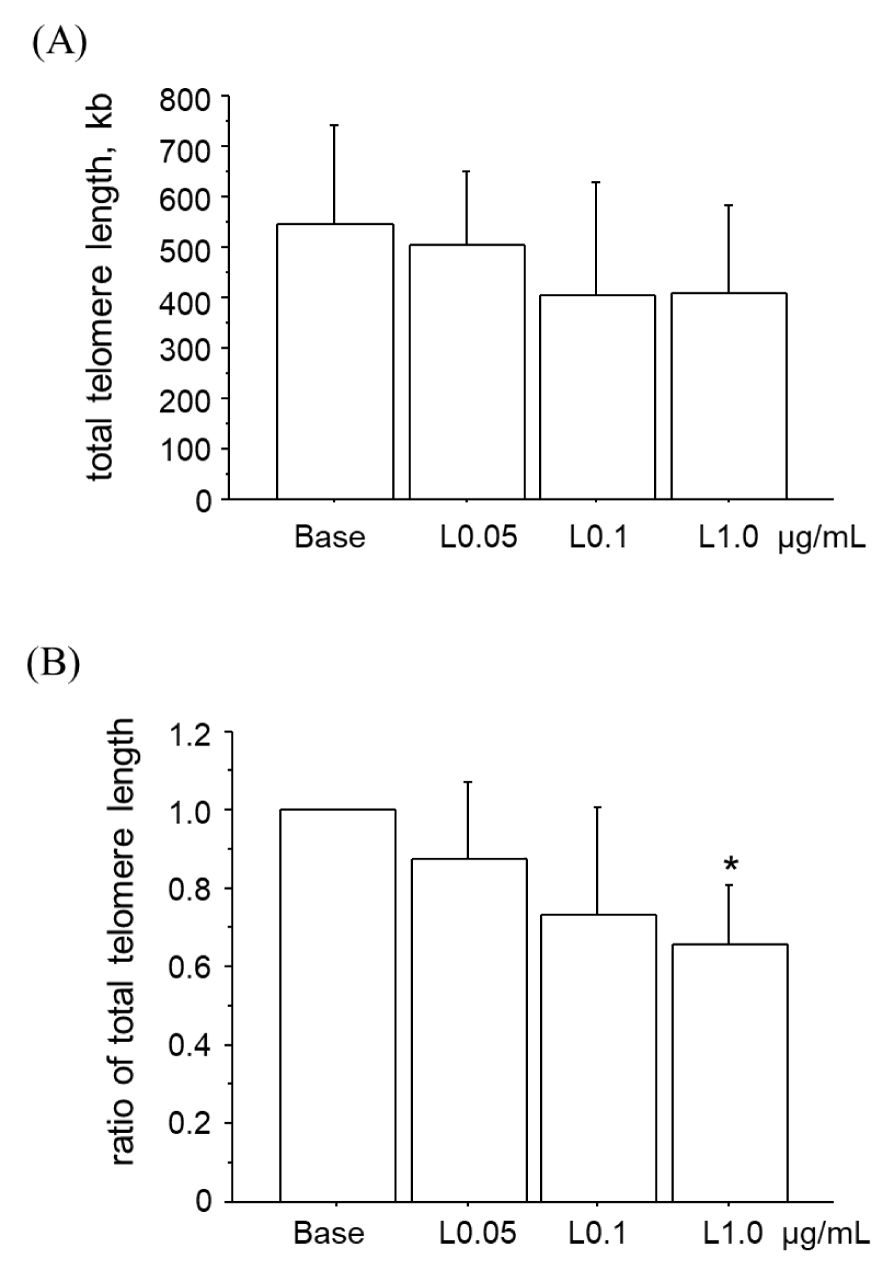
Figure 2 illustrates the total telomere length of HUVECs after approx. 72 hr of culture following the initiation of stimulation with different concentrations of Pg-LPS until cell confluence was achieved. The total telomere length (Figure 2A) showed a decreasing trend in response to the concentration of Pg-LPS, but the trend was not significant (p = 0.237). However, compared to the total telomere length of the unstimulated HUVECs (Figure 2B; the ratio of total telomere length), the total telomere length was significantly decreased in response to the concentration of Pg-LPS (p = 0.002). Specifically, a significant difference was observed (p = 0.000 <0.0083: Bonferroni correction) between the unstimulated HUVECs (Base group) and the HUVECs stimulated with Pg-LPS at the concentration 1.0 µg/mL (the L1.0µg/mL group), resulting in a 34% reduction in total telomere length compared to the unstimulated HUVECs.
Discussion
The results of this study demonstrated that periodontal disease shortens the telomere length of human cells, i.e., gingival crevicular epithelial cells and endothelial cells (HUVECs), both in vivo and in vitro. These results are consistent with reports on the leukocyte telomere length (LTL) in periodontal patients [3-5], and they suggest that the length of telomeres is sensitive to inflammation and to decreases in a high rate of blood-cell replication [1,5]. Our comparison of the telomere lengths of gingival crevicular epithelial cells from the diseased and healthy sides of the same individuals indicates that the observed differences in telomere lengths were influenced solely by the extent of periodontal disease, i.e., the degree of inflammation. However, we detected no significant correlation between the telomere length of the gingival epithelial cells and PPD or AL, whereas aging shortened the total telomere length similarly on both sides (Figure 1). In the papers by Masi [4] and Song [5], the shortening of LTL in periodontal patients was reported, and a negative correlation between LTL and alveolar bone loss or bleeding on probing (BOP) was observed. These reports had significantly larger sample sizes of 3,478 and 563 subjects, respectively, compared to our 25 subjects, suggesting a higher likelihood of detecting significant associations. Additionally, we measured telomere length in gingival crevicular epithelial cells instead of leukocytes. Due to the potentially shorter turnover of leukocytes in the bloodstream compared to gingival crevicular epithelial cells, LTL may be more susceptible to inflammation at the time of sampling. Conversely, the telomere length in gingival crevicular epithelial cells may be influenced by the duration of ongoing inflammation. In our measurement results, although not statistically significant, the correlation coefficient between the duration from the first visit to sampling and the telomere length on the diseased side was -0.206 (p = 0.324), indicating a negative correlation (Data not shown). It is highly intriguing that differences in telomere length can occur due to the effects of inflammation, even within the same tissue of the same individual.
In human somatic cells, the length of telomeres gradually shortens with each cell division; however, in germ cells, the enzyme telomerase, which extends telomeric DNA, is expressed, ensuring an infinite replicative lifespan. The expression of telomerase in gingival tissue and gingival crevicular fluid is rarely observed in healthy individuals but is enhanced in periodontal disease patients [12]. Moreover, a positive correlation has been reported between periodontal parameters such as gingival index, plaque index, probing depth, clinical attachment loss, and telomerase expression. In this case, telomere length in patients with periodontal disease may be longer than that in healthy individuals, potentially contradicting our current measurement results. Therefore, future investigations should consider not only telomere length measurement but also telomerase expression and activity assessment.
The results of our in vitro study using HUVECs demonstrated that periodontal bacteria-derived lipopolysaccharides (Pg-LPS) have a concentration-dependent effect, particularly at the 1.0 µg/mL concentration, significantly shortening the telomere length of HUVECs. If HUVECs were replaced with gingival crevicular epithelial cells, we suspect that the difference in the Pg-LPS concentration at the local level of periodontal tissue corresponds to the difference in the telomere length of human gingival crevicular epithelial cells. However, it's important to note that Pg-LPS may also affect the gingival crevicular epithelial cells on the healthy side, as Pg-LPS can flow into the bloodstream from the diseased side, reaching the healthy side. It is of interest to investigate these findings in other human periodontal tissues, other than HUVECs.
Telomere attrition causes cell senescence when the telomere length reaches a critically short length [8]. Shortened LTL has been associated with inflammation-related diseases including cardiovascular disease [13], type 2 diabetes [14], dementia [15], and cancer [16]. The telomere length in leukocytes is thus considered a useful surrogate marker for the status of various diseases. Cells undergoing senescence, which is a state of cell aging, secrete various factors such as inflammatory cytokines and chemokines with pro-inflammatory and pro-cancer effects (e.g., interleukin [IL]-6, tumor necrosis factor [TNF]α, MCP-1, etc.). This phenomenon is referred to as the Senescence-Associated Secretory Phenotype (SASP). As aging progresses, the accumulation of senescent cells in the body leads to chronic inflammation and carcinogenesis in the surrounding tissues through the secretion of SASP factors from these senescent cells [17]. The prevalence and severity of periodontal disease increase with age, and thus aging is one of the significant risk factors for periodontal disease. Hajishengallis G. [18] proposed that periodontal disease occurs as a result of aging, in which changes in periodontal tissues are accompanied by inflammation caused by infection with periodontal bacteria. Hajishengallis G. [18] also suggested that the immune and inflammatory states change with aging (immunosenescence), making individuals more susceptible to periodontal disease. Thus, the onset of periodontal disease is highly dependent on aging.
In 2014, Jurk D, et al. [19] reported that nfkb1 knockout mice, which develop chronic and progressive mild inflammation, experience early aging; the underlying mechanism involves DNA damage by reactive oxygen species, leading to telomere shortening and consequently the accumulation of aging cells. This is another way in which inflammation accelerates aging. Based on the above reports, it has been speculated that there is a bidirectional relationship between periodontal disease and aging. Considering our present findings, it is conceivable that (i) periodontal disease accelerates aging, specifically telomere shortening in periodontal tissue, and (ii) the aging of periodontal tissue, in turn, may further promote periodontal disease through the Senescence-Associated Secretory Phenotype (SASP). If this is the case, treatment to sever this bidirectionality would be required to maintain the health of periodontal tissues.
We reported that Pg-LPS upregulates the secretion of MCP-1 in HUVECs, particularly in a high-concentration glucose environment, and we thus speculated that periodontal pathogens could initiate or contribute to the progression of atherosclerosis, if the pathogens move out of a periodontal lesion into the bloodstream reaching the vasculature [11]. Our present study's finding that Pg-LPS shortened the telomere length of HUVECs also suggests the potential promotion of atherosclerosis as a result of accelerating aging. This is because cellular senescence has also been confirmed to be a contributing factor in the promotion of atherosclerosis [20]. Regarding the production of MCP-1, we suspect that the shortening of telomere lengths in HUVECs, i.e., cellular aging caused by Pg-LPS, would enhance the cells' MCP-1 production. We plan to investigate this possibility in future research and aim to elucidate how periodontal disease promotes atherosclerosis through inflammation and aging.
In conclusion, periodontal disease shortened the telomere lengths, a biomarker of cellular aging, in crevicular epithelial cells from patients with periodontal disease. Our study also revealed that periodontal bacteria-derived LPS, which is believed to act on blood vessels from periodontal lesions through the bloodstream, shortens telomere lengths in vascular endothelial cells.
Acknowledgments
We gratefully acknowledge the superb assistance from Waka Yokota, Ayase Tanaka, Rio Yamasaki, Rina Ogata, Nozomi Sato, and Kaede Takachi.
Funding
This work was funded by Kyushu Dental University, Kitakyushu, Fukuoka, Japan.
Competing Interest
All authors declare no conflicts of interest.
References
- Zhang J, Rane G, Dai X, Shanmugam MK, Arfuso F, Samy RP, Lai MK, Kappei D, Kumar AP, Sethi G. Ageing and the telomere connection: An intimate relationship with inflammation.Ageing Res Rev. 2016 Jan;25:55-69. doi: 10.1016/j.arr.2015.11.006. Epub 2015 Nov 23. PMID: 26616852.
- Steffens JP, Masi S, D'Aiuto F, Spolidorio LC. Telomere length and its relationship with chronic diseases – New perspectives for periodontal research.Arch Oral Biol. 2013 Feb;58(2):111-7. doi: 10.1016/j.archoralbio.2012.09.009. Epub 2012 Nov 30. PMID: 23201158.
- Sanders AE, Divaris K, Naorungroj S, Heiss G, Risques RA. Telomere length attrition and chronic periodontitis: An ARIC Study nested case-control study. J Clin Periodontol. 2015 Jan;42(1):12-20. doi: 10.1111/jcpe.12337. Epub 2014 Dec 26. PMID: 25418689.
- Masi S, Salpea KD, Li K, Parkar M, Nibali L, Donos N, Patel K, Taddei S, Deanfield JE, D'Aiuto F, Humphries SE. Oxidative stress, chronic inflammation, and telomere length in patients with periodontitis. Free Radic Biol Med. 2011 Mar 15;50(6):730-5. doi: 10.1016/j.freeradbiomed.2010.12.031. Epub 2010 Dec 30. PMID: 21195167.
- Song W, Yang J, Niu Z. Association of periodontitis with leukocyte telomere length in US adults: A cross-sectional analysis of NHANES 1999 to 2002. J Periodontol. 2021 Jun;92(6):833-843. doi: 10.1002/JPER.20-0269. Epub 2020 Oct 27. PMID: 32996594.
- Thomson WM, Zeng J, Broadbent JM, Foster Page LA, Shalev I, Moffitt TE, Caspi A, Williams SM, Braithwaite AW, Robertson SP, Poulton R. Telomere length and periodontal attachment loss: A prospective cohort study. J Clin Periodontol. 2016 Feb;43(2):121-7. doi: 10.1111/jcpe.12499. Epub 2016 Feb 19. PMID: 26713854.
- Takahashi K, Nishida H, Takeda H, Shin K. Telomere length in leukocytes and cultured gingival fibroblasts from patients with aggressive periodontitis. J Periodontol. 2004 Jan;75(1):84-90. doi: 10.1902/jop.2004.75.1.84. PMID: 15025219.
- Levy MZ, Allsopp RC, Futcher AB, Greider CW, Harley CB. Telomere end-replication problem and cell aging. J Mol Biol. 1992 Jun 20;225(4):951-60. doi: 10.1016/0022-2836(92)90096-3. PMID: 1613801.
- Kim CO, Huh AJ, Han SH, Kim JM. Analysis of cellular senescence induced by lipopolysaccharide in pulmonary alveolar epithelial cells. Arch Gerontol Geriatr. 2012 Mar-Apr;54(2):e35-41. doi: 10.1016/j.archger.2011.07.016. Epub 2011 Aug 25. PMID: 21871670.
- Zhao M, Chen X. Effect of lipopolysaccharides on adipogenic potential and premature senescence of adipocyte progenitors. Am J Physiol Endocrinol Metab. 2015 Aug 15;309(4):E334-44. doi: 10.1152/ajpendo.00601.2014. Epub 2015 Jun 23. PMID: 26105007.
- Sonoki K, Muraoka K, Hikiji H. Enhancement of porphyromonas gingivalis-lipopolysaccharide induced MCP-1 expression by high glucose in human endothelial cells. Journal of Dental and Oral Biology. 2020;1:6. doi: 10.47496/nl.JDOB.2020.01.01.
- Katarkar A, Saha A, Mukherjee S, Kundu D, Bandyopadhyay P, Chaudhuri K. Telomerase expression in individuals with chronic and aggressive periodontitis. J Periodontol. 2015 May;86(5):656-65. doi: 10.1902/jop.2015.140540. Epub 2015 Feb 9. PMID: 25660499.
- Samani NJ, Boultby R, Butler R, Thompson JR, Goodall AH. Telomere shortening in atherosclerosis. Lancet. 2001 Aug 11;358(9280):472-3. doi: 10.1016/S0140-6736(01)05633-1. PMID: 11513915.
- Cheng F, Luk AO, Shi M, Huang C, Jiang G, Yang A, Wu H, Lim CKP, Tam CHT, Fan B, Lau ESH, Ng ACW, Wong KK, Carroll L, Lee HM, Kong AP, Keech AC, Chow E, Joglekar MV, Tsui SKW, So WY, So HC, Hardikar AA, Jenkins AJ, Chan JCN, Ma RCW. Shortened leukocyte telomere length is associated with glycemic progression in type 2 diabetes: A prospective and mendelian randomization analysis. Diabetes Care. 2022 Mar 1;45(3):701-709. doi: 10.2337/dc21-1609. PMID: 35085380
- Honig LS, Kang MS, Schupf N, Lee JH, Mayeux R. Association of shorter leukocyte telomere repeat length with dementia and mortality. Arch Neurol. 2012 Oct;69(10):1332-9. doi: 10.1001/archneurol.2012.1541. PMID: 22825311.
- Samavat H, Luu HN, Beckman KB, Jin A, Wang R, Koh WP, Yuan JM. Leukocyte telomere length, cancer incidence and all-cause mortality among Chinese adults: Singapore Chinese Health Study. Int J Cancer. 2021 Jan 15;148(2):352-362. doi: 10.1002/ijc.33211. Epub 2020 Aug 5. PMID: 33459354.
- Nelson G, Kucheryavenko O, Wordsworth J, von Zglinicki T. The senescent bystander effect is caused by ROS-activated NF-κB signalling. Mech Ageing Dev. 2018 Mar;170:30-36. doi: 10.1016/j.mad.2017.08.005. Epub 2017 Aug 25.PMID: 28837845.
- Hajishengallis G. Aging and its impact on innate immunity and inflammation: Implications for periodontitis. J Oral Biosci. 2014 Feb 1;56(1):30-37. doi: 10.1016/j.job.2013.09.001. PMID: 24707191.
- Jurk D, Wilson C, Passos JF, Oakley F, Correia-Melo C, Greaves L, Saretzki G, Fox C, Lawless C, Anderson R, Hewitt G, Pender SL, Fullard N, Nelson G, Mann J, van de Sluis B, Mann DA, von Zglinicki T. Chronic inflammation induces telomere dysfunction and accelerates ageing in mice. Nat Commun. 2014 Jun 24;2:4172. doi: 10.1038/ncomms5172. PMID: 24960204.
- Wang JC, Bennett M. Aging and atherosclerosis: Mechanisms, functional consequences, and potential therapeutics for cellular senescence. Circ Res. 2012 Jul 6;111(2):245-59. doi: 10.1161/CIRCRESAHA.111.261388. PMID: 22773427.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.