General Science. 2024 August 16;5(8):983-988. doi: 10.37871/jbres1978.
Numbness of the Left Leg Caused by Perfusional Deficit in Right Parietal Lobe in a Case of Whiplash-Associated Disorder: Comparison of Imaging Tools for Diagnosis
Shan-Wen Lui1, Kar-Wai Lui2,3, and Shin-Tsu Chang4,5*
2Department of Medical Imaging and Intervention, Chang Gung Memorial Hospital, Linkou, Taoyuan, Taiwan
3Department of Medical Imaging and Radiological Sciences, College of Medicine, Chang Gung University, Taoyuan, Taiwan
4Department of Physical Medicine and Rehabilitation, Kaohsiung Veterans General Hospital, Kaohsiung, Taiwan
5Department of Physical Medicine and Rehabilitation, Tri-Service General Hospital, Taipei, School of Medicine, National Defense Medical Center, Taiwan
- Whiplash-associated disorder
- Numbness
- Parieto-occipital hypoperfusion
- Scintigraphy rehabilitation
- Nociceptive-vascular hypothesis
Abstract
Background: Since Whiplash-Associated Disorder (WAD) causes severe pain and even disability among patients, early diagnosis and therapy are essential to reduce personal and social costs. Traditionally, the diagnosis of WAD is mainly conducted through the examination of clinical symptoms and anatomical imaging with Computed Tomography (CT) and Magnetic Resonance Imaging (MRI) but not Single-Photon Emission Computed Tomography (SPECT).
Case description: A 24-year-old woman reported having mild headaches and progressive left leg numbness six months after experiencing a whiplash injury during a rear-end motor vehicle crash. MRI, CT, and electromyographic examination of her left leg were all negative, but SPECT of her brain showed decreased activity in the right parieto-occipital lobes.
Conclusion: Our case highlights (1) the clinical significance of hypoperfusion in the parietal lobe inducing leg numbness instead of nerve dysfunction or nerve-to-muscle signal transmission, (2) the evolving usefulness of functional imaging patterns of SPECT, and (3) the concordance between clinical WAD symptoms and the functional imaging findings. We suggest that patients with WAD need study of nuclear scintigraphy of the brain for a more precise diagnosis.
Introduction
Whiplash-Associated Disorder (WAD) is commonly found among motorists who experience a motor vehicle crash. Recent publications show that approximately 30% of patients with WAD remain moderately to severely disabled [1,2], and up to 50% of patients never completely recuperate [3]. Because of the lack of specialized electrophysiological studies to diagnose WAD, patients with less or none grumble of pain or paresthesia or any radiologic substantiation of injury might be overlooked, resulting in substantial personal and social costs, including disability, work failure, and medical expenses [4].
The diagnosis of WAD mainly relies on clinical symptoms including headache, visual disturbances, neck pain or stiffness, memory impairment, concentration problems, arm pain or paresthesia or both, and psychological distress, as well as radiographic imaging studies to detect anatomical abnormalities [5]. Although Single-Photon Emission Computed Tomography (SPECT) is not routinely used to diagnose WAD, recent SPECT studies have revealed brain hypoperfusion in patients with WAD [6,7]. The use of SPECT to link brain hypoperfusion to the clinical symptoms of a patient with a functional neurogenic disorder has been reported [8].
We report a patient with WAD for whom we used SPECT imaging of the brain to make the diagnosis, which brain hypoperfusion was compatible with the clinical symptoms.
Case description
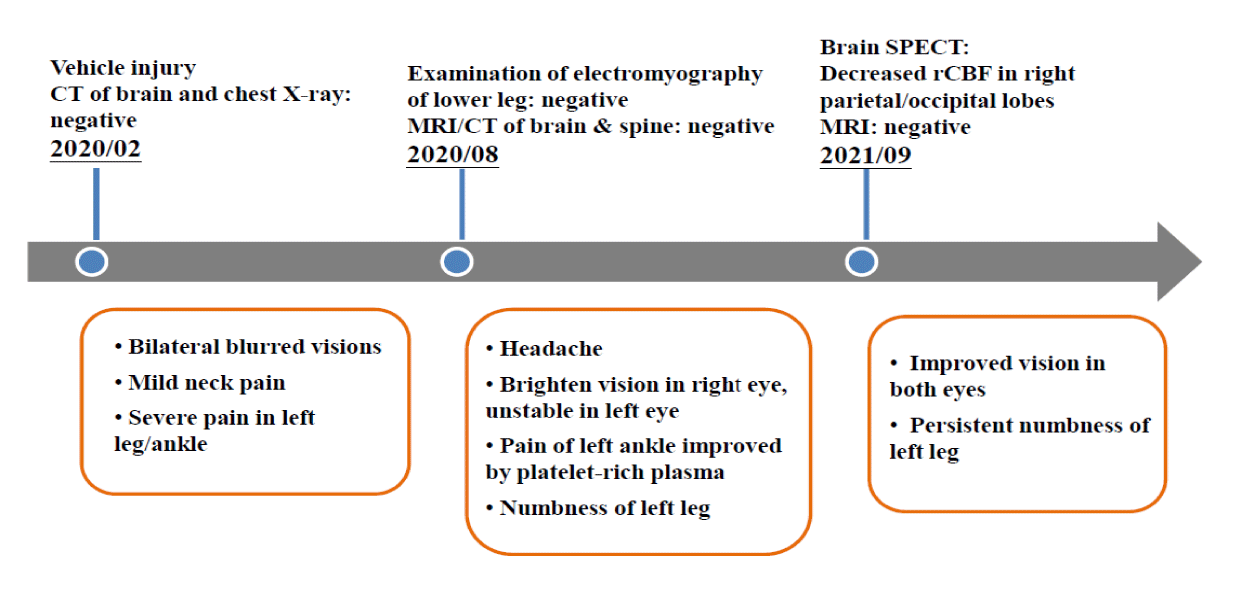
A 24-year-old female motorcyclist experienced a rear-end motorcycle-car crash four years previously. The motorcycle compressed her left ankle, causing severe lower leg pain and difficult walking. Mild neck pain and bilateral blurred visions also bothered her.
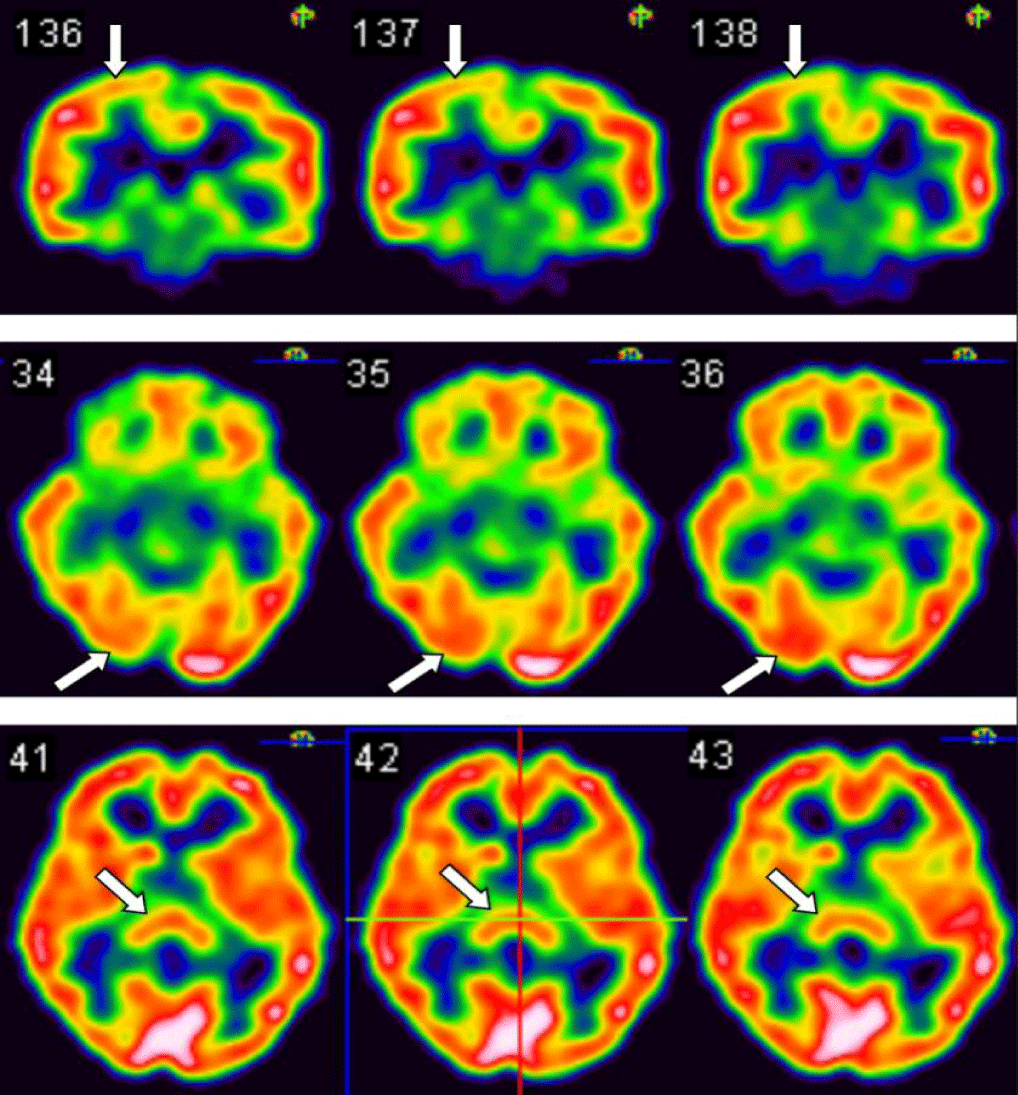
After six months of conservative treatment plus ankle injection, her left ankle pain and disability still persisted. Although her neck pain and blurred visions regressed, she developed new symptoms of headache and progressive left leg numbness. She was unable to stand, so she sought help from our hospital clinic. She could fluently and logically communicate with her physicians. MRI, CT, and electromyographic images of the spine and legs were negative for nerve dysfunction and abnormal nerve-to-muscle signal transmission as the possible causes of leg numbness. Injection therapy with platelet-rich plasma in her left ankle was given twice, and her ankle pain was relieved. The patient was discharged, and follow-up rehabilitation was prescribed. Nonetheless, the patient returned six months later, still reporting of left leg numbness. We examined the 99 mTc-Ethylene-Diylbis-L-Cysteine Diethyl Ester (ECD) SPECT images of her brain, which showed normal in the bilateral thalami, but obvious decreased perfusion in the right parietal (superior region) and right occipital lobes (Figure 1). As timeline shown in figure 2, the examination with CT and X-ray of multiple sites showed no destructive damage.
Discussion
In this case as shown on SPECT, we found the association not only between the patient’s bilateral blurred visions and occipital lobe hypoperfusion, but also hypoperfusion in the right parietal lobe and left leg numbness.
The patient reported numbness in the left leg. EMG findings were negative for nerve lesions, muscle dysfunction, and problems with nerve-to-muscle signal transmission, which conclusively excluded dysfunction of the left sciatic nerve. Sterling M, et al. [9] had shown that when patients who have had persisted moderate to severe warning signs within 6 months of WAD that some brain changes in mechanisms regarding central pain processing could possibly cause hypersensitivity symptoms. Although our patient had similarly moderate symptoms and injury to the central nervous system, she developed hyposensitivity in her leg. Due to hypoperfusion in the right parietal region on SPECT, psychological factors of disabilities were definitively excluded. Based on the above findings and those of previous studies [7,9], we conclude that the decreased blood flow in the parietal region caused by WAD affected the neurogenic pathway, which resulted in constant numbness of the left leg. A comparison between the patients reported by Sterling M, et al. [9] and our patient was shown in table 1.
| Table 1: Summary of the patient’s clinical characteristics and a published study of WAD. | |||||
| Severity | Injured area involved 6 months post-injury | Symptom | Precise anatomical structure | Psychological distress | |
| Our case | Moderate | Central nervous system | Hyposensitivity to pressure, pain and touch | SPECT image revealed hypoperfusion in the right parieto-occipital region | Definitively excluded |
| Sterling, et al. [9] | Moderate to severe | Central pain processing mechanisms | Hypersensitivity to pressure, pain, and temperature | No images presented | Unable to exclude |
| WAD: Whiplash-Associated Disorders; SPECT: Single-Photon Emission Computed Tomography. | |||||
Discomforts from patient with WAD are often ignored, possibly due to the difficulty in detecting abnormalities with CT/MRI and the fact that neuropsychological disturbances following whiplash are typically borderline [10]. These disturbances often present as non-specific symptoms, for instance, concentration and memory disturbances, which are common in other central disorders [11]. Although MRI offers brilliant soft tissue contrast and shuns ionizing radiation exposure, and CT provides detailed images of bone or hard structures, both modalities are limited in detecting functional changes in the brain, which reduces their utility in this aspect. Poor correlation between MRI and clinical manifestation of WAD has been reported by Karlsborg M, et al. [12], who concluded the prognosis of WAD is also not always associated with post-injury MRI [13]. In contrast, using SPECT can earlier detect cerebral blood flow abnormalities, allowing for timely interventions such as intravenous laser therapy to enhance the oxygen-carrying capacity and flexibility of red blood cells [14,15].
As a neuroimaging tool, SPECT allows for a better evaluation of brain functional status than other imaging tools. SPECT demonstrates the pathophysiological changes of neurological disorders that are so-called psychodynamic lesions [8] that correlate with clinical symptoms for making an accurate diagnosis, as with our patient. In the same way, in patients with dementia with Lewy bodies, significant decreased perfusion has been found through ECD-SPECT in frontal, parietal, temporal, and thalamus regions compared to the control group [16]. ECD-SPECT can be a helpful tool to help us understand the underlying mechanisms of WAD by providing functional insights into physiological processes and biological pathways. For lesions with cerebral blood flow alterations, SPECT can play a role in monitoring the brain plasticity. WAD emerges to be of mild to moderate injury intensity, and commonly occurs in the occipital lobe amid other brain areas [17]. Imaging with SPECT is essential in detecting acute lesions of a WAD [18], but might also play a role in the logistic ratiocination of unrelieved pathology.
Regional perfusion alterations in the brains of patients with WAD have been reported [7,19]. In a comprehensive review of WAD [7], the production of hypoperfusion in the parieto-occipital region was attributed to the nociceptive-vascular hypothesis. Vasoconstriction of brain vessels occurs with increased vasopeptide production triggered by nociceptive afferent nerves origininated from the upper cervical spine, which is under chronic stress and pain. These publications and concepts enhanced our understanding that in our patient’s case, hypoperfusion in the parieto-occipital lobe correlated with WAD.
Brain SPECT-CT is an imaging technique by using hybrid method in scintigraphic rehabilitation that merges SPECT and CT imaging to deliver detailed significance regarding the combination of the function and structure of the brain [20-33]. It is a great clinical tool to detect cerebral blood flow impairment and differentiate an ischemic event after stroke onset [34,35].
Finally, there are still some shortcomings in the diagnosis of WAD, and the connection between symptoms and mechanisms remains unclear, necessitating further research on neuroimaging techniques in WAD. For instance, the hybrid images of SPECT and MRI might help diagnose soft tissue injuries in patients with WAD, which has not yet been studied. Additionally, functional MRI, which can detect blood oxygen levels and produce images, aids in indirectly measuring neural activity and mapping the affected brain regions [36]. Its role in diagnosing WAD has not been fully recognized. With these diagnostic tools, combined with brain SPECT-CT, we might be able to further validate the nociceptive-vascular hypothesis and elucidate the underlying mechanisms of the disease. A comparison of the characteristics for MRI,CT, and SPECT-CT was made in table 2.
| Table 2: A comparison of the characteristics for MRI, CT, and SPECT-CT in brain pathology. | |||
| MRI | CT | SPECT-CT | |
| Principle | Using magnetic fields and radiofrequency pulses to produce images | Using computerized X-ray imaging from multiple angles to create images | Hybrid images with combination of SPECT and CT |
| Anatomical accuracy | Excellent | Excellent | Excellent |
| Soft tissue contrast | High | Low | Moderate |
| Radiation exposure | None | Moderate | Moderate |
| Functioning imaging | Absent | Absent | Simultaneous inspection of brain structure and corresponding location in the SPECT, but not real time |
| Cost | Most expensive | Less expensive than MRI | Less expensive than MRI |
| Manpower consumption | Low | Low | Moderate |
In conclusion, our report provides insight into the evolving functional imaging patterns that can be seen with SPECT in contrast to anatomical images like those produced by CT and MRI. Additionally, we have shown that hypoperfusion in the parietal lobe can induce leg numbness instead of nerve dysfunction or nerve-to-muscle signal transmission, and most importantly, we have shown concordance between clinical WAD symptoms and the SPECT-CT functional imaging findings. We suggest that those patients with WAD and clinical symptoms discordant to anatomical image findings should undergo nuclear scintigraphy of the brain for a more precise diagnosis.
References
- Rebbeck T, Sindhusake D, Cameron ID, Rubin G, Feyer AM, Walsh J, Gold M, Schofield WN. A prospective cohort study of health outcomes following whiplash associated disorders in an Australian population. Inj Prev. 2006 Apr;12(2):93-8. doi: 10.1136/ip.2005.010421. PMID: 16595423; PMCID: PMC2564458.
- Sterling M, Jull G, Kenardy J. Physical and psychological factors maintain long-term predictive capacity post-whiplash injury. Pain. 2006 May;122(1-2):102-8. doi: 10.1016/j.pain.2006.01.014. Epub 2006 Mar 9. PMID: 16527397.
- Carroll LJ, Holm LW, Hogg-Johnson S, Côté P, Cassidy JD, Haldeman S, Nordin M, Hurwitz EL, Carragee EJ, van der Velde G, Peloso PM, Guzman J; Bone and Joint Decade 2000-2010 Task Force on Neck Pain and Its Associated Disorders. Course and prognostic factors for neck pain in whiplash-associated disorders (WAD): results of the Bone and Joint Decade 2000-2010 Task Force on Neck Pain and Its Associated Disorders. Spine (Phila Pa 1976). 2008 Feb 15;33(4 Suppl):S83-92. doi: 10.1097/BRS.0b013e3181643eb8. PMID: 18204405.
- Sterling M. Whiplash-associated disorder: musculoskeletal pain and related clinical findings. J Man Manip Ther. 2011 Nov;19(4):194-200. doi: 10.1179/106698111X13129729551949. PMID: 23115472; PMCID: PMC3201650.
- Yadla S, Ratliff JK, Harrop JS. Whiplash: diagnosis, treatment, and associated injuries. Curr Rev Musculoskelet Med. 2008 Mar;1(1):65-8. doi: 10.1007/s12178-007-9008-x. PMID: 19468901; PMCID: PMC2684148.
- Vállez García D, Doorduin J, Willemsen AT, Dierckx RA, Otte A. Altered Regional Cerebral Blood Flow in Chronic Whiplash Associated Disorders. EBioMedicine. 2016 Aug;10:249-57. doi: 10.1016/j.ebiom.2016.07.008. Epub 2016 Jul 14. PMID: 27444853; PMCID: PMC5006659.
- Otte A. Pathophysiological interrelated deactivation/activation processes in the exhausted brain after whiplash injury. Hell J Nucl Med. 2019 May-Aug;22(2):92-95. doi: 10.1967/s002449911000. Epub 2019 Jul 7. PMID: 31273349.
- Galli S, Aybek S, Chokron S, Moulin T, Magnin E. Modification of both functional neurological symptoms and neuroimaging patterns with a good anatomoclinical concordance: a case report. BMC Neurol. 2019 Nov 4;19(1):270. doi: 10.1186/s12883-019-1475-3. PMID: 31684891; PMCID: PMC6830004.
- Sterling M, Jull G, Vicenzino B, Kenardy J. Sensory hypersensitivity occurs soon after whiplash injury and is associated with poor recovery. Pain. 2003 Aug;104(3):509-517. doi: 10.1016/S0304-3959(03)00078-2. PMID: 12927623.
- Radanov BP, Di Stefano G, Schnidrig A, Sturzenegger M, Augustiny KF. Cognitive functioning after common whiplash. A controlled follow-up study. Arch Neurol. 1993 Jan;50(1):87-91. doi: 10.1001/archneur.1993.00540010081022. PMID: 8418806.
- Otte A, Ettlin TM, Nitzsche EU, Wachter K, Hoegerle S, Simon GH, Fierz L, Moser E, Mueller-Brand J. PET and SPECT in whiplash syndrome: a new approach to a forgotten brain? J Neurol Neurosurg Psychiatry. 1997 Sep;63(3):368-72. doi: 10.1136/jnnp.63.3.368. PMID: 9328255; PMCID: PMC2169690.
- Karlsborg M, Smed A, Jespersen H, Stephensen S, Cortsen M, Jennum P, Herning M, Korfitsen E, Werdelin L. A prospective study of 39 patients with whiplash injury. Acta Neurol Scand. 1997 Feb;95(2):65-72. doi: 10.1111/j.1600-0404.1997.tb00071.x. PMID: 9059723.
- Sarrami P, Armstrong E, Naylor JM, Harris IA. Factors predicting outcome in whiplash injury: a systematic meta-review of prognostic factors. J Orthop Traumatol. 2017 Mar;18(1):9-16. doi: 10.1007/s10195-016-0431-x. Epub 2016 Oct 13. PMID: 27738773; PMCID: PMC5311004.
- Mi XQ, Chen JY, Liang ZJ, Zhou LW. In vitro effects of helium-neon laser irradiation on human blood: blood viscosity and deformability of erythrocytes. Photomed Laser Surg. 2004 Dec;22(6):477-82. doi: 10.1089/pho.2004.22.477. PMID: 15684746.
- Wasik M, Gorska E, Modzelewska M, Nowicki K, Jakubczak B, Demkow U. The influence of low-power helium-neon laser irradiation on function of selected peripheral blood cells. J Physiol Pharmacol. 2007 Nov;58 Suppl 5(Pt 2):729-37. PMID: 18204188.
- Risacher SL, Saykin AJ. Neuroimaging biomarkers of neurodegenerative diseases and dementia. Semin Neurol. 2013 Sep;33(4):386-416. doi: 10.1055/s-0033-1359312. Epub 2013 Nov 14. PMID: 24234359; PMCID: PMC3975244.
- Anarte-Lazo E, Abichandani D, Rodriguez-Blanco C, Bernal-Utrera C, Falla D. Headache features in people with whiplash associated disorders: A scoping review. Musculoskelet Sci Pract. 2023 Aug;66:102802. doi: 10.1016/j.msksp.2023.102802. Epub 2023 Jun 18. PMID: 37357053.
- Schollaert J, Van Goethem JWM. Imaging in Whiplash-Associated Disorders. Semin Musculoskelet Radiol. 2023 Oct;27(5):512-521. doi: 10.1055/s-0043-1772170. Epub 2023 Oct 10. PMID: 37816359.
- Otte A, Ettlin T, Fierz L, Mueller-Brand J. Parieto-occipital hypoperfusion in late whiplash syndrome: first quantitative SPET study using technetium-99m bicisate (ECD). Eur J Nucl Med. 1996 Jan;23(1):72-4. doi: 10.1007/BF01736993. PMID: 8586106.
- Lai MH, Wang TY, Chang CC, Li TY, Chang ST. Cerebellar diaschisis and contralateral thalamus hyperperfusion in a stroke patient with complex regional pain syndrome. J Clin Neurosci. 2008 Oct;15(10):1166-8. doi: 10.1016/j.jocn.2007.06.017. Epub 2008 Aug 15. PMID: 18707887.
- Hsu KC, Chang ST. Correlation of thalamic blood flow redistribution with persistent complex regional pain syndrome in a stroke patient with poor diabetic control. J Diabetes Investig. 2013 Nov 27;4(6):676-8. doi: 10.1111/jdi.12104. Epub 2013 Jul 12. PMID: 24843725; PMCID: PMC4020266.
- Chang CC, Ku CH, Chang ST. Postural asymmetry correlated with lateralization of cerebellar perfusion in persons with chronic stroke: A role of crossed cerebellar diaschisis in left side. Brain Inj. 2017;31(1):90-97. doi: 10.1080/02699052.2016.1210229. Epub 2016 Nov 10. PMID: 27830942.
- Yang WH, Lin SP, Chang ST. Case report: Rapid improvement of crossed cerebellar diaschisis after intravascular laser irradiation of blood in a case of stroke. Medicine (Baltimore). 2017 Jan;96(2):e5646. doi: 10.1097/MD.0000000000005646. PMID: 28079797; PMCID: PMC5266159.
- He HC, Hsu MC, Hsu CS, Cheng YY, Chang ST. Bidirectionality of the dentato-rubro-thalamo-cortical tract allows concurrent hypoperfusion in ipsilateral cerebellum and contralateral cerebral hemisphere: A case report. Medicine (Baltimore). 2018 Oct;97(40):e12590. doi: 10.1097/MD.0000000000012590. PMID: 30290625; PMCID: PMC6200509.
- Chuang YC, Liu CC, Yu IC, Tsai YL, Chang ST. Shifting of global aphasia to Wernicke's aphasia in a patient with intact motor function: a case report. BMC Neurol. 2021 Mar 11;21(1):111. doi: 10.1186/s12883-021-02131-w. PMID: 33706719; PMCID: PMC7949244.
- Liu CC, Hsu CS, He HC, Cheng YY, Chang ST. Effects of intravascular laser phototherapy on delayed neurological sequelae after carbon monoxide intoxication as evaluated by brain perfusion imaging: A case report and review of the literature. World Journal of Clinical Cases 2021; 9(13):3048-3055.
- Li SA, Lin YP, Hsieh SP, Chang ST. Binary effects of intravascular laser irradiation of blood on motor recovery and homocysteine reduction in a case with ischemic hemiparesis: portrayed with brain perfusion images. BMC Neurol. 2022 Sep 27;22(1):370. doi: 10.1186/s12883-022-02896-8. PMID: 36162998; PMCID: PMC9513988.
- Lan CH, Wu YC, Chiang CC, Chang ST. Effects of intravascular photobiomodulation on motor deficits and brain perfusion images in intractable myasthenia gravis: A case report. World J Clin Cases. 2022 Aug 26;10(24):8718-8727. doi: 10.12998/wjcc.v10.i24.8718. PMID: 36157830; PMCID: PMC9453358.
- Chiang CC, Wu YC, Lan CH, Wang KC, Tang HC, Chang ST. Exploring CNS Involvement in Pain Insensitivity in Hereditary Sensory and Autonomic Neuropathy Type 4: Insights from Tc-99m ECD SPECT Imaging. Tomography. 2023 Dec 18;9(6):2261-2269. doi: 10.3390/tomography9060175. PMID: 38133079; PMCID: PMC10747491.
- Chu YC, Chang ST, Chan HY, Shen DHY, Chan HP. Abnormalities in regional cerebral blood flow due to headache in a COVID-19 infected patient observed on 99 mTC-ECD brain SPECT/CT. Reports. 2023;6(4):58. doi: 10.3390/reports6040058.
- Chen HH, Lin CY, Chen SJ, Huang WY, Kuo CW, Chang ST. Intravascular laser irradiation of blood as novel migraine treatment: an observational study. Eur J Med Res. 2023 Oct 25;28(1):457. doi: 10.1186/s40001-023-01438-3. PMID: 37876003; PMCID: PMC10598972.
- Lin YP, Ku CH, Chang CC, Chang ST. Effects of intravascular photobiomodulation on cognitive impairment and crossed cerebellar diaschisis in patients with traumatic brain injury: a longitudinal study. Lasers Med Sci. 2023 Apr 20;38(1):108. doi: 10.1007/s10103-023-03764-8. PMID: 37076743; PMCID: PMC10115718.
- Tanaka K, Yamada T, Torii T, Yoshimura T, Takase K, Togao O, Wakata Y, Hiwatashi A, Nakashima N, Kira J, Murai H. Pure dysarthria and dysarthria-facial paresis syndrome due to internal capsule and/or corona radiata infarction. BMC Neurol. 2015 Oct 7;15:184. doi: 10.1186/s12883-015-0439-5. PMID: 26445963; PMCID: PMC4597455.
- Masdeu JC, Brass LM. SPECT imaging of stroke. J Neuroimaging. 1995 Jul;5 Suppl 1:S14-22. doi: 10.1111/jon19955s1s14. PMID: 7626832.
- Laloux P, Richelle F, Jamart J, De Coster P, Laterre C. Comparative correlations of HMPAO SPECT indices, neurological score, and stroke subtypes with clinical outcome in acute carotid infarcts. Stroke. 1995 May;26(5):816-21. doi: 10.1161/01.str.26.5.816. PMID: 7740573.
- Elliott JM, Dayanidhi S, Hazle C, Hoggarth MA, McPherson J, Sparks CL, Weber KA 2nd. Advancements in Imaging Technology: Do They (or Will They) Equate to Advancements in Our Knowledge of Recovery in Whiplash? J Orthop Sports Phys Ther. 2016 Oct;46(10):862-873. doi: 10.2519/jospt.2016.6735. PMID: 27690836; PMCID: PMC7274526.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.