Environmental Sciences Group. 2024 September 11;5(9):1104-1106. doi: 10.37871/jbres1995.
Initial Understanding of Per and Poly-Fluoroalkyl Compounds (PFAS): Synopsis
Kalidas Mainali*
- PFAS
- Adsorption
- Biochar
Abstract
The increasing attention in academic and industry research on per and Poly-Fluoroalkyl compounds (PFAS) in the environment is a result of growing concern. If PFAS are exposed to human health, they are also known to be dangerous. Utilizing leftover lignocellulosic biomass, biochar is a pyrolytic material that can be used as an inexpensive adsorbent to remove environmental pollutants. It is essential to adjust the physical and chemical properties of biochar due to its low concentration and distinct chemical composition. Most notably, char's high surface area and C and N content are the primary controlling elements during PFAS adsorption. This summary provides background information on PFAS pollutants and possible remediation strategies.
Introduction
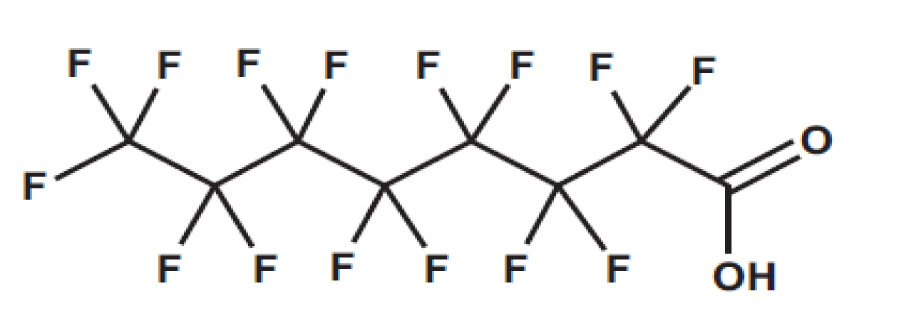
Since the 1950s, a wide range of synthetic fluorinated organic chemicals, known as perfluoroalkyl and Poly-Fluoroalkyl substances (PFAS), have been employed in various industrial and commercial settings [1,2]. Wastewater from Treatment Plants (WWTPs) is a significant source of per- and Poly-Fluoroalkyl Substances (PFAS) into drinking and natural waters) [3]. Strong carbon-fluorine linkages are associated with chemical and thermal stability, which is another characteristic that adds to the persistent presence of per- and Poly-Fluoroalkyl Substances (PFAS) in the environment, garnering them the label of "forever chemicals." Moreover, the exceptionally low surface tension provided by the fluorine moiety of PFAS contributes to their distinct hydrophobic and lipophobic qualities. PFASs have been extensively employed and then released into the environment, resulting in the identification of many of these compounds in drinking water, wildlife, municipal wastewater, ambient air, human blood, and nursing mother's milk [4]. In addition, PFAS are introduced via pesticide use and waste management techniques including landfills and rural chimneys [5]. Among these, PFOA has the potential to be a persistent PFAS pollutant since it never degrades further given the appropriate environmental circumstances, either biotically or abiotically. Longer chain PFAS are effectively removed from water using traditional physicochemical techniques like membrane filtration, ion exchange, and activated carbon adsorption. This is because the majority of PFAS are resistant to conventional water treatment methods like oxidation, coagulation/flocculation, filtration, and metabolic degradation. However, these methods are not cost-effective or efficient when it comes to lower amounts of PFAS in surface water. It is also important to comprehend the basic principles of PFAS behavior in water before developing suitable treatment strategies. These include but are not limited to, the per- and Poly-Fluoroalkyl Substances' (PFAS) chemical makeup in water (including their hydrophobicity, functional groups, and other characteristics), possible pollution sources, PFAS persistence and bioaccumulation, and potential health hazards. Immune effects, cancer, and metabolic effects are mains causes of PFAS exposure on human health [6]. Figure 1 shows the typical structure of PFAS molecule.
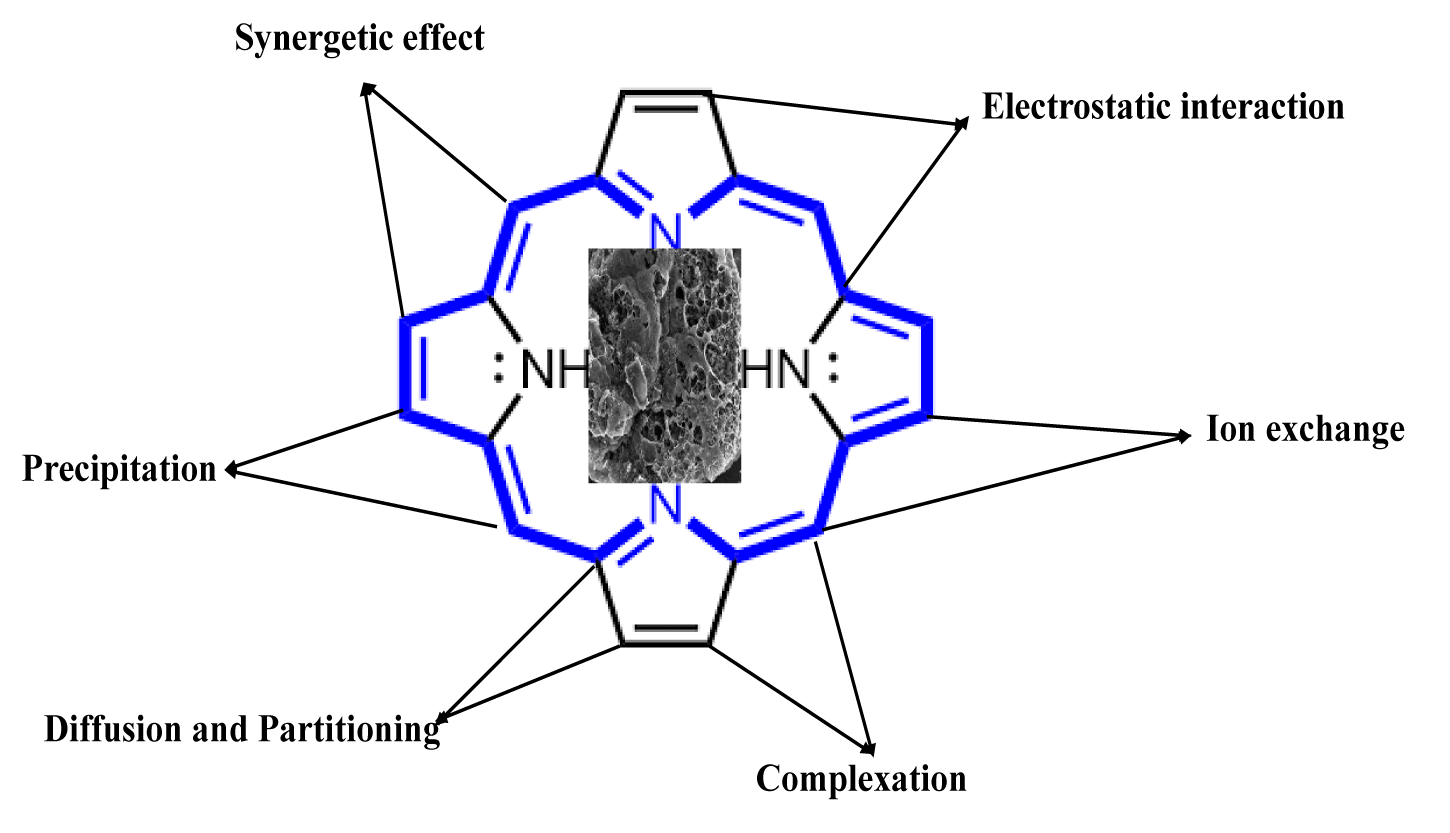
Alternatively, biochar can be used to remove these persistent chemicals [7,8]. The biochar-specific chemical properties such as large surface area, well distribution of pore size and micropores, rich oxygen-containing functional groups, high cation exchange capacity, and C and N content play an important role during the adsorption process. These properties can be easily tuned during biochar production, formation, and modification. Biochar employs various adsorption mechanisms such as electrostatic attraction, pore fillings, diffusion-partitioning, hydrophobic interaction, Ion exchange, surface complexation etc [9,10]. As compared to raw biochar which are directly produced from waste biomass, the modified engineered char showed better adsorption capacity. More importantly, it is vital to understand the nature of adsorbate which can easily adsorb on adsorbent (biochar surface). Secondly, we can easily modify biochar surfaces and inherent properties during the production process. It is widely reported that oxygen-containing functional groups such as carbonyl (-CO) and hydroxy groups (-OH), can enhance adsorption capacity for positive ions adsorbate (contaminant). Similarly, introducing metal and metal oxides on the biochar surface can improve the affinity of biochar to anion nature contaminants [10,11]. However, in the case of PFAS, the unique nature of compounds and persistent behavior, the adsorption process is challenging. Scattered literature reports support that high C and N content along with high surface area biochar can easily contribute to remove PFAS via the adsorption process. The primary mechanism to regulate C and N is the selective carbonization process, which is affected by the Maillard reaction [12,13]. Figure 2 shows the typical mechanism involved during the PFAS adsorption process.
The abundance of protein-containing waste such as dairy manure, sewage sludge, brewery’s grain wastes, food waste, yard wastes, and chlorophyll can be N precursors during the biochar preparation process [10,14,15]. The utilization of these massive wastes hits two birds with one stone such as waste management and creating value-added products such as N-doped chars which can be used for environmental applications via adsorption, catalysis, etc. Modulating the N ratio during the co-carbonization process helped to fix N in the char structure. Various synthesis processes including pyrolysis, hydrothermal carbonization, microwave pyrolysis, and activation are used to create N-doped chars. The mass production of N-rich biochar from these wastes can be used directly in waste streams to remove PFAS. However, each process and technology have its drawbacks and challenges. Several factors affect the engineering biochar production process and adsorption process. We need to consider all these factors and future research should focus on it.
Concluding Remarks
An enticing tactic for PFAS elimination is the production of biochar from renewable resources. Low-cost, readily available agricultural wastes can serve as sustainable resources for the production of tailored biochars. High surface area chars with high C and N contents are effective at removing PFAS from wastewater and other sources of contamination. Nonetheless, PFAS mitigation in the environment is a recent hot topic, and we must quickly solve socioeconomic issues. Future studies should concentrate on creating biochar from a variety of adaptable wastes and assessing how well those designed chars remove per- and Poly-Fluoroalkyl Substances (PFAS) from sources of contamination.
References
- Zhang D, He Q, Wang M, Zhang W, Liang Y. Sorption of Perfluoroalkylated Substances (PFASs) onto granular activated carbon and biochar. Environ Technol. 2021 May;42(12):1798-1809. doi: 10.1080/09593330.2019.1680744. Epub 2019 Oct 30. PMID: 31625466.
- Pilli S, Pandey AK, Pandey V, Pandey K, Muddam T, Thirunagari BK, Thota ST, Varjani S, Tyagi RD. Detection and removal of poly and perfluoroalkyl polluting substances for sustainable environment. J Environ Manage. 2021 Nov 1;297:113336. doi: 10.1016/j.jenvman.2021.113336. Epub 2021 Jul 26. PMID: 34325368.
- Jessica MS, Peng S, Ray JR. Novel perfluorooctanesulfonate-imprinted polymer immobilized on spent coffee grounds biochar for selective removal of perfluoroalkyl acids in synthetic wastewater. ACS ES&T Engineering. 2023;3(4): 520-532. doi: 10.1021/acsestengg.2c00336.
- Fronchetti GL, Reza T. Fundamental insight on how carbon chain length affects per-and polyfluoroalkyl substances adsorption onto hydrophobic deep eutectic solvents. Journal of Molecular Liquids. 2024;398:124238. doi: 10.1016/j.molliq.2024.124238.
- Jha G, Kankarla V, McLennon E, Pal S, Sihi D, Dari B, Diaz D, Nocco M. Per- and Polyfluoroalkyl Substances (PFAS) in Integrated Crop-Livestock Systems: Environmental Exposure and Human Health Risks. Int J Environ Res Public Health. 2021 Nov 28;18(23):12550. doi: 10.3390/ijerph182312550. PMID: 34886275; PMCID: PMC8657007.
- Pizzurro DM, Seeley M, Kerper LE, Beck BD. Interspecies differences in Per-Fluoroalkyl Substances (PFAS) toxicokinetics and application to health-based criteria. Regul Toxicol Pharmacol. 2019 Aug;106:239-250. doi: 10.1016/j.yrtph.2019.05.008. Epub 2019 May 9. PMID: 31078680.
- Barquilha CER, Braga MCB. Adsorption of organic and inorganic pollutants onto biochars: Challenges, operating conditions, and mechanisms. Bioresource Technology Reports. 2021;15:100728. doi: 10.1016/j.biteb.2021.100728.
- Margarida SS, Gonçalves M, Brito B, Nobre C. Waste-derived chars: A Comprehensive review. In Waste. 2024;2(3):218-239. doi: 10.3390/waste2030013.
- Kalidas M, Haghighi MS, Pelaez-Samaniego MR, Sierra-Jimenez V, Garcia-Perez M. Production and applications of N-doped carbons from bioresources: A review. Catalysis Today. 2023;423:114248. doi: 10.1016/j.cattod.2023.114248.
- Kalidas M, Mullen CA, Sarker MI, Haghighi MS, Garcia-Perez M. Production of N-Mg doped biochars for phosphate adsorption from renewable sources. Biomass and Bioenergy. 2024;185:107221. doi: 10.1016/j.biombioe.2024.107221.
- Li R, Wang JJ, Zhou B, Awasthi MK, Ali A, Zhang Z, Lahori AH, Mahar A. Recovery of phosphate from aqueous solution by magnesium oxide decorated magnetic biochar and its potential as phosphate-based fertilizer substitute. Bioresour Technol. 2016 Sep;215:209-214. doi: 10.1016/j.biortech.2016.02.125. Epub 2016 Mar 4. PMID: 26995322.
- Kalidas M, Garcia-Perez M. Understanding organic N interactions with cellulose during pyrolysis to produce N-doped carbonaceous materials. Journal of Analytical and Applied Pyrolysis. 2023;169:105791. doi: 10.1016/j.jaap.2022.105791.
- Krebsbach S, He J, Adhikari S, Olshansky Y, Feyzbar F, Davis LC, Oh TS, Wang D. Mechanistic understanding of Perfluorooctane Sulfonate (PFOS) sorption by biochars. Chemosphere. 2023 Jul;330:138661. doi: 10.1016/j.chemosphere.2023.138661. Epub 2023 Apr 10. PMID: 37044140.
- Wenran G, Lin Z, Chen H, Yan S, Huang Y, Hu X, Zhang S. A review on N-doped biochar for enhanced water treatment and emerging applications. Fuel Processing Technology. 2022;237:107468. doi: 10.1016/j.fuproc.2022.107468.
- Hanieh B, Papari S, Moreside E, Berruti F. High-temperature pyrolysis for elimination of per-and Poly-Fluoroalkyl Substances (PFAS) from biosolids. Processes. 2022;10(11):2187. doi: 10.3390/pr10112187.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.