Medicine Group. 2024 September 11;5(9):1089-1103. doi: 10.37871/jbres1994.
Reduced Number of Oligodendrocytes in the Cingulum in Schizophrenia: A Design-Based Stereology Study
Andrea Schmitt1-3*#, Katharina Ammann4#, Alisa Vollhardt4, Thomas Schneider-Axmann1, Florian J. Raabe1,3, Vladislav Yakimov1, Helmut Heinsen5,6, Patrick R. Hof7, Peter Falkai1,3# and Christoph Schmitz4#
2Laboratory of Neuroscience (LIM27), Institute of Psychiatry, University of São Paulo, São Paulo 05403-903, Brazil
3Max Planck Institute of Psychiatry, 80804 Munich, Germany
4Chair of Neuroanatomy, Institute of Anatomy, Faculty of Medicine, Institute of Anatomy, Ludwig Maximilian University (LMU) Munich, 80336 Munich, Germany
5Morphological Brain Research Unit, Department of Psychiatry, University of Würzburg, 97080 Würzburg, Germany
6Institute of Forensic Pathology, University of Würzburg, 97078 Würzburg, Germany
7Nash Family Department of Neuroscience, Friedman Brain Institute, and Center for Discovery and Innovation, Icahn School of Medicine at Mount Sinai, New York, NY 10029-6574, USA
#These authors contributed equally to this work and share first and last authorship
- Schizophrenia
- Oligodendrocytes
- Cingulum
- White matter
- Postmortem
- Stereology
Abstract
Schizophrenia is a severe mental disorder with a neurodevelopmental origin whose pathophysiological processes are poorly understood. Magnetic resonance imaging studies have shown structural alterations, such as volume loss in the temporal lobe, including the hippocampus and amygdala, thalamus, and frontal and temporal cortex, while other studies have found reduced oligodendrocyte numbers in the hippocampal CA4 subregion and dorsolateral prefrontal cortex. The decreased number of oligodendrocytes in CA4 was reported to be related to cognitive deficits. The hippocampus and other parts of the limbic system are connected to the white matter of the cingulum bundle, but it had remained unknown whether schizophrenia affects the number of oligodendrocytes also in the cingulum.
In a post mortem study, we applied a design-based stereological approach to investigate the number and density of oligodendrocytes in gallocyanin-stained serial sections in the cingulum of the left and right hemispheres in 12 brains from male donors with schizophrenia and 11 brains from age- and sex-matched controls without mental illness. In addition, we evaluated whether oligodendrocyte numbers and densities in the cingulum correlated with the corresponding data in the hippocampal CA4 subregion of the same brains.
In schizophrenia, both the mean volume of the left cingulum (difference, -12.7%) and the mean total oligodendrocyte number in the left cingulum (difference, -19.9%) were statistically significantly lower than in healthy controls (p = 0.049 and p = 0.037). No correlations were found for covariates (age, post mortem interval, fixation time). The individual oligodendrocyte numbers in the right cingulum correlated with those in the right hippocampal CA4, and the individual oligodendrocyte densities in the left cingulum correlated with that in the left hippocampal CA4.
This post mortem study shows, for the first time, a statistically significant reduction in the mean volume of and the mean oligodendrocyte number in the left cingulum of patients with schizophrenia. Our results support findings of a reduction of oligodendrocytes in limbic regions in schizophrenia, reinforcing the hypothesis that deficits in myelination and trophic support of long projecting axons play a role in the functional disconnectivity of the hippocampus and prefrontal cortex in schizophrenia..
Introduction
Schizophrenia (SZ) is a severe mental disorder with a worldwide prevalence of about 1% [1]. The first acute episode of SZ usually manifests itself between the ages of 18 and 35 and is often already noticeable during school or vocational training because it causes a drop in performance and social withdrawal [2]. About half of the patients experience incomplete remission of symptoms, with residual negative symptoms (e.g., avolition, apathy, lack of drive and affective flattening) and cognitive deficits (e.g., attention, working memory, executive function, episodic memory and social cognition) [3-5]. These long-lasting symptoms cause difficulties in coping with social and professional life and result in unemployment or limited earning capacity in about 70% of patients [6]. According to the Global Burden of Disease Study, SZ is one of the 25 diseases worldwide with the highest number of disability-impaired life years in the age group of 25 to 49 years [7]. At the same time, SZ causes high costs to society and health care systems because of its early onset and often chronic course [8].
The pathophysiological mechanisms of SZ are poorly understood. The neurodevelopmental hypothesis, which is based on genome-wide association and epidemiological studies, proposes that SZ is associated with interacting genetic and environmental risk factors, such as maternal infection, obstetric and birth complications, and childhood trauma, which influence connectivity in neuronal circuits during vulnerable brain developmental periods [9]. These risk factors induce disturbances of macro- and microconnectivity in neuronal networks in the gray and white matter. Meta-analyses of Magnetic Resonance Imaging (MRI) studies have shown structural alterations, such as volume reductions in the temporal lobe, including the hippocampus and amygdala, thalamus, prefrontal including the anterior cingulate cortex and cingulum [10-12]. Voxel-Based Morphometry (VBM) and Diffusion Tensor Imaging (DTI) are two distinct methodologies for brain structural investigation, suitable for detecting different aspects of white matter alterations in neuropsychiatric disorders. In a meta-analysis of VBM and DTI studies focusing on white matter alterations in patients with SZ compared to healthy controls, VBM studies showed volume reductions in several regions including the bilateral cingulum, anterior commissure, corpus callosum, fornix, internal capsule, right anterior segment of Arcuate Fasciculus (AF), left cortico-ponto-cerebellum tract, right superior cerebellar penduculus, bilateral AF, bilateral corticospinal tract, bilateral Inferior Fronto-Occipital Fasciculus (IFOF), bilateral Inferior Longitudinal Fasciculus (ILF), bilateral inferior cerebellar penduculus, bilateral optic radiation, bilateral posterior segment of AF, bilateral first part of Superior Longitudinal Fasciculus (SLF1), bilateral central part of SLF (SLF2), bilateral final part of SFL (SLF3) and the bilateral Uncinate Fasciculus (UF) [13]. Furthermore, DTI studies showed white matter microstructure reductions in the bilateral cingulum as well, together with reductions in the anterior commissure, corpus callosum, fornix, internal capsule, left anterior segment of AF, left long segment of AF, bilateral AF, bilateral cingulum, bilateral cortico-ponto-cerebellum tract, bilateral cortico-spinal tract, bilateral IFOF, bilateral ILF, bilateral inferior cerebellar penduculus, bilateral optic radiation, bilateral posterior segment of AF, bilateral SLF1, bilateral SFL2, bilateral SLF3, bilateral superior cerebellar penduculus and the bilateral uncinate fasciculus [13]. The conjunction analysis from VBM and DTI studies found no overlapping clusters on the same white matter tract, indicating that DTI and VBM actually detect different aspects of white matter alterations [13]. Diffusion Tensor Imaging (DTI) studies revealed impaired white matter integrity, such as lower Fractional Anisotropy (FA) in white matter fiber tracts connecting these brain regions, and these findings point to deficits in myelination of fiber tracts [14,15]. A meta-analysis of DTI studies across 2937 individuals with four psychiatric disorders and healthy controls identified lower FA in patients with SZ in the anterior corona radiata, corpus callosum, fornix and cingulum [16]. A recent meta-analysis compared SZ and bipolar patients and showed disorder-specific decreased FA in the left cingulum in SZ [17].
The cingulum is a white matter bundle located between the cingulate cortex and the dorsal surface of the corpus callosum that reaches up to the temporal lobe [18]. It connects the anterior thalamic nucleus, cingulate gyrus and parahippocampal region, and plays a role in emotional and social cognition, both of which are impaired in SZ [19-21]. In SZ, speed of processing, attention, executive function and social cognition were found to correlate with disrupted white matter integrity in the cingulum bundle, particularly in the left hemisphere [22-26]. Moreover, patients with delusions of control have lower FA in the cingulum than patients without such delusions [27]. A 7 T magnetization transfer study confirmed aberrant myelination of the cingulum in patients with such delusions (Schneiderian delusions) [28]. Lower functional connectivity during rest and working memory tasks was detected in the cingulum around the cingulate cortex and hippocampus of patients with SZ and correlated with cognitive performance [29].
The cingulum contains many oligodendrocytes, which generate myelin sheaths around neuronal axons. They provide trophic support to the axons [30] and improve axonal conduction velocity [31], thus contributing to proper functional connectivity. Within the limbic system, design-based stereological studies revealed reduced mean oligodendrocyte numbers in the hippocampal CA4 subregion in SZ [32,33], a finding that was replicated in an independent sample [34]. The reduced oligodendrocyte number in the CA4 subregion was more pronounced in patients with definite cognitive deficits [35,36]. In SZ, the number of oligodendrocytes was also reported to be decreased in the white matter directly subjacent to the Dorsolateral Prefrontal Cortex (DLPFC) [37]. In at least a subgroup of patients with SZ, the number of oligodendrocytes in the CA4 subregion and DLPFC may be lower because of deficits in oligodendrocyte maturation and differentiation of oligodendrocyte precursor cells, and these changes may be related to cognitive deficits [38]. However, a more widespread reduction of oligodendrocytes in limbic regions may underlie the impaired connectivity in SZ, and to date, it remains unknown whether patients with SZ have a loss of oligodendrocytes also in the white matter of the cingulum.
In view of these changes in white matter integrity in the cingulum and its association with cognitive deficits predominantly in the left hemisphere, we tested (using design-based stereology) the hypothesis that mean oligodendrocyte numbers and densities are decreased in the cingulum in SZ, and, if so, that the reductions are more pronounced in the left hemisphere.
Materials and Methods
Post mortem brains
The autopsied brain tissue examined in this study was provided by the Heinsen Collection (University of Wuerzburg, Wuerzburg, Germany), which is managed by H.H. The further use of the brains for scientific purposes was approved by the ethics committee of the University of Wuerzburg.
This study evaluated both hemispheres of 12 post mortem brains from multi-episode male patients with SZ and poor outcome (participant codes, S1-S12; age range, 22 to 63 years; mean age, 50.5 years; mean Post Mortem Interval [PMI], 38.1 hours). As a control group, we used 11 post mortem brains of age-matched men who had not had a mental disorder (participant codes, C2-C13; age range, 36 to 65 years; mean age, 54.5 years; mean PMI, 23.5 hours). Participants’ clinical characteristics, i.e., age at death, age at disease onset, cause of death, PMI, fixation duration and clinical diagnosis (for patients with SZ only) are listed in table 1. All patients with SZ fulfilled the diagnostic criteria of the Diagnostic Statistical Manual, 4th revision, and International Statistical Classification of Diseases and Related Health Problems, 10th revision. They had been treated in German clinical facilities (state psychiatric hospitals and local district hospitals) and had all received treatment with "typical" antipsychotics for decades. However, we were unable to calculate lifetime medication exposure because most of the patients were not hospitalized for the entire duration of their disease, so we did not have documentation on their entire medication history. Both groups had a comparable, white European ethnic background.
| Table 1: Clinical characteristics of male patients with schizophrenia and controls. | |||||||
| No. | A | O | Cause of death | PMI | Fix | Diagnosis | |
| y | y | h | d | DSM-IV | ICD-10 | ||
| S1 | 22 | 19 | Suicide by jumping from a high building | 88 | 130 | 295.30 | F20.00 |
| S2 | 36 | 28 | Suicide by strangulation | < 72 | 115 | 295.30 | F20.00 |
| S3 | 46 | 24 | Systemic hypothermia | < 24 | 327 | 295.30 | F20.01 |
| S4 | 50 | 17 | Peritonitis | < 24 | 203 | 295.30 | F20.00 |
| S5 | 50 | 22 | Suicide by strangulation | 18 | 170 | 295.30 | F20.00 |
| S6 | 51 | 17 | Septicemia | 33 | 127 | 295.60 | F20.50 |
| S7 | 54 | 20 | Septicemia | 27 | 250 | 295.60 | F20.50 |
| S8 | 55 | 22 | Right-sided heart failure | 25 | 84 | 295.30 | F20.00 |
| S9 | 57 | 37 | Septicemia | 76 | 163 | 295.30 | F20.00 |
| S10 | 60 | 24 | Pulmonary embolism | < 48 | 311 | 295.30 | F20.01 |
| S11 | 62 | 19 | Aspiration | 7 | 171 | 295.30 | F20.00 |
| S12 | 63 | 22 | Acute myocardial infarction | 15 | 338 | 295.60 | F20.50 |
| C2 | 36 | - | Gunshot | 24 | 143 | - | - |
| C3 | 47 | - | Acute myocardial infarction | < 24 | 133 | - | - |
| C5 | 50 | - | Avalanche accident | 23 | 498 | - | - |
| C6 | 51 | - | Septicemia | 7 | 285 | - | - |
| C7 | 54 | - | Acute myocardial infarction | 18 | 168 | - | - |
| C8 | 56 | - | Acute myocardial infarction | 60 | 3570 | - | - |
| C9 | 58 | - | Acute myocardial infarction | 28 | 126 | - | - |
| C10 | 60 | - | Gastrointestinal hemorrhage | 18 | 101 | - | - |
| C11 | 60 | - | Gastrointestinal hemorrhage | 27 | 302 | - | - |
| C12 | 62 | - | Acute myocardial infarction | < 24 | 3696 | - | - |
| C13 | 65 | - | Bronchopneumonia | 6 | 2289 | - | - |
| S: Patient with schizophrenia; C: Age-matched control; A: Age at death; O: Age at disease onset; PMI: Postmortem Interval (i.e., the time between death and autopsy); Fix: Fixation time. | |||||||
Exclusion criteria for both groups were neurological diseases that would have required treatment or resulted in cognitive impairment (e.g., apoplexy with aphasia), known history of seizure disorders (e.g., epilepsy), severe head injury with loss of consciousness, diabetes mellitus with poor glycemic control (free plasma glucose > 200 mg/dl), and alcohol and drug intoxications. On the other hand, head trauma could not be excluded in participants who had died by suicide. Brains were examined for neuropathologies to rule out tumors, infarcts, heterotopias, evidence of autolysis, staining artifacts and gliosis.
Tissue processing
All tissues were fixed and processed in the same way at the Morphological Brain Research Unit (University of Wuerzburg, Wuerzburg, Germany). For detailed information, Kreczmanski P, et al. [39], who investigated the same post mortem brains in previous studies. To prepare the histological specimens, the brainstem, including the cerebellum, was first separated at the level of the rostral pons, and the hemispheres were separated medio-sagittally. The separated hemispheres were then fixed in 4% formalin solution for at least three months. After being pretreated with glycerol-dimethylsulfoxidformalin and embedded in gelatin, the hemispheres were deep frozen in isopentane at -60 °C. Series of frontal/coronal sections with a thickness of 600 to 700 µm were prepared with a cryomicrotome (Jung, Nussloch, Germany; Heinsen H. [40] for further details). One control brain (participant C7) was embedded in celloidin [41] and sectioned into a series of 440-μm thick coronal sections with a sled microtome (Polycut, Cambridge Instruments, UK). Every second to third section of each brain was stained with gallocyanin, a Nissl stain [40], and the remaining sections were placed in formalin and stored for further use. One section of the brain of each participant aged over 40 years was not stained with gallocyanin and was processed through the central parts of the entorhinal and transentorhinal cortex by the Gallyas method to detect neurofibrillary changes [42]. In addition, each section was coded and tumors, infarcts, heterotopia, signs of autolysis, staining artifacts and gliosis were excluded. This procedure took place under the same conditions for all brains (except for sections from control group participant C7, which were embedded in celloidin instead of gelatin). The mean (SD) numbers of whole-brain sections used were as follows: patients with SZ, 20.75 (0.92); controls, 21.55 (1.62). The whole cingulum was analyzed in both hemispheres (Figure 1).
Design-based stereological analyses
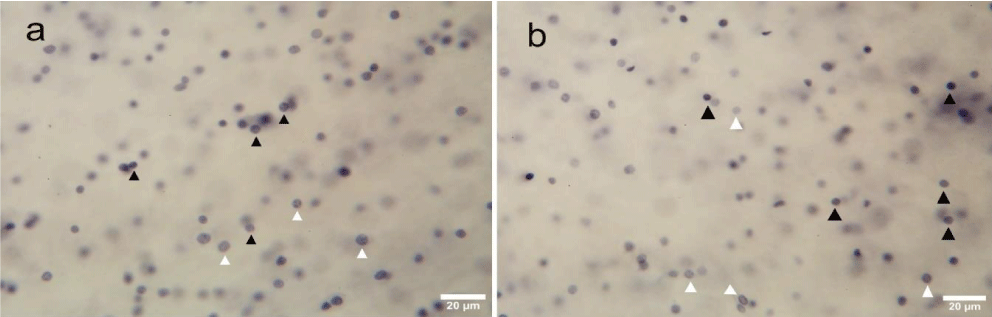
We used histological and morphological criteria to identify oligodendrocytes as small, dark cells [34] in photomicrographs of histologically stained cells using a 40x objective (Figure 2). Mature oligodendroglia showed no staining in the cytoplasm and no nucleolus but did show pronounced nuclear staining with scattered chromatin. We also assessed immature oligodendrocytes which are larger and more pale than mature ones [43]. Oligodendrocytes could be distinguished from astrocytes because the latter show less dense staining [34] (Figure 2(a,b)). All stereological examinations were performed with a light microscope (Zeiss Axio Imager.M2 Microscope; Carl Zeiss Microscopy GmbH, Jena, Germany); Zeiss Objectives (1,25×; 40×); a stage controller; and software (Stereo Investigator 2018.2.2 64 bit; MBF Bioscience, Williston, VT, USA). The delineation of the cingulum (Figure 1) was performed using a 1.25x objective. The volume of the cingulum was determined according to the Cavalieri principle [44], and the total number of oligodendrocytes was determined using the optical fractionator method [45]. We used the prediction methods described by Schmitz [46] and Schmitz and Hof [47] to calculate the predicted coefficient of error in the estimates of total numbers of oligodendrocytes. In all examinations of the sections, the investigator was blinded to participant group. The details of the stereological procedures for counting oligodendrocytes are listed in table 2.
| Table 2: Details of the stereological procedures used for oligodendrocyte counting. | |
| Objective used | 40x |
| B µm2 | 900 |
| H µm | 20 |
| D µm | 925 |
| ∑CS | 487 |
| ∑Q- | 627 |
| Cepred | 0.041 |
| B and H base and height of the unbiased virtual counting spaces; D distance between the unbiased virtual counting spaces in mutually orthogonal directions x and y; ∑CS mean sum of unbiased virtual counting spaces used in one hemisphere of one participant; ∑Q- mean number of counted oligodendrocytes in one hemisphere of one participant; CEpred, mean predicted coefficient of error of estimated cell numbers. | |
Statistical analyses
The dependent variables were cingulate volume and the number and density of oligodendrocytes in both hemispheres. The independent factor was diagnosis (patients with SZ, controls), and the intervening variables were age, PMI and fixation time. All Spearman correlations between dependent variables and age, PMI and fixation time were non-significantly different from zero, so they were not considered as covariates in the main analysis. To adequately test the hypothesis of a hemisphere-specific deficit, analyses of variance with factor diagnosis were calculated for all dependent variables separately for the left and right hemisphere. Additionally, repeated measures analyses of variance were calculated with the between-subject factor diagnosis and within-subject factor hemisphere. We also investigated whether the volume and oligodendrocyte numbers and densities in the cingulum were correlated with the corresponding values in the hippocampal subregion CA4 of the same sample [34]. The significance level of α = 0.05 was used for all analyses. All tests were two-tailed. SPSS version 28 (IBM, Armonk, NY, USA) was used for statistical analyses.
Results
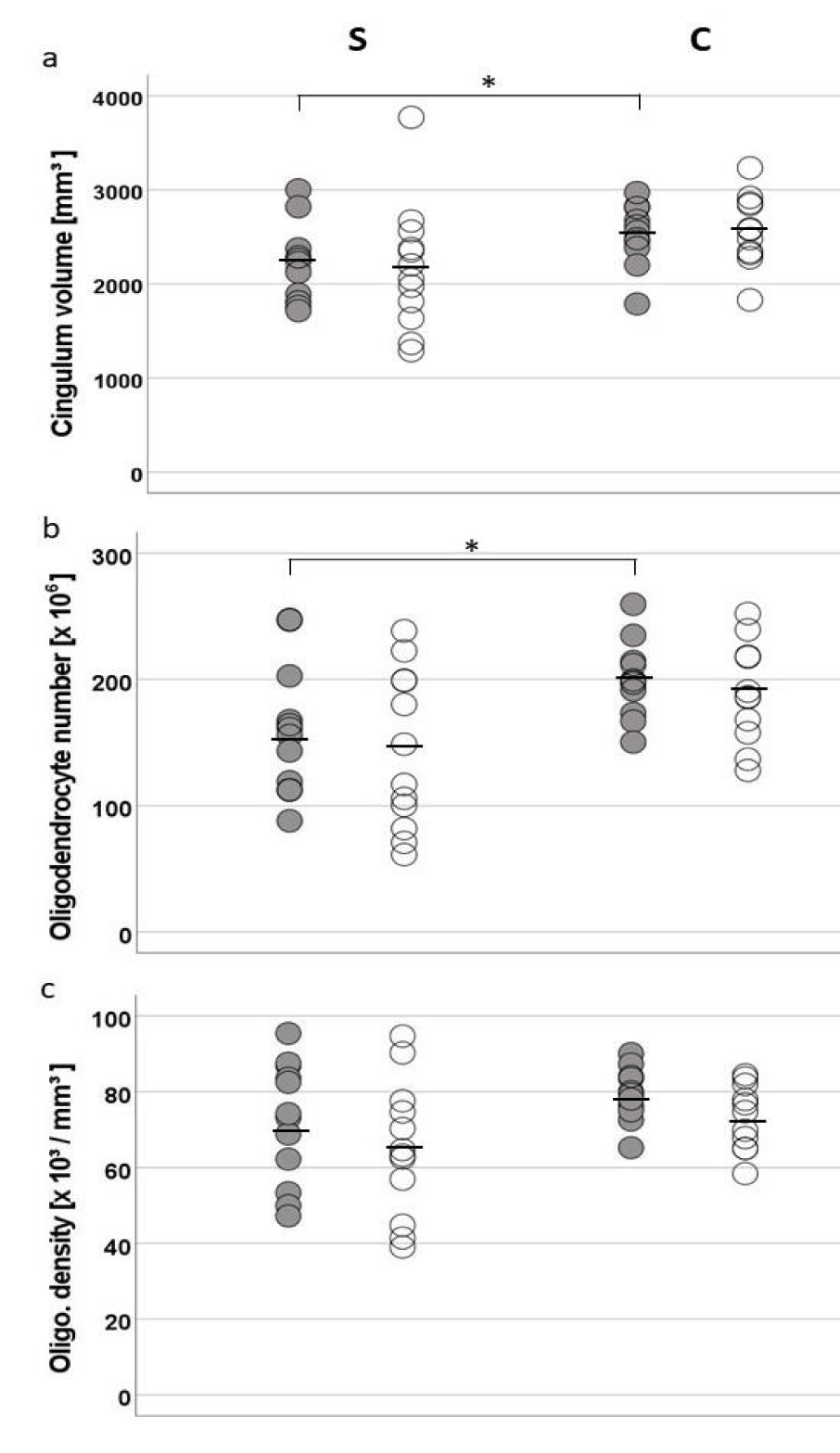
The mean cingulum volume was statistically significantly lower in patients with SZ than in controls in the left hemisphere (mean [SD]: SZ, 2205 [399] mm³; controls, 2525 [328] mm³; F(1, 21) = 4.382; p = 0.049) but not in the right hemisphere (mean [SD]; SZ, 2173 [669] mm³; controls, 2571 [384] mm³; F(1, 21) = 2.98; p = 0.099) (Figure 3a).
The mean total number of oligodendrocytes in the cingulum was lower in SZ patients (mean [SD]; left hemisphere, 160.12 [51.21] million; right hemisphere, 143.81 [62.33] million) than in controls (mean [SD]; left hemisphere, 199.83 [30.77] million; right hemisphere, 189.21 [40.13] million). Oligodendrocyte numbers were statistically significantly lower in the left hemisphere (-19.9%; F(1, 21) = 4.96; p = 0.037) but not in the right hemisphere (-24%; F(1, 21) = 4.222; p = 0.053) (Figure 3b). The mean number of oligodendrocytes did not differ between the two hemispheres (F(1, 21) = 2.89, p = 0.104). We observed an effect of the factor hemisphere on oligodendrocyte densities in that mean densities were larger in the left hemisphere (mean [SD]; SZ,72072 [15986] cells/mm³; controls, 79241 [7031] cells/mm³) than in the right hemisphere (mean [SD]; SZ, 65024 [17884] cells/mm³; controls, 73273 [8623] cells/mm³; F(1, 21) = 6.552; p = 0.018). However, we found no statistically significant differences in mean oligodendrocyte densities between the patients with SZ and controls in the left (F(1, 21) = 1.874, p = 0.186) and right (F(1, 21) = 1.925, p = 0.180) hemispheres (Figure 3c). The results from the brain of control participant C7 (which was embedded in celloidin) did not systematically deviate from those of the gelatin-embedded brains.
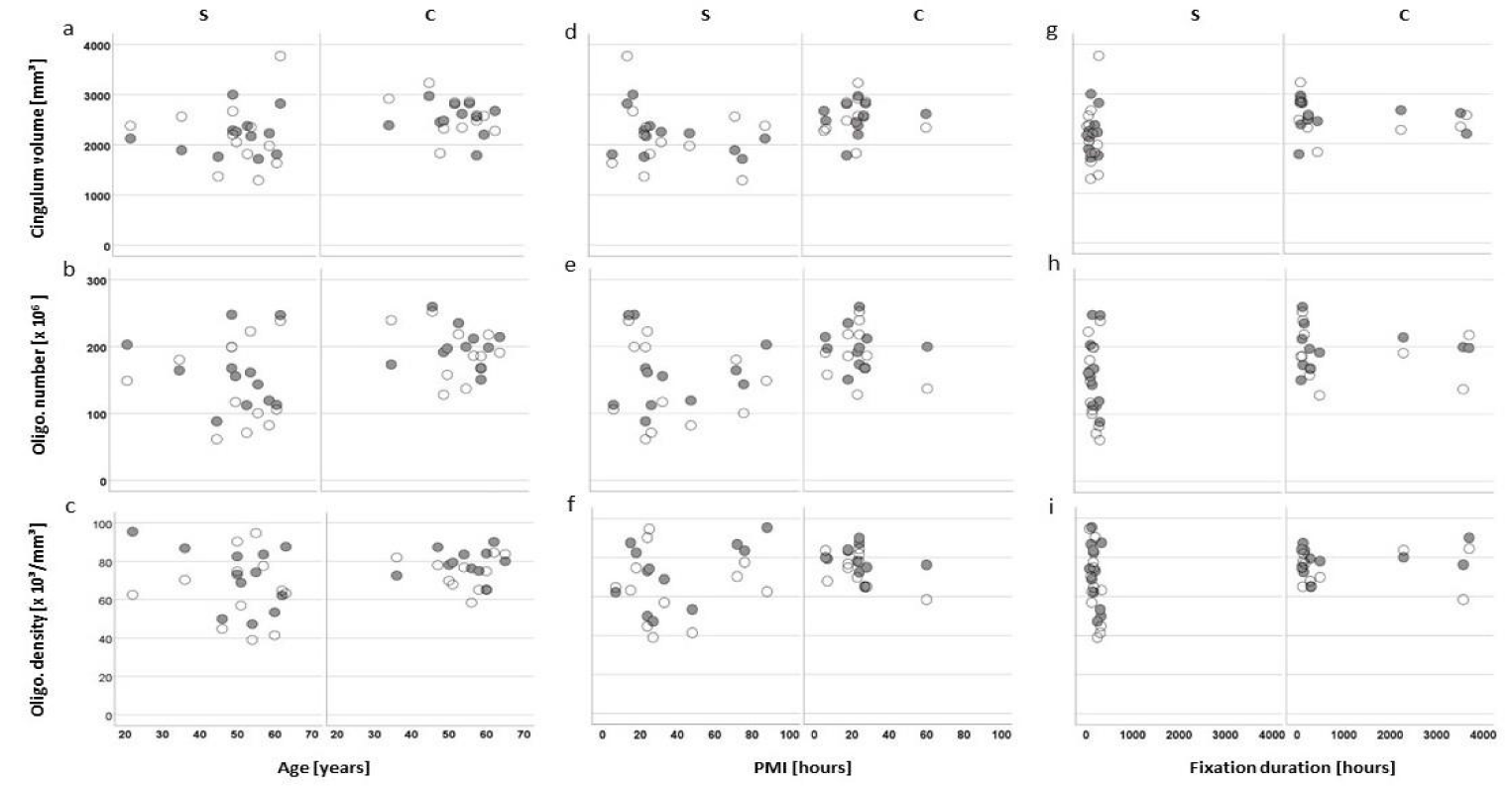
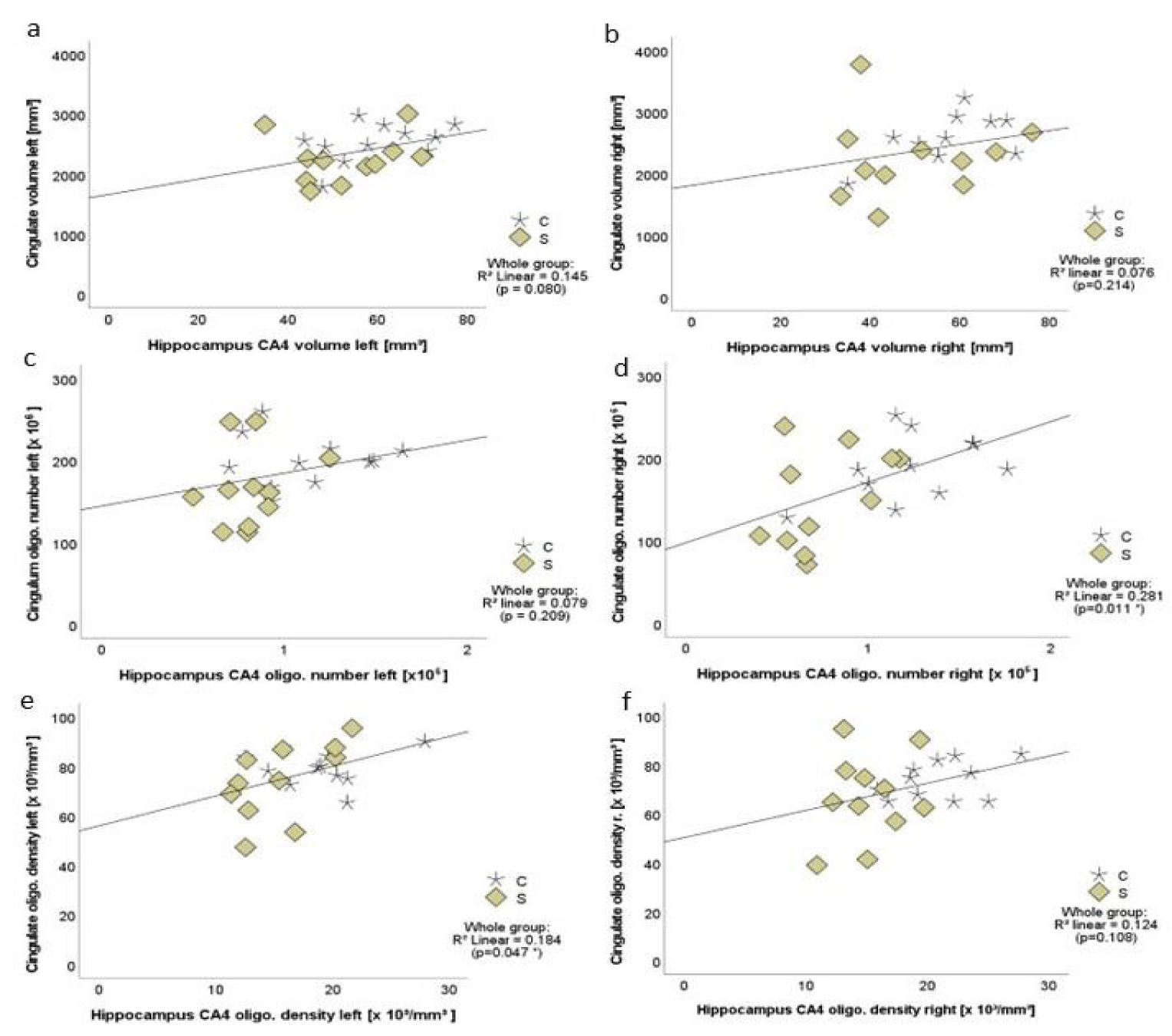
In patients with SZ and controls, we found no statistically significant correlations of any dependent variable with age (all p < 0.346 , p > 0.257), PMI (all ρ < 0.446, p > 0.169) or duration of fixation (all p < 0.503, p > 0.095) (Figure 4(a-i)). In the whole sample, we found a statistically significant correlation between the individual oligodendrocyte numbers in the right cingulum and right CA4 (r = 0.530, p = 0.011; figure 5(a-d)) and between the individual oligodendrocyte densities in the left cingulum and left CA4 (r = 0.429, p = 0.047; figure 5(e,f)).
Discussion
The present study detected a statistically significantly lower mean volume of the left cingulum and a statistically significant reduction in the mean oligodendrocyte number in the left cingulum in male patients with SZ compared with age- and sex-matched controls. In contrast, mean oligodendrocyte densities were not different between the groups, which may be due to a bias in cell density measurements caused by tissue shrinkage after fixation and staining procedures. This issue is addressed by design-based stereological methods [44,47,48]. In principle, cell numbers can change without a corresponding change in the density of the same cells [49] and cell density measurements cannot serve as a surrogate of cell number measurements because cell densities may change without alterations in the number of the same cells [47].
Our results are in agreement with reported reductions in mean oligodendrocyte numbers in other brain regions, such as the CA4 subregion of the hippocampus, in patients with SZ in the same sample [34] and in an independent cohort [32,33], as well as in the DLPFC of another series [37]. In a previous post mortem analysis, we found a positive relationship between the oligodendrocyte number in the hippocampal subregions CA4 and CA2/3 and the volume of the hypothalamus as predictors for a diagnosis of SZ [50]. Our results support the notion that an oligodendrocyte pathology may contribute to cognitive deficits in limbic circuits in SZ. We have investigated the volume and total number of oligodendrocytes in the gray matter of the hippocampus CA4 within the same brains and found no volume reduction, whereas in the left cingulum, a volume reduction of 12.7% on average was detected. The total number of oligodendrocytes was reduced in the CA4 subregion in the left hemisphere by averaged 28%, in the right hemisphere by averaged 39%, in the left cingulum by averaged 19.9% and in the right cingulum by averaged 24% (the reduction in the right cingulum was not statistically significant). Collectively these data indicate a larger effect of diagnosis on the volume and a slightly smaller effect on the total number of oligodendrocytes in the left cingulum in SZ. Furthermore, we found a statistically significant correlation between the individual total number of oligodendrocytes in the right cingulum and right CA4 and between the oligodendrocyte density in the left CA4 and left cingulum. Therefore, we hypothesize a more widespread process in the brain. A lateralization of brain abnormalities has often been described in SZ. We found a statistically significant reduction of the volume and total number of oligodendrocytes only in the left cingulum. This pronounced reduction in our multi-episode patients with poor outcome reflects the finding from a recent DTI study, that poor outcome patients had a specific reduction of fractional anisotropy in the left cingulum compared to good outcome patients [51].
Previously, we hypothesized that an oligodendrocyte deficit underlies the cognitive deficits in SZ [36,38]. An earlier study found that a reduced oligodendrocyte number in the hippocampus was associated with impaired cognition [35]. However, in the present study we did not have information on disturbed cognitive abilities of the patients with SZ. The cingulum is involved in emotional expression, attention, motivation and working memory [19,21,52], and all of these functions are impaired in SZ [3,20]. DTI studies found reduced FA (as a marker of white matter fiber density, axonal diameter and myelination) in the cingulum of patients with SZ [16,17]. Moreover, in SZ reduced performance in cognitive domains such as processing speed, attention, executive function and social cognition correlates with impaired white matter integrity and connectivity in the cingulum [22-27]. Previous DTI studies detected stronger findings in the left cingulum, and our post mortem results also point to significant changes in volume of the left cingulum and an oligodendrocyte pathology predominantly in the left hemisphere.
The present study is the first to detect an oligodendrocyte deficit in the white matter region of the cingulum in SZ. Oligodendrocytes play an important role in the myelination of neuronal axons and thus in neuronal network connectivity based on myelination [53,54]. Therefore our findings suggest a deficit in myelination and trophic support of long projecting axons in the limbic white matter and reinforce the hypothesis that the disconnectivity of multiple brain regions within neuronal networks based on oligodendrocyte pathology plays a major role in SZ. Myelination is an important process during postnatal brain development. White matter pathways develop in the early years of childhood in particular, but they continue to be myelinated into puberty and young adulthood. The late myelinating regions include the associative cortical areas such as the cingulate and prefrontal cortex [55]. A disturbed myelination process during this critical window of brain development may have implications for the onset of SZ in young adulthood [9].
Exposure to substantial stressors during adolescence may trigger a cascade of events that result in SZ in adulthood [56]. However, our post mortem study does not help to clarify these neurodevelopmental aspects because we investigated the brains from older adults, in whom the long-term disease process may have shown its effect. Animal studies have shown that juvenile social isolation, a stress paradigm during puberty, leads to SZ-related behavior deficits in prepulse inhibition of the acoustic startle response, deficits in working memory, decreased social exploration, altered oligodendrocyte morphology, reduced myelin thickness and decreased oligodendrocyte protein expression [57,58]. Genome-wide association studies revealed that Transcription Factor 4 (TCF4) is a risk gene for SZ and is related to cognitive deficits [59,60]. In double heterozygous Tcf4 oligodendrocyte lineage transcription factor 2 (Olig2) null mutant mice, Tcf4 was shown to be the preferred heterodimerization partner for Olig2 in oligodendrocytes and required for oligodendrocyte precursor cell differentiation. Double heterozygous Tcf4/Olig2 mice have deficits in oligodendrocyte maturation and display reduced numbers of oligodendrocytes, as shown in the spinal cord [61]. Moreover, implantation of human induced pluripotent stem cell-derived oligodendrocyte progenitor cells from patients with childhood-onset SZ into myelin-deficient shiverer mice had a negative effect on the myelination properties of those cells in the mouse chimeras [62]. It has been proposed that a deficit in differentiation and maturation of oligodendrocyte precursor cells is an underlying pathophysiology of cognitive deficits and impaired frontotemporal connectivity in SZ and that this deficit could be restored by pro-myelinating drugs and aerobic exercise [38].
One limitation of our study is that the results are restricted to male subjects. Male patients with SZ tend to develop the condition earlier than females. Additionally, male patients with SZ typically experience more severe psychotic symptoms and generally have poorer outcomes, including more pronounced negative symptoms and cognitive issues [63,64]. Another constrain is that our sample was relatively small limiting its power. Therefore, the results should be confirmed in an independent, larger sample. We cannot exclude effects of the disease process itself and long-term antipsychotic treatment in our patient group, but a stereological study of the parietal cortex of monkeys after treatment with typical and second-generation antipsychotics showed no significant reduction of the number of oligodendrocytes immunostained with the oligodendrocyte enzyme-related antibody 2′,3′-cyclic-nucleotide 3'-phosphodiesterase [65]. In the hippocampus of mice, treatment with typical antipsychotics increased the number of Olig2-expressing oligodendrocytes and activated oligodendrocyte precursor cells [66]. We identified oligodendrocytes by histological gallocyanin staining, but we did not confirm the results by immunohistochemical labeling because antibodies usually do not penetrate through thick tissue sections like those used in the present study [48]. Future immunohistochemical studies using antibodies for oligodendrocyte precursors (e.g., breast carcinoma amplified sequence 1 (BCAS1) [67] or mature oligodendrocytes) are necessary to conclude if a deficit in maturation and differentiation of oligodendrocyte precursors is the underlying cause of the described cell loss [68].
Conclusion
In summary, we found a lower mean volume and lower mean oligodendrocyte number in the left cingulum in male patients with SZ, but we did not find changes in mean oligodendrocyte density. Missing oligodendrocytes reduce the volume of the myelin sheaths, and may result in a reduction of the volume of the cingulum. Since the cingulum is smaller, the fewer oligodendrocytes are compressed in a smaller volume and, thus, the density of these cells is not changed. This context has already been shown in a study on neuron numbers and densities from animal brains [49]. A decreased oligodendrocyte number may indicate that mature oligodendrocytes are damaged or that differentiation of oligodendrocyte precursor cells is impaired; the latter can be clarified in a future study by immunohistochemical staining of oligodendrocyte precursor cells at the early myelination stage [38]. From magnetic resonance imaging studies it is known that SZ is associated with volume loss in brain regions such as the hippocampus, insula, anterior cingular cortex, gyrus frontalis medius, temporal cortex and thalamus [12,69-72], and with deficits in structural and functional connectivity in specific networks in SZ, comprising the default-mode network comprising the cingulate gyrus, the salience network comprising the dorsal anterior cingulate cortex oder the fronto-parietal network [73-75]. Therefore it is unlikely that an oligodendrocyte loss is limited to one specific brain region. Up to now, a loss of oligodendrocytes has been detected by design-based stereology in the gray matter of the hippocampus CA4 subregion, the dorsolateral prefrontal cortex and now for the first time in the white matter of the cingulum, supporting the hypothesis of a microstructural deficit in brain circuits in SZ [68]. Additionally, future studies should investigate limbic gray and white matter regions in SZ and assess other brain regions as other axonal pathways are likely to be comparably affected. Such studies may result in new insights that can lead to innovative treatment strategies that target remyelination of oligodendrocyte precursor cells and thus improve impaired connectivity in SZ.
Declaration of Interests
P.F. was co-editor of the German (DGPPN) schizophrenia treatment guidelines, co-author of the WFSBP schizophrenia treatment guidelines, and currently on advisory board of Boehringer Ingelheim and he was on advisory boards of Neuron Pharmaceutical, Abbott, Janssen, NeuraxPharm. He received speaker fees from Janssen, Lundbeck, Otsuka, Servier, and Richter. The other authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Author Contributions
Conceptualization, P.F., C.S., and A.S.; methodology, C.S., A.V., P.R.H., and K.A.; formal analysis, C.S. and T.S.-A.; investigation, K.A. and A.V.; resources, H.H.; data curation, T.S.-A.; writing and original draft preparation, A.S., K.A., P.F., P.R.H., F.J.R., and V.Y.; writing, review, and editing, A.S., P.F., F.J.R., V.Y., K.A., P.R.H., H.H., T.S.-A., and C.S.; visualization, K.A., A.V., and C.S..; supervision, C.S. and P.F. All authors have read and agreed to the published version of the manuscript.
Funding Sources
The study was supported by the Else Kröner-Fresenius Foundation for the Residency/PhD track “Translational Psychiatry” to A.S., F.J.R and P.F. and the International Max Planck Research School for Translational Psychiatry (IMPRS-TP) to A.S., F.J.R., V.Y., and P.F. and by the Munich Clinician Scientist Program (MCSP) of the Faculty of Medicine at LMU Munich to F.J.R.
Acknowledgment
The authors thank Jacquie Klesing, Board-certified Editor in the Life Sciences (ELS), for editing assistance with the manuscript. Jacquie Klesing received compensation for her work from the LMU University Hospital, Munich, Germany.
Declaration of Generative AI and AI-Assisted Technologies in the Writing Process
No generative AI and AI-assisted technologies have been used in the writing process.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available for ethical reasons.
References
- Jablensky A. Epidemiology of schizophrenia: the global burden of disease and disability. Eur Arch Psychiatry Clin Neurosci. 2000;250(6):274-85. doi: 10.1007/s004060070002. PMID: 11153962.
- Häfner H, Nowotny B, Löffler W, an der Heiden W, Maurer K. When and how does schizophrenia produce social deficits? Eur Arch Psychiatry Clin Neurosci. 1995;246(1):17-28. doi: 10.1007/BF02191811. PMID: 8773215.
- Nuechterlein KH, Green MF, Kern RS, Baade LE, Barch DM, Cohen JD, Essock S, Fenton WS, Frese FJ 3rd, Gold JM, Goldberg T, Heaton RK, Keefe RS, Kraemer H, Mesholam-Gately R, Seidman LJ, Stover E, Weinberger DR, Young AS, Zalcman S, Marder SR. The MATRICS Consensus Cognitive Battery, part 1: test selection, reliability, and validity. Am J Psychiatry. 2008 Feb;165(2):203-13. doi: 10.1176/appi.ajp.2007.07010042. Epub 2008 Jan 2. PMID: 18172019.
- Kahn RS, Keefe RS. Schizophrenia is a cognitive illness: time for a change in focus. JAMA Psychiatry. 2013 Oct;70(10):1107-12. doi: 10.1001/jamapsychiatry.2013.155. PMID: 23925787.
- Schmitt A, Maurus I, Falkai P. Treatment of negative symptoms in schizophrenia: a challenge for clinical research. Eur Arch Psychiatry Clin Neurosci. 2023 Apr;273(3):525-526. doi: 10.1007/s00406-023-01595-w. PMID: 36977788; PMCID: PMC10085889.
- Wolfram an der Heiden. Course and outcome, in Schizophrenia. Weinberger DR, editor. Blackwell Oxford; 2011. p.104-141.
- GBD 2019 Diseases and Injuries Collaborators. Global burden of 369 diseases and injuries in 204 countries and territories, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet. 2020 Oct 17;396(10258):1204-1222. doi: 10.1016/S0140-6736(20)30925-9. Erratum in: Lancet. 2020 Nov 14;396(10262):1562. doi: 10.1016/S0140-6736(20)32226-1. PMID: 33069326; PMCID: PMC7567026.
- Gustavsson A, Svensson M, Jacobi F, Allgulander C, Alonso J, Beghi E, Dodel R, Ekman M, Faravelli C, Fratiglioni L, Gannon B, Jones DH, Jennum P, Jordanova A, Jönsson L, Karampampa K, Knapp M, Kobelt G, Kurth T, Lieb R, Linde M, Ljungcrantz C, Maercker A, Melin B, Moscarelli M, Musayev A, Norwood F, Preisig M, Pugliatti M, Rehm J, Salvador-Carulla L, Schlehofer B, Simon R, Steinhausen HC, Stovner LJ, Vallat JM, Van den Bergh P, van Os J, Vos P, Xu W, Wittchen HU, Jönsson B, Olesen J; CDBE2010Study Group. Cost of disorders of the brain in Europe 2010. Eur Neuropsychopharmacol. 2011 Oct;21(10):718-79. doi: 10.1016/j.euroneuro.2011.08.008. Epub 2011 Sep 15. Erratum in: Eur Neuropsychopharmacol. 2012 Mar;22(3):237-8. den Bergh, Peter Van [corrected to Van den Bergh, Peter]. PMID: 21924589.
- Schmitt A, Falkai P, Papiol S. Neurodevelopmental disturbances in schizophrenia: evidence from genetic and environmental factors. J Neural Transm (Vienna). 2023 Mar;130(3):195-205. doi: 10.1007/s00702-022-02567-5. Epub 2022 Nov 12. PMID: 36370183; PMCID: PMC9660136.
- Haijma SV, Van Haren N, Cahn W, Koolschijn PC, Hulshoff Pol HE, Kahn RS. Brain volumes in schizophrenia: a meta-analysis in over 18 000 subjects. Schizophr Bull. 2013 Sep;39(5):1129-38. doi: 10.1093/schbul/sbs118. Epub 2012 Oct 5. PMID: 23042112; PMCID: PMC3756785.
- van Erp TGM, Walton E, Hibar DP, Schmaal L, Jiang W, Glahn DC, Pearlson GD, Yao N, Fukunaga M, Hashimoto R, Okada N, Yamamori H, Bustillo JR, Clark VP, Agartz I, Mueller BA, Cahn W, de Zwarte SMC, Hulshoff Pol HE, Kahn RS, Ophoff RA, van Haren NEM, Andreassen OA, Dale AM, Doan NT, Gurholt TP, Hartberg CB, Haukvik UK, Jørgensen KN, Lagerberg TV, Melle I, Westlye LT, Gruber O, Kraemer B, Richter A, Zilles D, Calhoun VD, Crespo-Facorro B, Roiz-Santiañez R, Tordesillas-Gutiérrez D, Loughland C, Carr VJ, Catts S, Cropley VL, Fullerton JM, Green MJ, Henskens FA, Jablensky A, Lenroot RK, Mowry BJ, Michie PT, Pantelis C, Quidé Y, Schall U, Scott RJ, Cairns MJ, Seal M, Tooney PA, Rasser PE, Cooper G, Shannon Weickert C, Weickert TW, Morris DW, Hong E, Kochunov P, Beard LM, Gur RE, Gur RC, Satterthwaite TD, Wolf DH, Belger A, Brown GG, Ford JM, Macciardi F, Mathalon DH, O'Leary DS, Potkin SG, Preda A, Voyvodic J, Lim KO, McEwen S, Yang F, Tan Y, Tan S, Wang Z, Fan F, Chen J, Xiang H, Tang S, Guo H, Wan P, Wei D, Bockholt HJ, Ehrlich S, Wolthusen RPF, King MD, Shoemaker JM, Sponheim SR, De Haan L, Koenders L, Machielsen MW, van Amelsvoort T, Veltman DJ, Assogna F, Banaj N, de Rossi P, Iorio M, Piras F, Spalletta G, McKenna PJ, Pomarol-Clotet E, Salvador R, Corvin A, Donohoe G, Kelly S, Whelan CD, Dickie EW, Rotenberg D, Voineskos AN, Ciufolini S, Radua J, Dazzan P, Murray R, Reis Marques T, Simmons A, Borgwardt S, Egloff L, Harrisberger F, Riecher-Rössler A, Smieskova R, Alpert KI, Wang L, Jönsson EG, Koops S, Sommer IEC, Bertolino A, Bonvino A, Di Giorgio A, Neilson E, Mayer AR, Stephen JM, Kwon JS, Yun JY, Cannon DM, McDonald C, Lebedeva I, Tomyshev AS, Akhadov T, Kaleda V, Fatouros-Bergman H, Flyckt L; Karolinska Schizophrenia Project; Busatto GF, Rosa PGP, Serpa MH, Zanetti MV, Hoschl C, Skoch A, Spaniel F, Tomecek D, Hagenaars SP, McIntosh AM, Whalley HC, Lawrie SM, Knöchel C, Oertel-Knöchel V, Stäblein M, Howells FM, Stein DJ, Temmingh HS, Uhlmann A, Lopez-Jaramillo C, Dima D, McMahon A, Faskowitz JI, Gutman BA, Jahanshad N, Thompson PM, Turner JA. Cortical Brain Abnormalities in 4474 Individuals With Schizophrenia and 5098 Control Subjects via the Enhancing Neuro Imaging Genetics Through Meta Analysis (ENIGMA) Consortium. Biol Psychiatry. 2018 Nov 1;84(9):644-654. doi: 10.1016/j.biopsych.2018.04.023. Epub 2018 May 14. PMID: 29960671; PMCID: PMC6177304.
- Cui Y, Li C, Liu B, Sui J, Song M, Chen J, Chen Y, Guo H, Li P, Lu L, Lv L, Ning Y, Wan P, Wang H, Wang H, Wu H, Yan H, Yan J, Yang Y, Zhang H, Zhang D, Jiang T. Consistent brain structural abnormalities and multisite individualised classification of schizophrenia using deep neural networks. Br J Psychiatry. 2022 Dec;221(6):732-739. doi: 10.1192/bjp.2022.22. PMID: 35144702.
- Vitolo E, Tatu MK, Pignolo C, Cauda F, Costa T, Ando' A, Zennaro A. White matter and schizophrenia: A meta-analysis of voxel-based morphometry and diffusion tensor imaging studies. Psychiatry Res Neuroimaging. 2017 Dec 30;270:8-21. doi: 10.1016/j.pscychresns.2017.09.014. Epub 2017 Sep 23. PMID: 28988022.
- Ellison-Wright I, Bullmore E. Meta-analysis of diffusion tensor imaging studies in schizophrenia. Schizophr Res. 2009 Mar;108(1-3):3-10. doi: 10.1016/j.schres.2008.11.021. Epub 2009 Jan 6. PMID: 19128945.
- Jiang J, Zhao YJ, Hu XY, Du MY, Chen ZQ, Wu M, Li KM, Zhu HY, Kumar P, Gong QY. Microstructural brain abnormalities in medication-free patients with major depressive disorder: a systematic review and meta-analysis of diffusion tensor imaging. J Psychiatry Neurosci. 2017 May;42(3):150-163. doi: 10.1503/jpn.150341. PMID: 27780031; PMCID: PMC5403660.
- Koshiyama D, Fukunaga M, Okada N, Morita K, Nemoto K, Usui K, Yamamori H, Yasuda Y, Fujimoto M, Kudo N, Azechi H, Watanabe Y, Hashimoto N, Narita H, Kusumi I, Ohi K, Shimada T, Kataoka Y, Yamamoto M, Ozaki N, Okada G, Okamoto Y, Harada K, Matsuo K, Yamasue H, Abe O, Hashimoto R, Takahashi T, Hori T, Nakataki M, Onitsuka T, Holleran L, Jahanshad N, van Erp TGM, Turner J, Donohoe G, Thompson PM, Kasai K, Hashimoto R; COCORO. White matter microstructural alterations across four major psychiatric disorders: mega-analysis study in 2937 individuals. Mol Psychiatry. 2020 Apr;25(4):883-895. doi: 10.1038/s41380-019-0553-7. Epub 2019 Nov 29. PMID: 31780770; PMCID: PMC7156346.
- Zhao G, Lau WKW, Wang C, Yan H, Zhang C, Lin K, Qiu S, Huang R, Zhang R. A Comparative Multimodal Meta-analysis of Anisotropy and Volume Abnormalities in White Matter in People Suffering From Bipolar Disorder or Schizophrenia. Schizophr Bull. 2022 Jan 21;48(1):69-79. doi: 10.1093/schbul/sbab093. PMID: 34374427; PMCID: PMC8781378.
- Bubb EJ, Metzler-Baddeley C, Aggleton JP. The cingulum bundle: Anatomy, function, and dysfunction. Neurosci Biobehav Rev. 2018 Sep;92:104-127. doi: 10.1016/j.neubiorev.2018.05.008. Epub 2018 May 16. PMID: 29753752; PMCID: PMC6090091.
- Brunet-Gouet E, Decety J. Social brain dysfunctions in schizophrenia: a review of neuroimaging studies. Psychiatry Res. 2006 Dec 1;148(2-3):75-92. doi: 10.1016/j.pscychresns.2006.05.001. Epub 2006 Nov 7. PMID: 17088049.
- Green MF, Bearden CE, Cannon TD, Fiske AP, Hellemann GS, Horan WP, Kee K, Kern RS, Lee J, Sergi MJ, Subotnik KL, Sugar CA, Ventura J, Yee CM, Nuechterlein KH. Social cognition in schizophrenia, Part 1: performance across phase of illness. Schizophr Bull. 2012 Jun;38(4):854-64. doi: 10.1093/schbul/sbq171. Epub 2011 Feb 23. PMID: 21345917; PMCID: PMC3406534.
- Fan FM, Tan SP, Yang FD, Tan YL, Zhao YL, Chen N, Li BB, Song CS, Wang YH, Jin Z, Zhou DF, Milham MP, Zou YZ, Zuo XN. Ventral medial prefrontal functional connectivity and emotion regulation in chronic schizophrenia: a pilot study. Neurosci Bull. 2013 Feb;29(1):59-74. doi: 10.1007/s12264-013-1300-8. Epub 2013 Jan 14. PMID: 23319314; PMCID: PMC5561861.
- Nestor PG, Kubicki M, Gurrera RJ, Niznikiewicz M, Frumin M, McCarley RW, Shenton ME. Neuropsychological correlates of diffusion tensor imaging in schizophrenia. Neuropsychology. 2004 Oct;18(4):629-637. doi: 10.1037/0894-4105.18.4.629. PMID: 15506830; PMCID: PMC2790923.
- Nestor PG, Kubicki M, Spencer KM, Niznikiewicz M, McCarley RW, Shenton ME. Attentional networks and cingulum bundle in chronic schizophrenia. Schizophr Res. 2007 Feb;90(1-3):308-15. doi: 10.1016/j.schres.2006.10.005. Epub 2006 Dec 5. PMID: 17150337; PMCID: PMC1906862.
- Kubicki M, Niznikiewicz M, Connor E, Ungar L, Nestor P, Bouix S, Dreusicke M, Kikinis R, McCarley R, Shenton M. Relationship Between White Matter Integrity, Attention, and Memory in Schizophrenia: A Diffusion Tensor Imaging Study. Brain Imaging Behav. 2009 Jun 1;3(2):191-201. doi: 10.1007/s11682-009-9061-8. PMID: 20556231; PMCID: PMC2885800.
- Karbasforoushan H, Duffy B, Blackford JU, Woodward ND. Processing speed impairment in schizophrenia is mediated by white matter integrity. Psychol Med. 2015 Jan;45(1):109-20. doi: 10.1017/S0033291714001111. Epub 2014 May 15. PMID: 25066842; PMCID: PMC5297385.
- Kim NS, Lee TY, Hwang WJ, Kwak YB, Kim S, Moon SY, Lho SK, Oh S, Kwon JS. White Matter Correlates of Theory of Mind in Patients With First-Episode Psychosis. Front Psychiatry. 2021 Mar 5;12:617683. doi: 10.3389/fpsyt.2021.617683. PMID: 33746794; PMCID: PMC7973210.
- Whitford TJ, Kubicki M, Pelavin PE, Lucia D, Schneiderman JS, Pantelis C, McCarley RW, Shenton ME. Cingulum bundle integrity associated with delusions of control in schizophrenia: Preliminary evidence from diffusion-tensor tractography. Schizophr Res. 2015 Jan;161(1):36-41. doi: 10.1016/j.schres.2014.08.033. Epub 2014 Oct 11. PMID: 25311780; PMCID: PMC4303408.
- Palaniyappan L, Al-Radaideh A, Mougin O, Das T, Gowland P, Liddle PF. Aberrant myelination of the cingulum and Schneiderian delusions in schizophrenia: a 7T magnetization transfer study. Psychol Med. 2019 Aug;49(11):1890-1896. doi: 10.1017/S0033291718002647. Epub 2018 Sep 19. PMID: 30229713..
- Gao Y, Li M, Huang AS, Anderson AW, Ding Z, Heckers SH, Woodward ND, Gore JC. Lower functional connectivity of white matter during rest and working memory tasks is associated with cognitive impairments in schizophrenia. Schizophr Res. 2021 Jul;233:101-110. doi: 10.1016/j.schres.2021.06.013. Epub 2021 Jun 29. PMID: 34215467; PMCID: PMC8442250.
- Nave KA. Myelination and support of axonal integrity by glia. Nature. 2010 Nov 11;468(7321):244-52. doi: 10.1038/nature09614. PMID: 21068833.
- Cassoli JS, Guest PC, Malchow B, Schmitt A, Falkai P, Martins-de-Souza D. Disturbed macro-connectivity in schizophrenia linked to oligodendrocyte dysfunction: from structural findings to molecules. NPJ Schizophr. 2015 Sep 23;1:15034. doi: 10.1038/npjschz.2015.34. PMID: 27336040; PMCID: PMC4849457.
- Schmitt A, Steyskal C, Bernstein HG, Schneider-Axmann T, Parlapani E, Schaeffer EL, Gattaz WF, Bogerts B, Schmitz C, Falkai P. Stereologic investigation of the posterior part of the hippocampus in schizophrenia. Acta Neuropathol. 2009 Apr;117(4):395-407. doi: 10.1007/s00401-008-0430-y. Epub 2008 Sep 6. PMID: 18777029.
- Falkai P, Malchow B, Wetzestein K, Nowastowski V, Bernstein HG, Steiner J, Schneider-Axmann T, Kraus T, Hasan A, Bogerts B, Schmitz C, Schmitt A. Decreased Oligodendrocyte and Neuron Number in Anterior Hippocampal Areas and the Entire Hippocampus in Schizophrenia: A Stereological Postmortem Study. Schizophr Bull. 2016 Jul;42 Suppl 1(Suppl 1):S4-S12. doi: 10.1093/schbul/sbv157. PMID: 27460617; PMCID: PMC4960426..
- Schmitt A, Tatsch L, Vollhardt A, Schneider-Axmann T, Raabe FJ, Roell L, Heinsen H, Hof PR, Falkai P, Schmitz C. Decreased Oligodendrocyte Number in Hippocampal Subfield CA4 in Schizophrenia: A Replication Study. Cells. 2022 Oct 15;11(20):3242. doi: 10.3390/cells11203242. PMID: 36291109; PMCID: PMC9600243.
- Falkai P, Steiner J, Malchow B, Shariati J, Knaus A, Bernstein HG, Schneider-Axmann T, Kraus T, Hasan A, Bogerts B, Schmitt A. Oligodendrocyte and Interneuron Density in Hippocampal Subfields in Schizophrenia and Association of Oligodendrocyte Number with Cognitive Deficits. Front Cell Neurosci. 2016 Mar 30;10:78. doi: 10.3389/fncel.2016.00078. PMID: 27065804; PMCID: PMC4811909.
- Schmitt A, Falkai P. Cellular pathology in the limbic system in schizophrenia. Eur Arch Psychiatry Clin Neurosci. 2023 Sep;273(6):1207-1208. doi: 10.1007/s00406-023-01659-x. PMID: 37542559; PMCID: PMC10449720.
- Hof PR, Haroutunian V, Friedrich VL Jr, Byne W, Buitron C, Perl DP, Davis KL. Loss and altered spatial distribution of oligodendrocytes in the superior frontal gyrus in schizophrenia. Biol Psychiatry. 2003 Jun 15;53(12):1075-85. doi: 10.1016/s0006-3223(03)00237-3. PMID: 12814859.
- Falkai P, Rossner MJ, Raabe FJ, Wagner E, Keeser D, Maurus I, Roell L, Chang E, Seitz-Holland J, Schulze TG, Schmitt A. Disturbed Oligodendroglial Maturation Causes Cognitive Dysfunction in Schizophrenia: A New Hypothesis. Schizophr Bull. 2023 Nov 29;49(6):1614-1624. doi: 10.1093/schbul/sbad065. PMID: 37163675; PMCID: PMC10686333.
- Kreczmanski P, Heinsen H, Mantua V, Woltersdorf F, Masson T, Ulfig N, Schmidt-Kastner R, Korr H, Steinbusch HW, Hof PR, Schmitz C. Volume, neuron density and total neuron number in five subcortical regions in schizophrenia. Brain. 2007 Mar;130(Pt 3):678-92. doi: 10.1093/brain/awl386. Epub 2007 Feb 15. PMID: 17303593.
- Heinsen H. Serial thick, frozen, gallocyanin stained sections of human central nervous system. J Histotechnol. 1991;14:167-173.
- Heinsen H, Arzberger T, Schmitz C. Celloidin mounting (embedding without infiltration) - a new, simple and reliable method for producing serial sections of high thickness through complete human brains and its application to stereological and immunohistochemical investigations. J Chem Neuroanat. 2000 Oct;20(1):49-59. doi: 10.1016/s0891-0618(00)00067-3. PMID: 11074343.
- Heinsen H, Rüb U, Bauer M, Ulmar G, Bethke B, Schüler M, Böcker F, Eisenmenger W, Götz M, Korr H, Schmitz C. Nerve cell loss in the thalamic mediodorsal nucleus in Huntington's disease. Acta Neuropathol. 1999 Jun;97(6):613-22. doi: 10.1007/s004010051037. PMID: 10378380.
- van Kooten IA, Palmen SJ, von Cappeln P, Steinbusch HW, Korr H, Heinsen H, Hof PR, van Engeland H, Schmitz C. Neurons in the fusiform gyrus are fewer and smaller in autism. Brain. 2008 Apr;131(Pt 4):987-99. doi: 10.1093/brain/awn033. Epub 2008 Mar 10. PMID: 18332073.
- Schmitz C, Hof PR. Design-based stereology in neuroscience. Neuroscience. 2005;130(4):813-31. doi: 10.1016/j.neuroscience.2004.08.050. PMID: 15652981.
- West MJ, Slomianka L, Gundersen HJ. Unbiased stereological estimation of the total number of neurons in thesubdivisions of the rat hippocampus using the optical fractionator. Anat Rec. 1991 Dec;231(4):482-97. doi: 10.1002/ar.1092310411. PMID: 1793176.
- Schmitz C. Variation of fractionator estimates and its prediction. Anat Embryol (Berl). 1998 Nov;198(5):371-97. doi: 10.1007/s004290050191. PMID: 9801058.
- Schmitz C, Hof PR. Recommendations for straightforward and rigorous methods of counting neurons based on a computer simulation approach. J Chem Neuroanat. 2000 Oct;20(1):93-114. doi: 10.1016/s0891-0618(00)00066-1. PMID: 11074347.
- Glaser ER, Green G, Hendricks S. Stereology for biological research: With a focus on neuroscience. Williston, VT, USA: MBF Press ; 2007:2.
- Schmitz C, Born M, Dolezel P, Rutten BP, de Saint-Georges L, Hof PR, Korr H. Prenatal protracted irradiation at very low dose rate induces severe neuronal loss in rat hippocampus and cerebellum. Neuroscience. 2005;130(4):935-48. doi: 10.1016/j.neuroscience.2004.08.034. PMID: 15652991.
- Falkai P, Raabe F, Bogerts B, Schneider-Axmann T, Malchow B, Tatsch L, Huber V, Slapakova L, Dobrowolny H, Schmitz C, Cantuti-Castelvetri L, Simons M, Steiner J, Schmitt A. Association between altered hippocampal oligodendrocyte number and neuronal circuit structures in schizophrenia: a postmortem analysis. Eur Arch Psychiatry Clin Neurosci. 2020 Jun;270(4):413-424. doi: 10.1007/s00406-019-01067-0. Epub 2019 Sep 24. PMID: 31552495.
- Zeng J, Zhang W, Xiao Y, Fu G, Liu L, Tang B, Hu N, Sweeney JA, Lui S, Gong Q. Pretreatment abnormalities in white matter integrity predict one-year clinical outcome in first episode schizophrenia. Schizophr Res. 2021 Feb;228:241-248. doi: 10.1016/j.schres.2020.12.011. Epub 2021 Jan 22. PMID: 33486391.
- Fitzsimmons J, Rosa P, Sydnor VJ, Reid BE, Makris N, Goldstein JM, Mesholam-Gately RI, Woodberry K, Wojcik J, McCarley RW, Seidman LJ, Shenton ME, Kubicki M. Cingulum bundle abnormalities and risk for schizophrenia. Schizophr Res. 2020 Jan;215:385-391. doi: 10.1016/j.schres.2019.08.017. Epub 2019 Aug 30. PMID: 31477373.
- Schmitt A, Simons M, Cantuti-Castelvetri L, Falkai P. A new role for oligodendrocytes and myelination in schizophrenia and affective disorders? Eur Arch Psychiatry Clin Neurosci. 2019 Jun;269(4):371-372. doi: 10.1007/s00406-019-01019-8. PMID: 31076838.
- Raabe FJ, Slapakova L, Rossner MJ, Cantuti-Castelvetri L, Simons M, Falkai PG, Schmitt A. Oligodendrocytes as A New Therapeutic Target in Schizophrenia: From Histopathological Findings to Neuron-Oligodendrocyte Interaction. Cells. 2019 Nov 23;8(12):1496. doi: 10.3390/cells8121496. PMID: 31771166; PMCID: PMC6952785.
- Höistad M, Segal D, Takahashi N, Sakurai T, Buxbaum JD, Hof PR. Linking white and grey matter in schizophrenia: oligodendrocyte and neuron pathology in the prefrontal cortex. Front Neuroanat. 2009 Jul 2;3:9. doi: 10.3389/neuro.05.009.2009. PMID: 19636386; PMCID: PMC2713751.
- Gomes FV, Grace AA. Adolescent Stress as a Driving Factor for Schizophrenia Development-A Basic Science Perspective. Schizophr Bull. 2017 May 1;43(3):486-489. doi: 10.1093/schbul/sbx033. PMID: 28419390; PMCID: PMC5464111.
- Gomes FV, Grace AA. Adolescent Stress as a Driving Factor for Schizophrenia Development-A Basic Science Perspective. Schizophr Bull. 2017 May 1;43(3):486-489. doi: 10.1093/schbul/sbx033. PMID: 28419390; PMCID: PMC5464111.
- Makinodan M, Rosen KM, Ito S, Corfas G. A critical period for social experience-dependent oligodendrocyte maturation and myelination. Science. 2012 Sep 14;337(6100):1357-60. doi: 10.1126/science.1220845. PMID: 22984073; PMCID: PMC4165613.
- Pardiñas AF, Holmans P, Pocklington AJ, Escott-Price V, Ripke S, Carrera N, Legge SE, Bishop S, Cameron D, Hamshere ML, Han J, Hubbard L, Lynham A, Mantripragada K, Rees E, MacCabe JH, McCarroll SA, Baune BT, Breen G, Byrne EM, Dannlowski U, Eley TC, Hayward C, Martin NG, McIntosh AM, Plomin R, Porteous DJ, Wray NR, Caballero A, Geschwind DH, Huckins LM, Ruderfer DM, Santiago E, Sklar P, Stahl EA, Won H, Agerbo E, Als TD, Andreassen OA, Bækvad-Hansen M, Mortensen PB, Pedersen CB, Børglum AD, Bybjerg-Grauholm J, Djurovic S, Durmishi N, Pedersen MG, Golimbet V, Grove J, Hougaard DM, Mattheisen M, Molden E, Mors O, Nordentoft M, Pejovic-Milovancevic M, Sigurdsson E, Silagadze T, Hansen CS, Stefansson K, Stefansson H, Steinberg S, Tosato S, Werge T; GERAD1 Consortium; CRESTAR Consortium; Collier DA, Rujescu D, Kirov G, Owen MJ, O'Donovan MC, Walters JTR. Common schizophrenia alleles are enriched in mutation-intolerant genes and in regions under strong background selection. Nat Genet. 2018 Mar;50(3):381-389. doi: 10.1038/s41588-018-0059-2. Epub 2018 Feb 26. Erratum in: Nat Genet. 2019 Jul;51(7):1193. doi: 10.1038/s41588-019-0450-7. PMID: 29483656; PMCID: PMC5918692.
- Wirgenes KV, Sønderby IE, Haukvik UK, Mattingsdal M, Tesli M, Athanasiu L, Sundet K, Røssberg JI, Dale AM, Brown AA, Agartz I, Melle I, Djurovic S, Andreassen OA. TCF4 sequence variants and mRNA levels are associated with neurodevelopmental characteristics in psychotic disorders. Transl Psychiatry. 2012 May 8;2(5):e112. doi: 10.1038/tp.2012.39. PMID: 22832956; PMCID: PMC3365258.
- Wedel M, Fröb F, Elsesser O, Wittmann MT, Lie DC, Reis A, Wegner M. Transcription factor Tcf4 is the preferred heterodimerization partner for Olig2 in oligodendrocytes and required for differentiation. Nucleic Acids Res. 2020 May 21;48(9):4839-4857. doi: 10.1093/nar/gkaa218. PMID: 32266943; PMCID: PMC7229849.
- Windrem MS, Osipovitch M, Liu Z, Bates J, Chandler-Militello D, Zou L, Munir J, Schanz S, McCoy K, Miller RH, Wang S, Nedergaard M, Findling RL, Tesar PJ, Goldman SA. Human iPSC Glial Mouse Chimeras Reveal Glial Contributions to Schizophrenia. Cell Stem Cell. 2017 Aug 3;21(2):195-208.e6. doi: 10.1016/j.stem.2017.06.012. Epub 2017 Jul 20. PMID: 28736215; PMCID: PMC5576346.
- Häfner H, Riecher-Rössler A, An Der Heiden W, Maurer K, Fätkenheuer B, Löffler W. Generating and testing a causal explanation of the gender difference in age at first onset of schizophrenia. Psychol Med. 1993 Nov;23(4):925-40. doi: 10.1017/s0033291700026398. PMID: 8134516.
- Barendse MEA, Lara GA, Guyer AE, Swartz JR, Taylor SL, Shirtcliff EA, Lamb ST, Miller C, Ng J, Yu G, Tully LM. Sex and pubertal influences on the neurodevelopmental underpinnings of schizophrenia: A case for longitudinal research on adolescents. Schizophr Res. 2023 Feb;252:231-241. doi: 10.1016/j.schres.2022.12.011. Epub 2023 Jan 20. PMID: 36682313; PMCID: PMC10725041.
- Konopaske GT, Dorph-Petersen KA, Sweet RA, Pierri JN, Zhang W, Sampson AR, Lewis DA. Effect of chronic antipsychotic exposure on astrocyte and oligodendrocyte numbers in macaque monkeys. Biol Psychiatry. 2008 Apr 15;63(8):759-65. doi: 10.1016/j.biopsych.2007.08.018. Epub 2007 Oct 22. PMID: 17945195; PMCID: PMC2386415.
- Wang H, Xu H, Niu J, Mei F, Li X, Kong J, Cai W, Xiao L. Haloperidol activates quiescent oligodendroglia precursor cells in the adult mouse brain. Schizophr Res. 2010 Jun;119(1-3):164-74. doi: 10.1016/j.schres.2010.02.1068. Epub 2010 Mar 25. PMID: 20346631.
- Fard MK, van der Meer F, Sánchez P, Cantuti-Castelvetri L, Mandad S, Jäkel S, Fornasiero EF, Schmitt S, Ehrlich M, Starost L, Kuhlmann T, Sergiou C, Schultz V, Wrzos C, Brück W, Urlaub H, Dimou L, Stadelmann C, Simons M. BCAS1 expression defines a population of early myelinating oligodendrocytes in multiple sclerosis lesions. Sci Transl Med. 2017 Dec 6;9(419):eaam7816. doi: 10.1126/scitranslmed.aam7816. PMID: 29212715; PMCID: PMC7116798.
- Falkai P, Rossner MJ, Raabe FJ, Wagner E, Keeser D, Maurus I, Roell L, Chang E, Seitz-Holland J, Schulze TG, Schmitt A. Disturbed Oligodendroglial Maturation Causes Cognitive Dysfunction in Schizophrenia: A New Hypothesis. Schizophr Bull. 2023 Nov 29;49(6):1614-1624. doi: 10.1093/schbul/sbad065. PMID: 37163675; PMCID: PMC10686333.
- Adriano F, Caltagirone C, Spalletta G. Hippocampal volume reduction in first-episode and chronic schizophrenia: a review and meta-analysis. Neuroscientist. 2012 Apr;18(2):180-200. doi: 10.1177/1073858410395147. Epub 2011 Apr 29. PMID: 21531988.
- van Erp TG, Hibar DP, Rasmussen JM, Glahn DC, Pearlson GD, Andreassen OA, Agartz I, Westlye LT, Haukvik UK, Dale AM, Melle I, Hartberg CB, Gruber O, Kraemer B, Zilles D, Donohoe G, Kelly S, McDonald C, Morris DW, Cannon DM, Corvin A, Machielsen MW, Koenders L, de Haan L, Veltman DJ, Satterthwaite TD, Wolf DH, Gur RC, Gur RE, Potkin SG, Mathalon DH, Mueller BA, Preda A, Macciardi F, Ehrlich S, Walton E, Hass J, Calhoun VD, Bockholt HJ, Sponheim SR, Shoemaker JM, van Haren NE, Hulshoff Pol HE, Ophoff RA, Kahn RS, Roiz-Santiañez R, Crespo-Facorro B, Wang L, Alpert KI, Jönsson EG, Dimitrova R, Bois C, Whalley HC, McIntosh AM, Lawrie SM, Hashimoto R, Thompson PM, Turner JA. Subcortical brain volume abnormalities in 2028 individuals with schizophrenia and 2540 healthy controls via the ENIGMA consortium. Mol Psychiatry. 2016 Apr;21(4):547-53. doi: 10.1038/mp.2015.63. Epub 2015 Jun 2. Erratum in: Mol Psychiatry. 2016 Apr;21(4):585. doi: 10.1038/mp.2015.118. Pol, H E H [Corrected to Hulshoff Pol, H E]. PMID: 26033243; PMCID: PMC4668237.
- Kuo SS, Pogue-Geile MF. Variation in fourteen brain structure volumes in schizophrenia: A comprehensive meta-analysis of 246 studies. Neurosci Biobehav Rev. 2019 Mar;98:85-94. doi: 10.1016/j.neubiorev.2018.12.030. Epub 2019 Jan 4. PMID: 30615934; PMCID: PMC6401304.
- Chopra S, Segal A, Oldham S, Holmes A, Sabaroedin K, Orchard ER, Francey SM, O'Donoghue B, Cropley V, Nelson B, Graham J, Baldwin L, Tiego J, Yuen HP, Allott K, Alvarez-Jimenez M, Harrigan S, Fulcher BD, Aquino K, Pantelis C, Wood SJ, Bellgrove M, McGorry PD, Fornito A. Network-Based Spreading of Gray Matter Changes Across Different Stages of Psychosis. JAMA Psychiatry. 2023 Dec 1;80(12):1246-1257. doi: 10.1001/jamapsychiatry.2023.3293. PMID: 37728918; PMCID: PMC10512169.
- Dong D, Wang Y, Chang X, Luo C, Yao D. Dysfunction of Large-Scale Brain Networks in Schizophrenia: A Meta-analysis of Resting-State Functional Connectivity. Schizophr Bull. 2018 Jan 13;44(1):168-181. doi: 10.1093/schbul/sbx034. PMID: 28338943; PMCID: PMC5767956.
- Sha Z, Wager TD, Mechelli A, He Y. Common Dysfunction of Large-Scale Neurocognitive Networks Across Psychiatric Disorders. Biol Psychiatry. 2019 Mar 1;85(5):379-388. doi: 10.1016/j.biopsych.2018.11.011. Epub 2018 Nov 23. PMID: 30612699.
- Brandl F, Avram M, Weise B, Shang J, Simões B, Bertram T, Hoffmann Ayala D, Penzel N, Gürsel DA, Bäuml J, Wohlschläger AM, Vukadinovic Z, Koutsouleris N, Leucht S, Sorg C. Specific Substantial Dysconnectivity in Schizophrenia: A Transdiagnostic Multimodal Meta-analysis of Resting-State Functional and Structural Magnetic Resonance Imaging Studies. Biol Psychiatry. 2019 Apr 1;85(7):573-583. doi: 10.1016/j.biopsych.2018.12.003. Epub 2018 Dec 11. PMID: 30691673.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.