Medicine Group. 2024 August 16;5(8):989-995. doi: 10.37871/jbres1979.
Patterns of Recurrence in Patients with Non-Metastatic Head and Neck Squamous Cell Cancer. Can Radiotherapy Interruptions Influence The Patients’ Prognosis?
Bosurgi A1*, Pontoriero A1, Brogna A2, Siragusa C2, Parisi S1 and Pergolizzi S1
2Medical Physics Unit, A.O.U. G. Martino of Messina, 98100 Messina, Italy
Abstract
Aims: To determine the impact of interruptions/suspensions of radiotherapy in relation to the stage and histopathological features in patients with Non-Metastatic Head and Neck Squamous Cell Cancer (NMHNSCC), undergoing chemotherapy and Intensity-Modulated Radiation Therapy (IMRT).
Materials and methods: We retrospectively analyzed patients with T3-T4 N+ NMHNSCC treated with IMRT in our department from January 2014 to December 2023, matching the following inclusion criteria: age > 18 years, availability of 18F-FDG CT/PET before and after the treatment, T3-4 N+ disease, adjuvant or exclusive Concurrent Chemoradiation Therapy (cCTRT) +/- Induction Chemotherapy (ICT). The patients were divided into two groups according to the nodal disease stage: Group A with limited (N1-N2b) and group B (N2c-N3) with extensive nodal involvement. Prescribed doses and Treatment volumes extension were determined according to international guidelines. Data concerning time of treatment, interruptions, dose prescriptions, sites of progression, and histopathological features were collected.
Results: A total of 107 eligible patients were collected. Median age was 68.6 years (range 53-78 years). Median time of follow-up 36.7 months (range 12-60 months).
Group A included 79 patients; of these, 62.02% (49) achieved a complete response, 12.66% (10) showed no response to treatment; 25.32% (20) showed loco-regional progression. Median End of Treatment Delay (EoTD) for patients experiencing recurrences was 9.05 days, being > 5 days in 85% (17) of them.
Group B included 28 patients. Of these, only 14.29% (4) achieved a complete metabolic response. 21.43% (6) showed no response to treatments. 60.71% (17) showed loco-regional progression.
Median EoTD for these patients experiencing pathologic relapses was 7.4 days, being > 5 days in 71.43% (20) of them. LC for all patients was 49.53% (53). Cumulative 1 year-PFS was 71.1%. Cumulative 3 years-PFS was 55.4%.
The PFS analysis was also performed in relation to patients’ HPV status confirming worse clinical outcomes in those with HPV-unrelated tumors.
Conclusion: This study shows the role of a correctly timed radiotherapy. It was also confirmed that patients, not achieving complete remission, experience disease progression in sites located outside the areas where higher radiation doses were delivered, but still included in the treatment field. Thus, patients with tumor progression after chemoradiation should be included in clinical trials testing immunotherapy, alone or combined with chemotherapy.
Introduction
Head and Neck squamous cell carcinoma is the seventh most common tumor worldwide, being responsible of roughly 4.5% of cancer diagnoses and death [1]; it may involve different sites such as oral cavity, oropharynx hypopharynx, nasal cavity, nasopharynx and salivary glands.
Risk factors include alcohol and tabagism, Epstain Barr Virus (EBV), notoriously a negative prognostic factor for nasopharyngeal carcinoma [2] and Human Papilloma Virus (HPV), with HPV related tumors being more common in developed nations, confirming the importance of vaccinations on both genders [3] from an age of 11-12 and educational campaigns in favor of secure sexual practices.
Tumor staging generally implies the patient’s clinical examination, ORL assessment, radiological staging through MRI with gadolinium, Head-Neck-Thorax CT scan with iodine contrast enhancement [4] and 18F-FDG CT-PET scan. Treatment includes surgery for resectable, not-locally advanced tumors, Radiotherapy, as an exclusive approach for non-resectable tumors or as an adjuvant treatment within 4-6 weeks from surgery [5] for all the other cases, and chemotherapy, which may be employed in patients with locally advanced disease as a neoadjuvant approach for patients not directly eligible for surgery, in order to reduce the tumor burden or as an adjuvant approach consisting of Induction Chemotherapy (ICT) and/or Concurrent Chemoradiation (cCTRT).
Tumor recurrences and the importance of patients’ stratification
Despite the many different approaches at our disposal, local and nodal recurrences represent a critical aspect in the management of these patients, given the extreme biological and histopathological variability of these tumors.
Haring CT, et al. [6], by collecting the data of 447 patients that were treated in their department between 2008 and 2019, confirmed that most of recurrences occur within 2 years after the end of treatment. Besides that, they also underlined that current surveillance guidelines do not consider patient- and tumor-specific risk factors (tumor subsites, stage, and HPV status), which greatly affect treatment response and patterns of failure.
HPV proved a great importance as a positive prognostic factor in patients with HPV related-tumors resulting in better clinical outcomes (especially for oropharyngeal ones), if compared with unrelated tumors [7,8], to the point that some even proposed treatments de-intensification strategies for HPV positive patients [9]. The RTOG 1016 trial [10] compared the outcome of patients with HPV-related oropharyngeal cancer at stage III–IV (T1-2 N2-3 or T3-4 N0-3, according to the AJCC, 7th edition) treated with cisplatin or cetuximab; as expected, patients treated with cetuximab experienced lower grade toxicities. Unluckily, the study also showed that patients treated with cisplatin were associated with fewer failures in the radiation field than those treated with cetuximab.
Even though the de-intensification of the treatments showed contrasting results, it’s undeniable that a more accurate stratification of the patients according to the tumor biological features could help us establish the probability of eventual recurrences.
With the same intention, we tried to analyze the potential prognostic impact of factors more related to the therapy than to the pathology itself, as the time needed to carry out the RT course, which be different even for patients with the same kind of pathology, being it influenced by logistic factors, radio- and/or chemotherapy-related toxicities, and patients’ adherence, which other studies [11] proved to be also influenced by not tumor-related factors (i.e. socioeconomic ones). Specifically, we tried to determine if longer times of treatment could be associated with a worsening of patients’ outcome, in relation to the features of the disease (stage, involved sites, molecular expression and regimen of chemotherapy), by evaluating patients’ Progression Free Survival.
Materials and Methods
Inclusion criteria
We retrospectively analyzed NMHNSCC patients treated in our department from January 2014 to December 2023. The patients that were included in the study met the following criteria: age >18 years, availability of 18F-FDG CT/PET before and after the treatment [12], locally advanced (T3-4 N+) disease, treatment with adjuvant or exclusive cCTRT +/- iCT. They were divided into two groups according to the nodal disease stage: Group A with limited (N1-N2b) and Group B with extensive nodal disease (N2c-N3) (Table 1).
Radiotherapy Protocol: set-up, planning and treatment schedule
Radiotherapy was carried out, after proper set-up with CT scan and 5-point head, neck and shoulder thermoplastic mask for the immobilization of patients during the treatment [13]. Deformable Image Registration (DIR) of the centering CT images with those of 18F-FDG CT-PET (and MRI scans, when available) was also performed [14].
Intensity Modulated Radiation Therapy (IMRT) was carried out using an Elekta Synergy® Platform LINAC (linear accellerator), 6MV photons, daily Cone Beam CT (CBCT) to verify the patients’ setup [15] and the following prescription doses to the clinical target volumes, corresponding to those areas associated with variable risk of tumor relapse [16]: Low risk Clinical Target Volume (CTV1) receiving 54 Gy (1.8 Gy/fraction), intermediate risk Clinical Target Volume (CTV2) 60-62 Gy (2-2.07 Gy/fraction), gross nodal volume (GNV) corresponding to pathologically involved nodes receiving 63-66 Gy (2.1-2.2 Gy/fraction), Gross Tumor Volume (GTV) 66-69 Gy (2.2-2.3 Gy/ fraction).
Treatment volumes extension was decided according to DAHANCA guidelines [17].
Chemoradiation regimens
All the patients underwent cCTRT with one of the following schedules, in relation to age, comorbidities and tumor burden [18]: weekly Cisplatin (40 mg/mq for 6 cycles) or 3-weekly Cisplatin (100 mg/mq for 3 cycles).
ICT with TPF (docetaxel/cisplatin/5-FU) regimen was administered before cCTRT for those patients with HPV-unrelated Oropharyngeal Cancer (OPC) or EBV-related Nasopharyngeal Cancer (NPC), CT-PET detected high tumor burden and/or bilateral nodal involvement [19,20]. Weekly biochemical controls (for eventual CT-related toxicities) were performed.
Endpoints
For each patient, we collected the data regarding the time of treatment, eventual treatment interruptions and/or suspensions, prescribed doses to the target volumes, the sites of disease progression and tumor’s histopathological features.
Results
Starting from an initial number of 336 patients, 107 eligible patients were selected. Median Male/ female patients ratio was 3:1. Patients’median age was 68.6 years (range 53-78 years). Median time of follow-up was 36.7 months (range 12-60 months).
Group A included 79 patients with N1-N2b nodal disease; End of Treatment Delay (EoTD), which is expressed by supplementary days required to carry out the treatment beyond an Overall Treatment Time (OTT) of 41 days (corresponding to a 6-weeks treatment schedule), ranged between 0 and 28 days; of these, 62,02% (49) achieved a complete metabolic response as showed by post-RT FDGPET scan, 12,66% (10) showed no response to treatment with 4 of them with oropharyngeal HPV unrelated tumors; 25,32% (20) experienced loco-regional progression showing pathology relapse inside the radiotherapy field, including 8 with HPV-unrelated oral cavity/oropharyngeal cancers and 1 with nasopharyngeal EBV-related cancer. Median dose delivered to the areas of recurrences was 56.71 Gy (range 54-63 Gy). Median EoTD for Group A patients experiencing recurrences was 9.05 days, being > 5 days in 85% (17) of them.
Group B included 28 patients with N2c-N3 nodal disease. Of these, only 14.29% (4) achieved a complete metabolic response, all of them without histological risk factors related to HPV/EBV status. 21.43% (6) showed no response to treatments including 3 patients with HPV-unrelated oropharyngeal cancers and 1 with EBV-related nasopharyngeal ones. 60.71% (17) showed locoregional progression in sites with median delivered. Radiation doses of 61.16 Gy (range 52-65.3 Gy). No updated data on the disease status are available for one patient (3.57%) of this group. Median EoTD for Group B patients experiencing recurrences was 7.4 days, being > 5 days in 71.43% (20) of them.
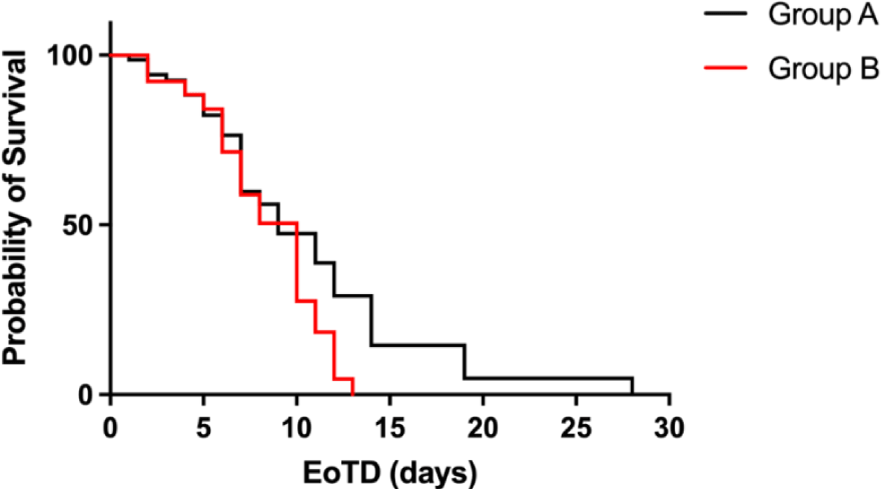
We then evaluated PFS in relation to EoTD in Group A and B; for both groups, EoTDs > 10 days are associated with Expected PoS reduction of 50% (Figure 1).
No meaningful differences were observed in terms of radio-/chemo-induced toxicities between the two groups.
LC for all patients was 49.53% (53), including those who obtained a complete response or disease chronicity after chemoradiation (specifically, one for each group).
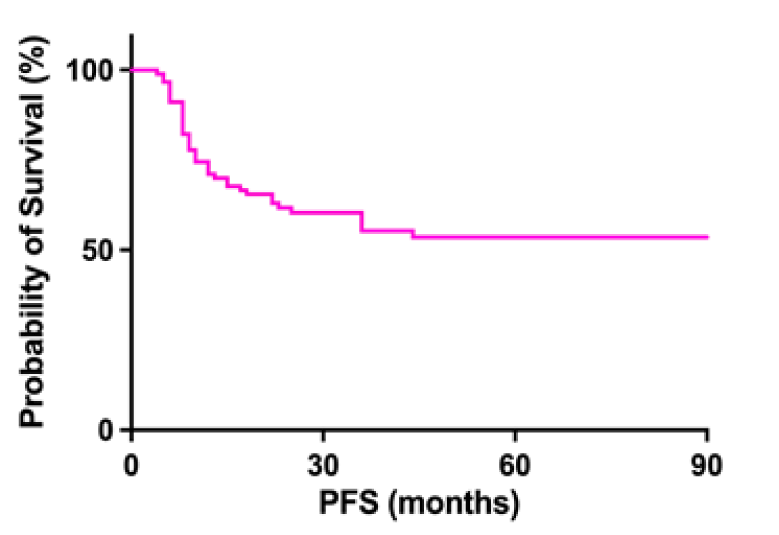
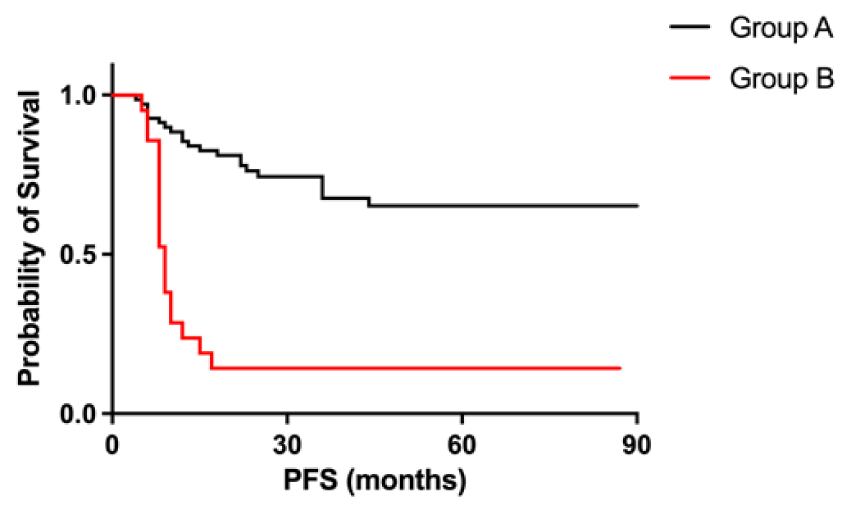
Cumulative 1 and 3 year-PFS were respectively 71.1% and 55.4% (Figure 2). 1 and 3 year-PFS respectively were 85.5% and 67.6% for Group A and 23.8% and 14.3% for Group B (Figure 3). Expected Probability of Survival (PoS) was 30% at 10 months in high risk patients vs., 90% in low risk patients.
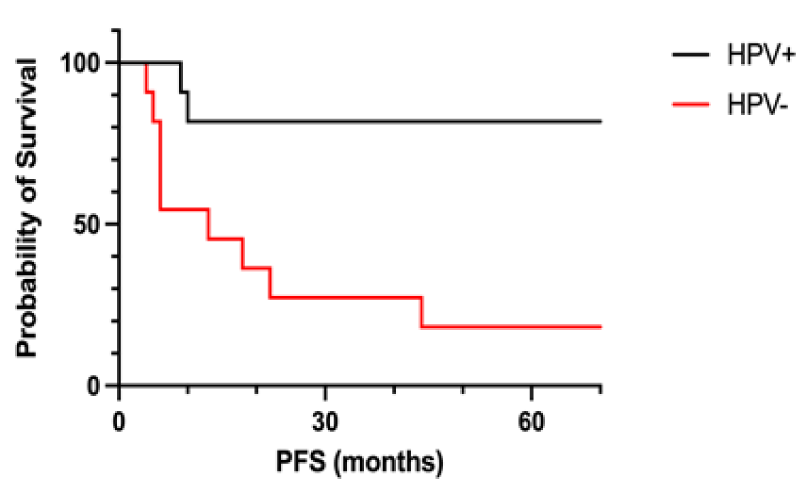
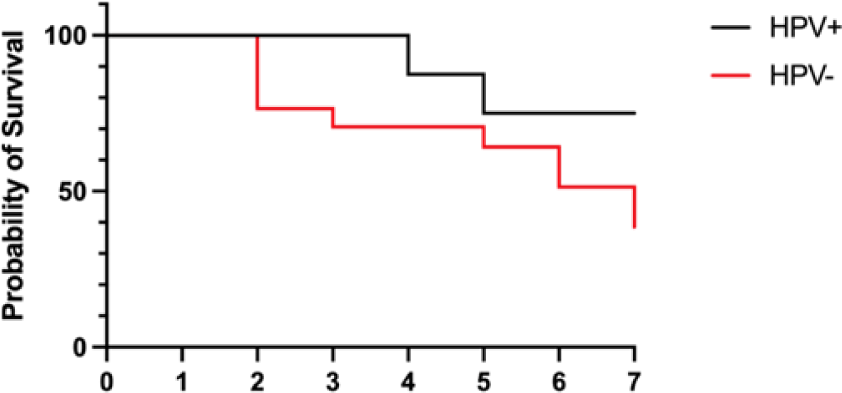
Furtherly, we analyzed PFS in patients with oral cavity/oropharyngeal cancer in relation to HPV Status: 1-year PFS was 81.8% for HPV+ and 45.4% for HPV- patients (Figure 4). Expected 1-year PoS was 50% for HPV- patients (vs., 82% for HPV+ patients).
Expected PoS with EoTD = 2 days was 77% in HPV- patients (vs., 100% in HPV+ patients) and EoTD = 6 days was 50% in HPV- patients (vs., 75% in HPV+ patients) (Figure 5).
Discussion
The results we obtained confirmed the importance of a stratification of the patients in relation to tumor staging and biological features.
As showed in literature [21], approximately 50% of the patients with locally advanced NMHNSCC is bound to experience recurrences. Our analysis confirmed this result (LC 49.5%), leading us to a critical point: the need of making a correct stratification of patients with head neck cancers, right from diagnosis with the possibility of developing predictive algorithms for the prognostic outcomes of these patients.
Ang KK, et al. [22], in the RTOG 0129 study retrospectively analyzed 721 patients with IIIIV stage head and neck cancer and found a 3-year PFS of 73.7% and 43.4% in patients with HPV related and unrelated tumors, respectively (vs., 81.8% and 27.3% we actually obtained). The difference in the PFS values of the two studies could be related to the important numeric gap between the two populations (743 vs., 107) and the non-homogeneous management of the patients (different radiotherapy fractionations and chemotherapy regimens), still representing the only extensive study with these aims available in literature, for this specific class of patients.
Still, in both cases, HPV-unrelated cancers were associated with lower values in terms of PFS if compared with HPV-related ones.
This study, in concordance with prior analyses [23,24], also showed that a correctly delivered radiotherapy with no or few delays was associated with better outcomes in patients with locally advanced disease, being the first one to determine, a time cut-off beyond which an increase of recurrences can be observed in patients undergoing homogenous treatment courses in terms of administered doses and length of treatment schedule. It was also the first study to evaluate the administered dose to the sites of recurrences in this class of patients, with HPV-unrelated tumors being associated with worse results in terms of PFS, with EoTD ≥ 2 days.
The high incidence of recurrences proves that these tumors are not to be underestimated. Therefore, further therapeutic strategies should be considered.
The patients experiencing tumor progression, not having other therapeutic options, could benefit from the inclusion in clinical trials [25] concerning the employment of chemotherapy and/or immunotherapy, with the latter requiring the analysis of the tumor’s molecular profile, that should be preventively performed right after diagnosis, in order to reduce the time required for the start of the therapy (given the high growth rate of these tumors).
Conclusion
This study shows the role of a correctly timed radiotherapy without long delays, which could compromise the patients’ clinical outcome, being worse in patients with HPV-unrelated disease and high tumor burden. It was also confirmed that patients, not achieving complete remission, experience disease progression in sites located outside the GTV/GNV, but still included in the low/ intermediate risk CTVs (CTV1/2). Given the lack of further treatment options, patients undergoing tumor progression after chemoradiation should be included in clinical trials implying the use of immunotherapy, alone or combined with chemotherapy.
References
- Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021 May;71(3):209-249. doi: 10.3322/caac.21660. Epub 2021 Feb 4. PMID: 33538338.
- Alami IE, Gihbid A, Charoute H, Khaali W, Brahim SM, Tawfiq N, Cadi R, Belghmi K, El Mzibri M, Khyatti M. Prognostic value of Epstein-Barr virus DNA load in nasopharyngeal carcinoma: a meta-analysis. Pan Afr Med J. 2022 Jan 3;41:6. doi: 10.11604/pamj.2022.41.6.28946. PMID: 35145598; PMCID: PMC8797042.
- El Hussein MT, Dhaliwal S. HPV vaccination for prevention of head and neck cancer among men. Nurse Pract. 2023 Oct 1;48(10):25-32. doi: 10.1097/01.NPR.0000000000000099. PMID: 37751612.
- Wippold FJ 2nd. Head and neck imaging: the role of CT and MRI. J Magn Reson Imaging. 2007 Mar;25(3):453-65. doi: 10.1002/jmri.20838. PMID: 17279529.
- Sun K, Tan JY, Thomson PJ, Choi SW. Influence of time between surgery and adjuvant radiotherapy on prognosis for patients with head and neck squamous cell carcinoma: A systematic review. Head Neck. 2023 Aug;45(8):2108-2119. doi: 10.1002/hed.27401. Epub 2023 May 16. PMID: 37194205.
- Haring CT, Kana LA, Dermody SM, Brummel C, McHugh JB, Casper KA, Chinn SB, Malloy KM, Mierzwa M, Prince MEP, Rosko AJ, Shah J, Stucken CL, Shuman AG, Brenner JC, Spector ME, Worden FP, Swiecicki PL. Patterns of recurrence in head and neck squamous cell carcinoma to inform personalized surveillance protocols. Cancer. 2023 Sep 15;129(18):2817-2827. doi: 10.1002/cncr.34823. Epub 2023 May 10. PMID: 37162461.
- Wookey VB, Appiah AK, Kallam A, Ernani V, Smith LM, Ganti AK. HPV Status and Survival in Non-Oropharyngeal Squamous Cell Carcinoma of the Head and Neck. Anticancer Res. 2019 Apr;39(4):1907-1914. doi: 10.21873 / anticanres.13299. PMID: 30952732.
- Rettig EM, D'Souza G. Epidemiology of head and neck cancer. Surg Oncol Clin N Am. 2015 Jul;24(3):379-96. doi: 10.1016/j.soc.2015.03.001. Epub 2015 Apr 9. PMID: 25979389.
- Vermorken JB, Psyrri A, Mesía R, Peyrade F, Beier F, de Blas B, Celik I, Licitra L. Impact of tumor HPV status on outcome in patients with recurrent and/or metastatic squamous cell carcinoma of the head and neck receiving chemotherapy with or without cetuximab: retrospective analysis of the phase III EXTREME trial. Ann Oncol. 2014 Apr;25(4):801-807. doi: 10.1093/annonc/mdt574. Epub 2014 Feb 26. PMID: 24577117; PMCID: PMC3969553.
- Gillison ML, Trotti AM, Harris J, Eisbruch A, Harari PM, Adelstein DJ, Jordan RCK, Zhao W, Sturgis EM, Burtness B, Ridge JA, Ringash J, Galvin J, Yao M, Koyfman SA, Blakaj DM, Razaq MA, Colevas AD, Beitler JJ, Jones CU, Dunlap NE, Seaward SA, Spencer S, Galloway TJ, Phan J, Dignam JJ, Le QT. Radiotherapy plus cetuximab or cisplatin in human papillomavirus-positive oropharyngeal cancer (NRG Oncology RTOG 1016): a randomised, multicentre, non-inferiority trial. Lancet. 2019 Jan 5;393(10166):40-50. doi: 10.1016/S0140-6736(18)32779-X. Epub 2018 Nov 15. Erratum in: Lancet. 2020 Mar 7;395(10226):784. doi: 10.1016/S0140-6736(20)30454-2. PMID: 30449625; PMCID: PMC6541928.
- Miller JL, Evers J. Barriers to Adherence to Cancer Treatments Among Head and Neck Cancer Patients. J Adv Pract Oncol. 2022 Jul;13(5):515-523. doi: 10.6004/jadpro.2022.13.5.5. Epub 2022 Jul 27. PMID: 35910497; PMCID: PMC9328454.
- Rettig EM, D’Souza G. Epidemiology of head and neck cancer. Surg Oncol Clin N Am. 2015;24:379-396. doi: 10.1016/j.soc.2015.03.001.
- Wong WL. PET-CT for Staging and Detection of Recurrence of Head and Neck Cancer. Semin Nucl Med. 2021 Jan;51(1):13-25. doi: 10.1053/j.semnuclmed.2020.09.004. Epub 2020 Oct 22. PMID: 33246535.
- Leech M, Coffey M, Mast M, Moura F, Osztavics A, Pasini D, Vaandering A. ESTRO ACROP guidelines for positioning, immobilisation and position verification of head and neck patients for radiation therapists. Tech Innov Patient Support Radiat Oncol. 2017 Jan 5;1:1-7. doi: 10.1016/j.tipsro.2016.12.001. PMID: 32095536; PMCID: PMC7033761.
- Ireland RH, Dyker KE, Barber DC, Wood SM, Hanney MB, Tindale WB, Woodhouse N, Hoggard N, Conway J, Robinson MH. Nonrigid image registration for head and neck cancer radiotherapy treatment planning with PET/CT. Int J Radiat Oncol Biol Phys. 2007 Jul 1;68(3):952-7. doi: 10.1016/j.ijrobp.2007.02.017. Epub 2007 Apr 18. PMID: 17445999; PMCID: PMC2713594.
- Den RB, Doemer A, Kubicek G, Bednarz G, Galvin JM, Keane WM, Xiao Y, Machtay M. Daily image guidance with cone-beam computed tomography for head-and-neck cancer intensity-modulated radiotherapy: a prospective study. Int J Radiat Oncol Biol Phys. 2010 Apr;76(5):1353-9. doi: 10.1016/j.ijrobp.2009.03.059. Epub 2009 Jun 18. PMID: 19540071.
- Merlotti A, Alterio D, Vigna-Taglianti R, Muraglia A, Lastrucci L, Manzo R, Gambaro G, Caspiani O, Miccichè F, Deodato F, Pergolizzi S, Franco P, Corv R, Russi EG, Sanguineti G; Italian Association of Radiation Oncology. Technical guidelines for head and neck cancer IMRT on behalf of the Italian association of radiation oncology - head and neck working group. Radiat Oncol. 2014 Dec 29;9:264. doi: 10.1186/s13014-014-0264-9. PMID: 25544268; PMCID: PMC4316652.
- Jensen K, Friborg J, Hansen CR, Samsøe E, Johansen J, Andersen M, Smulders B, Andersen E, Nielsen MS, Eriksen JG, Petersen JBB, Elstrøm UV, Holm AI, Farhadi M, Morthorst MH, Skyt PS, Overgaard J, Grau C. The Danish Head and Neck Cancer Group (DAHANCA) 2020 radiotherapy guidelines. Radiother Oncol. 2020 Oct;151:149-151. doi: 10.1016/j.radonc.2020.07.037. Epub 2020 Aug 8. PMID: 32781011.
- Melotek JM, Cooper BT, Koshy M, Silverman JS, Spiotto MT. Weekly versus every-three-weeks platinum-based chemoradiation regimens for head and neck cancer. J Otolaryngol Head Neck Surg. 2016 Nov 24;45(1):62. doi: 10.1186/s40463-016-0175-x. PMID: 27881143; PMCID: PMC5121964.
- Vermorken JB. Where and when to use induction chemotherapy in head and neck squamous cell cancer. In: Vermorken JB, Budach V, Leemans CR, Machiels JP, Nicolai P, O’Sullivan B, editors. Critical issues in head and neck oncology. Springer: Cham; 2021. doi: 10.1007/978-3-030-63234-2_11.
- Jiang YT, Chen KH, Liang ZG, Yang J, Qu S, Li L, Zhu XD. A Nomogram to Identify the Optimal Candidates for Induction Chemotherapy in Advanced N-Stage Nasopharyngeal Carcinoma. Cancer Manag Res. 2022 Aug 31;14:2583-2596. doi: 10.2147/CMAR.S377731. PMID: 36068822; PMCID: PMC9441150.
- Ang KK, Harris J, Wheeler R, Weber R, Rosenthal DI, Nguyen-Tân PF, Westra WH, Chung CH, Jordan RC, Lu C, Kim H, Axelrod R, Silverman CC, Redmond KP, Gillison ML. Human papillomavirus and survival of patients with oropharyngeal cancer. N Engl J Med. 2010 Jul 1;363(1):24-35. doi: 10.1056/NEJMoa0912217. Epub 2010 Jun 7. PMID: 20530316; PMCID: PMC2943767.
- Fesinmeyer MD, Mehta V, Blough D, Tock L, Ramsey SD. Effect of radiotherapy interruptions on survival in medicare enrollees with local and regional head-and-neck cancer. Int J Radiat Oncol Biol Phys. 2010 Nov 1;78(3):675-81. doi: 10.1016/j.ijrobp.2009.08.004. Epub 2010 Feb 3. PMID: 20133084.
- Thomas K, Martin T, Gao A, Ahn C, Wilhelm H, Schwartz DL. Interruptions of Head and Neck Radiotherapy Across Insured and Indigent Patient Populations. J Oncol Pract. 2017 Apr;13(4):e319-e328. doi: 10.1200/JOP.2016.017863. Epub 2017 Mar 7. Erratum in: J Oncol Pract. 2017 Jul;13(7):466. doi: 10.1200/JOP.2017.024190. PMID: 28267393.
- Rao YJ, Goodman JF, Haroun F, Bauman JE. Integrating Immunotherapy into Multimodal Treatment of Head and Neck Cancer. Cancers (Basel). 2023 Jan 21;15(3):672. doi: 10.3390/cancers15030672. PMID: 36765627; PMCID: PMC9913370.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.