Medicine Group. 2024 September 11;5(9):1107-1110. doi: 10.37871/jbres1996.
Prophylaxis by Administration of Stable Iodine: Principle, Evolution and Recommendations
Maâmar Souidi1*, Marc Benderitter1 and Francois Caire-Maurisier2
2Army Health Academy, 75005 Paris, France
- Potassium Iodide
- Thyroid
- Nuclear accident
- Iodine Thyroid blocking
- Repetitive prophylaxis
Abstract
Prophylaxis by stable iodine “KI 65 mg, breakable tablet” is a pharmacological countermeasure of radiological protection adapted for the prevention of thyroid cancer after exposure to radioactive iodine. A first modification of the marketing authorization of “KI 65 mg, breakable tablet” for repeated prophylaxis for adults and children above 12 years old was obtained in France in 2020. At this point in time, the recommendation of a unique “KI” intake by pregnant and breastfeeding women and children below 12 years old remain valid.
Introduction
Release of radioactive products
An uncontrolled nuclear accident can lead to the release of fission products, including the radioactive iodine. Depending on the nature of the accident, the quantity and type of radioactive products dispersed differ. Details of the quantities and nature of the radioactive products dispersed in the 2 nuclear accidents at Chernobyl-1986 and Fukushima-2011 have been described in International Atomic Energy Agency (IAEA) and The United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR) reports [1,2]. The dispersion of radioactive iodine isotopes, particularly iodine-131 (half-life of 8 days) in the event of an accidental release, is a major issue due to its potential impact on health. Radioactive iodine-131 can be released in a plume or cloud, contaminating the environment (air, water, soil, etc.) and depositing on skin and clothing, leading to external exposure. Inhalation of contaminated air and ingestion of contaminated food and drinking water can lead to internal exposure to radiation and uptake of radioactive iodine, mainly by one target organ, the thyroid, which concentrates almost all the iodine contained in the human body. Exposure to radioactive iodine can then have deleterious long term effects on the thyroid gland, including cancer.
Thyroid cancer
Epidemiological studies of exposed populations, particularly following the Chernobyl accident, show a significant increase in the risk of thyroid cancer in children as the dose increases [3,4]. The 2018 UNSCEAR report [4] specifies that the fraction of thyroid cancer incidence attributable to radiation exposure in regions affected by the Chernobyl accident is around 25%. Children and adolescents (aged < 18) are particularly sensitive to radioactive iodine contamination. Pregnant and breast-feeding women are also sensitive to radioactive iodine exposure for their offspring. Epidemiological studies, on the other hand, provide only a weak suggestion of an increase in the incidence of thyroid cancer in the population exposed in adulthood [4]. Only one study of liquidators shows an excess of thyroid cancer with a dose-response relationship depending on the total dose to the thyroid [5].
Protection strategy
The strategy for protecting the population in the event of a radiological threat or release during a nuclear accident aims to limit exposure to as low a level as possible. The 4 food and drinking restriction main measures are: evacuation, sheltering and taking stable iodine in the form of potassium salts. Taking potassium iodide (KI) tablets is one of the main and most effective measures [6] for protecting people who may be exposed to these releases. In the event of radioactive iodine contamination, circulating radioactive iodine follows the same metabolic pathways as stable iodine [7]. Once captured by the thyroid gland, radioactive iodine causes damage to the DNA of thyrocytes. Depending on the activity delivered, this DNA damage can lead to genetic instability that initiates the cancerization process. The administration of excess stable iodine to people likely to be exposed to radioactive iodine releases is a protective measure recognized by the international organizations involved in radiation protection. It does not prevent radioactive iodine from entering WHO the body (by inhalation, ingestion or, more modestly, through the skin), but it does prevent it from accumulating in the thyroid gland. Taking stable iodine is a preventive measure that should be taken before exposure or, failing that, within the first few hours of contamination. This measure then loses most of its effectiveness or becomes totally ineffective if taken more than 24 to 48 hours after exposure. Once radioactive iodine has attached itself to the thyroid, stable iodine cannot reverse the deleterious effects [8]. The World Health Organization (WHO) therefore recommends [9]: prophylaxis with stable iodine as the reference treatment for radioactive iodine contamination; a stable iodine dosage adapted to the population categories; ingestion of stable iodine as early as possible, and if possible two hours before the radioactive emission. Prophylactic treatment with potassium iodide offers thyroid protection for people exposed to radioactive iodine but does not offer protection against other radioelements. This protection is the result, on the one hand of a dilution effect of radioactive iodine and, on the other hand, of the induction of a physiological regulation of the thyroid, the Wolff-Chaikoff effect [10], which reduces the thyroid's capacity to absorb iodine and to synthesize thyroid hormones. The time lag between uptake of stable iodine and exposure is important. Saturating the thyroid gland by ingesting stable iodine before (ideally in the 24 hours before exposure) or in the first few hours after exposure prevents the concentration of radioactive iodine and avoids or reduces the risk of damage to the gland. Once the thyroid gland has been blocked, it is protected for a period of 24 to 48 hours. The radioactive iodine is then eliminated naturally by the human body.
The different populations and dosage regimens
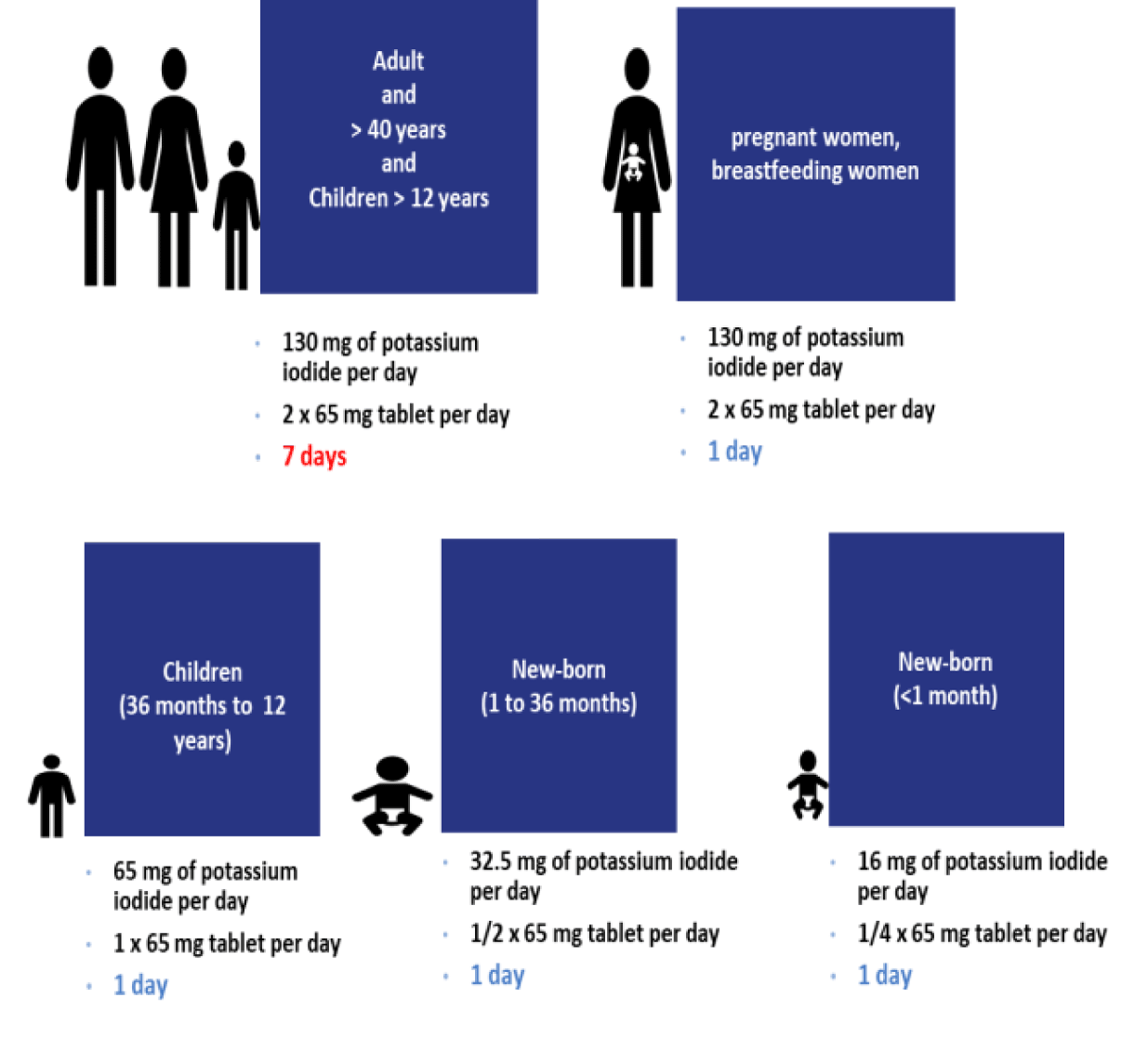
The dose of KI recommended by the WHO is adapted to age: Newborns under 1 month: 16 mg of KI; Children aged 1 month to 3 years: 32 mg of KI; Children aged 3 to 12 years: 65 mg of KI; Children aged over 12 years and adults: 130 mg of KI. However, the over-40s are not considered a priority. Unlike other age groups, treatment strategies will slightly vary from country to country [9]. Countries that have decided not to distribute stable iodine tablets to people over the age of 40 justify this choice on the basis of a less favorable benefit-risk ratio for this age group: the risk of developing thyroid cancer is expected lower, whereas the risk of developing cardiovascular adverse effects following the administration of stable iodine is expected greater [9,11]. In addition, the "iodine doctrine" has until now assumed that people would be exposed for a relatively short period to radioactive releases, from which they would be rapidly removed, thanks in particular to the synergistic action of other protective measures, such as evacuation or sheltering. However, the events that occurred in March 2011 at the Fukushima Daiichi nuclear power plant in Japan showed that, even if evacuation and sheltering in a highly degraded situation had been brought under control, optimuisation of the population is questionable because of prolonged releases of radioactive materials into the environment and/or repeated releases following the repetition of events affecting several nuclear reactors or spent fuel storage pools. In such a situation of prolonged and/or repeated discharges, the ingestion of a single dose of stable iodine would under no circumstances provide satisfactory protection for the thyroid glands of people likely to be exposed. The administration of further doses of stable iodine to the same population would be essential for the effective prevention of thyroid pathologies secondary to exposure to radioactive iodine. As part of this, the PRIODAC project (repeated Prophylaxis with Stable I0Dine in Accidental situations) was set up. The aims of this research program are to determine the methods for repeated administration of stable iodine over 7 days, to assess potential undesirable effects of repeated administration of stable iodine on the body's major physiological functions, to gain a better understanding of the molecular mechanisms regulating iodine metabolism, and to change the current marketing authorization (MA / France) for potassium iodide tablets dosed at 65 mg. The PRIODAC project has already produced initial results for children aged over 12 and adults and investigation is currently being extended to the remaining age groups: children under 12, pregnant women and nursing mothers. To date, an initial extension of the French MA for KI in adults and children aged over 12 has already been obtained in March 2020 for daily use, repeated if necessary for up to 7 days [12,13].
Conclusion
Single prophylaxis (130 mg) or repeated doses for up to 7 days with potassium iodide is a pharmacological countermeasure to prevent thyroid cancer in the context of short time or prolonged exposure to radioactive iodine.
However, at this stage, it is still advisable to exclude the repeated use of potassium iodide by pregnant or breast-feeding women and children under the age of 12. They should therefore be evacuated from the contaminated area as a matter of priority.
Acknowledgment
This work was co-funded by the French Government under the ‘Investments for the Future’ program (Nuclear Safety and Radioprotection Research (RSNR) action), managed by the French National Research Agency (ANR- 11-RSNR-0019)". The authors thank the ANR program manager Dr Antony Lebeau for his support and confidence and Dr Stéphane Flamant for grammatical correction and proofreading of the manuscript.
Conflict of Interests
The authors do not report any conflict of interests regarding the publication of this paper.
References
- IAEA. International Atomic Energy Agency, Frequently asked Chernobyl questions. 2016.
- UNSCEAR. United Nations Scientific Committee on the Effects of Atomic Radiation 2020/2021 Report to the General Assembly with Scientific Annexes Volume I, II, Scientific Annex B sources, effects and risks of ionizing radiation.
- Ron E. Thyroid cancer incidence among people living in areas contaminated by radiation from the Chernobyl accident. Health Phys. 2007 Nov;93(5):502-11. doi: 10.1097/01.HP.0000279018.93081.29. PMID: 18049226.
- UNSCEAR. Evaluation of data on thyroid cancer in regions affected by the Chernobyl accident: a white paper to guide the Scientific Committee’s future pro gramme of work. In: Sources and effects of ionizing radiation. United Nations Scientific Committee on the Effects of Atomic Radiation. 2018.
- UNSCEAR. sources and effects of ionizing radiation, United Nations Scientific Committee on the Effects of Atomic Radiation, Report to the General Assembly with Scientific Annexes Volume II, Scientific Annexes C, D and E. 2008.
- Nauman J, Wolff J. Iodide prophylaxis in Poland after the Chernobyl reactor accident: benefits and risks. Am J Med. 1993 May;94(5):524-532. doi: 10.1016/0002-9343(93)90089-8. PMID: 8498398.
- Carvalho DP, Dupuy C. Thyroid hormone biosynthesis and release. Mol Cell Endocrinol. 2017 Dec 15;458:6-15. doi: 10.1016/j.mce.2017.01.038. Epub 2017 Jan 31. PMID: 28153798.
- Zanzonico PB, Becker DV. Effects of time of administration and dietary iodine levels on potassium iodide (KI) blockade of thyroid irradiation by 131I from radioactive fallout. Health Phys. 2000 Jun;78(6):660-7. doi: 10.1097/00004032-200006000-00008. PMID: 10832925.
- Iodine Thyroid Blocking: Guidelines for Use in Planning for and Responding to Radiological and Nuclear Emergencies. Geneva: World Health Organization; 2017. PMID: 29630192.
- WOLFF J, CHAIKOFF IL. Plasma inorganic iodide as a homeostatic regulator of thyroid function. J Biol Chem. 1948 Jun;174(2):555-64. PMID: 18865621.
- Iervasi G, Nicolini G. Thyroid hormone and cardiovascular system: from basic concepts to clinical application. Intern Emerg Med. 2013 Apr;8 Suppl 1:S71-4. doi: 10.1007/s11739-013-0911-4. PMID: 23435988.
- Toft DJ, Schneider AB. Protecting the Thyroid in Times of Conflict (Ukraine 2022). Thyroid. 2022 Jun;32(6):607-610. doi: 10.1089/thy.2022.0135. Epub 2022 May 23. PMID: 35469429.
- Martin JC, Pourcher T, Phan G, Guglielmi J, Crambes C, Caire-Maurisier F, Lebsir D, Cohen D, Rosique C, Jing L, Hichri M, Salleron L, Darcourt J, Souidi M, Benderitter M. Review of the PRIODAC project on thyroid protection from radioactive iodine by repeated iodine intake in individuals aged 12. Eur Thyroid J. 2024 Jan 1;13(1):e230139. doi: 10.1530/ETJ-23-0139. Epub ahead of print. PMID: 38241789; PMCID: PMC10895330.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.