Medicine Group. 2024 August 21;5(8):996-1005. doi: 10.37871/jbres1980.
Age-Specific Survival in Lung Cancer in the Nordic Countries through a Half Century
Kari Hemminki1,7*, Frantisek Zitricky1, Asta Försti2,3, Otto Hemminki4,5, and Akseli Hemminki4,6
2Hopp Children's Cancer Center (KiTZ), Heidelberg, Germany
3Division of Pediatric Neurooncology, German Cancer Research Center (DKFZ), German Cancer Consortium (DKTK), Heidelberg, Germanyv 4Cancer Gene Therapy Group, Translational Immunology Research Program, University of Helsinki, Finland
5Department of Urology, Helsinki University Hospital, Helsinki, Finland
6Comprehensive Cancer Center, Helsinki University Hospital, Helsinki, Finland
7Division of Cancer Epidemiology, German Cancer Research Center (DKFZ), Im Neuenheimer Feld 580, D-69120, Heidelberg, Germany
- Age-groups
- Performance status
- Stage
- Conditional survival
- Surgery
Abstract
Background: Lung Cancer (LC) is often diagnosed at a late stage in relatively old persons whose survival may be compromised because of limited treatment. Our aim was to carry out an age-specific survival study on LC in Denmark (DK), Finland (FI), Norway (NO) and Sweden (SE) through a half century (1972-2021). Nordic survival data were compared with survival in the US Surveillance, Epidemiology and End Results (SEER) database
Methods: Relative 1- and 5-year survival data were extracted from the NORDCAN database and subjected to modelling by generalized additive models for survival trends over time. Conditional survival was calculated for those who had survived the first year to survive additional 4 years
Results: Very little improvement was found in survival for any age group before year 2000. By 2017-21, 5-year survival for men below age 50 years has increased to over 40% (FI somewhat less) and for equally aged women it was over 50% in countries other than FI. Survival reached over 20% for the 80-89-year-old male and female patients but it was 7% for men and 12% for women in FI. DK age-group-specific survival curves stayed well parallel to each other indicating equal improvement but for FI and SE men the curves diverged showing increasing age gaps. Nordic age-specific survival data were better than those in the US SEERS
Conclusion: Survival increased, after a 30-year standstill, after 2000 with increasing tempo towards 2017-21 and encompassing all age groups. This and the increase in conditional survival suggested that even the oldest patients were offered effective treatment. As LC remains a fatal disease all prevention modes needs to be emphasized, starting from primary prevention, to early detection and therapeutic improvements.
Introduction
Data from industrialized countries have shown that Lung Cancer (LC) mortality rates follow cigarette smoking prevalence with a lag times of 20-30 years [1-3]. Thus as the smoking epidemic in many countries commenced at around World War II the incidence of the then relatively rare LC started to climb to become a common cancer in the 1950s. Data from the Danish cancer registry, which is the oldest of its kind in the world, recorded an LC incidence (adjusted to the world population) of 10/100,000 for men and 3.5/100,000 for women in 1943; the rates were 5 times higher for men and 4 times higher for women 30 years later and kept on increasing [4]. The attributable fraction of cigarette smoking for LC has been estimated at > 80% in the industrialized countries [1]. There are also other known risk factors for LC, such as occupational exposures, air pollution, radon, obesity, type 2 diabetes and family history, and their contribution is most likely increasing as smoking prevalence deceases [5-9]. In Sweden with the lowest European LC incidence for men only 10% of male LC patients were never smokers in 2018-2022; overall 13.2% patients were never smokers, 35.2% were smokers and 51.4% were former smokers (https://cancercentrum.se/samverkan/cancerdiagnoser/lunga-och-lungsack/kvalitetsregister).
LC histology has changed over the years from the dominant squamous cell carcinoma (SCC), accounting for close the 40% of all in the Nordic countries in 1978-91, to an even more dominant adenocarcinoma, accounting for 55% in Sweden in 2021 [10]; the share for SCC has decreased to < 20% (20230921_nlcr_nationell_rapport2022.pdf (cancercentrum.se)). According to this report, the share of the clinically distinct small cell carcinoma has decreased in Sweden to 10%. Clinical stage at diagnosis was IV (metastatic) in > 60% of the patients in 1978-91 in the Nordic countries; in 2021 its share was 50% in Sweden while stage I had climbed to over 20% passing stage III (below 20%). In small cell LC stage IV has remained at over 70% (20230921_nlcr_nationell_rapport2022.pdf (cancercentrum.se)). Increasing detection of early stage LC has been achieved by more sensitive thorax Computed Tomography (CT) and Positron Emission Tomography (PET)/CT, and the patients may be amenable to minimal invasive surgery. Therapies for more advanced LC have improved and for metastatic disease all treatment modalities are applied including surgery, chemotherapy, chemoradiation, targeted therapy and most recently immunotherapy; the therapeutic choices are guided by appropriate laboratory data to be effective [10,11]. Small cell LC is a distinct disease which may only transiently respond to chemotherapy and radiation [11].
Survival in LC has increased globally but age differences are large in favor of young patients [12-18]. For example, a study covering 87 European cancer registries found a 5-year relative survival of 13% among all patients and an age-disadvantage [17]. In a recent international comparison even lower 5-year survival levels were reported for developing countries [18]. The age disparity, which is noted for many cancers, is explained in terms of late diagnoses, less active treatment and lower toleration of treatment among old patients because of frailty and comorbidities [14,17,19]. The influence of these factors on survival was illustrated in a large European study on LC [20]. However, stage distribution of LC at diagnosis does not show age differences which would suggest that stage may not explain the age differences [14,19]. Smoking may also influence survival in LC through interfering with radiation and other forms of therapy, at least in patients who continue smoking during their treatments [21-23].
We assess here age-specific relative survival in LC in the four Nordic countries of Denmark (DK), Finland (FI), Norway (NO) and Sweden (SE) through a half century from 1972 to 2021 based on the NORDCAN database. For background reference, overall LC survival in these countries was recently published [15]. These Nordic countries may be particularly interesting for LC survival analysis for at least three reasons: health care is guaranteed to the whole population, cancer data are of high quality over decades [24] and smoking prevalence has changed extensively between these populations (Supplementary Table 1) [25]. Shortly to the latter point, FI men were among the heaviest European smokers after World War II, later taken over by DK men while SE became the non-smoking champions [1,26,27] (www.pnlee.co.uk/ISS.htm). The epochal crossing of SE male and female lung cancer incidence rates took place in 2016 [28].
Methods
The data were obtained from NORDCAN database 2.0 [29,30]. The database was accessed at the International Agency for Cancer (IARC) website (https://nordcan.iarc.fr/en) [31], and the available tools were used to extract data on incidence, mortality and 1- and 5-year survival. NORDCAN uses International Classification of Diseases (ICD) version 10 codes for cancer.
Statistical modelling was performed using R statistical software (https://www.r-project.org) in the R studio environment (https://posit.co/). Trends in incidence and mortality were modelled by jointpoint regression log-normal models using the ‘segmented’ package [32,33]. Models with different number of knots were compared by Bayesian Information Criterion (BIC) and models with low BIC values were preferred.
Data on 1-year and 5-year relative survival were available from consecutive 5-year periods between 1972 and 2021. The survival estimates were based on ‘cohort approach’ for the first nine 5-year periods and ‘period approach’ for the last period (2017-2021), combining data from the previous 5-year period. The age-specific relative survival was estimated using the Pohar Perme estimator [29]. The national life tables were used to calculate the expected survival. The 5/1-year conditional survival was calculated as the ratio of the respective 5-year and 1-year relative survival estimates. The relative survival estimates were not available if less than 30 patients were alive in the beginning of follow-up.
The trends in relative survival were modelled using Bayesian generalized additive models as described before [34]. The modelling was performed on the cumulative hazard scale, permitting asymmetric Confidence Intervals (CIs) for the individual NORDCAN estimates. We fitted separate models for each metric and country. The models included the main effect of the group (combination of age-specific groups and sex) and non-linear effect of time, modelled with thin plate regression splines (mgcv package, k = 4). The female 60-69 year group was modelled as a reference level (intercept) with effects of the other groups having Gaussian prior (mean = 0, sigma = 1 for cumulative hazard associated with 1-year and 5/1-year relative survival; sigma = 2 for cumulative hazard associated with 5-year relative survival). For the purpose of modelling of temporal trends, the relative survival estimates for individual 5-year periods were assigned timepoint in the middle of the respective period. The CIs provided for survival estimates were used to model uncertainty in model input. The modelled trends were visualized after back-transforming from cumulative excess hazard scale to relative survival scale.
We further calculated 1st derivatives of temporal survival trends, corresponding to annual change in relative survival, to detect the periods when survival was changing with at least 95% plausibility (the associated 95% credible intervals did not cross zero in the sliding 5-year periods).
As a reference we obtain LC data from the US Surveillance, Epidemiology and End Results (SEER) web site for years 2016–20 for Whites and Hispanics (https://seer.cancer.gov/statistics-network/explorer/application.html?site=1&data_type=1&graph_type=2&compareBy=sex&chk_sex_3=3&chk_sex_2=2&rate_type=2&race=1&age_range=1&hdn_stage=101&advopt_precision=1&advopt_show_ci=on&hdn_view=0&advopt_display=2#graphArea).
Results
Demographics
In the first year of the study in 1972, 6039 men and 1227 women were diagnosed with LC in the four Nordic countries, with crude age-specific rates of 55.8/100,000 and 11.1/100,000. The estimated median diagnostic ages were 66 and 69 years. In 2021 case numbers for men were 7920 and for women they were 7841 with crude age-specific rates of 57.7/100,000 and 57.3/100,000; estimated median diagnostic ages were 76 and 73 years, respectively. According to US SEER data, LC median diagnostic ages were 71 and 72 years for men and women in 2016-20.
Incidence and mortality in the Nordic countries
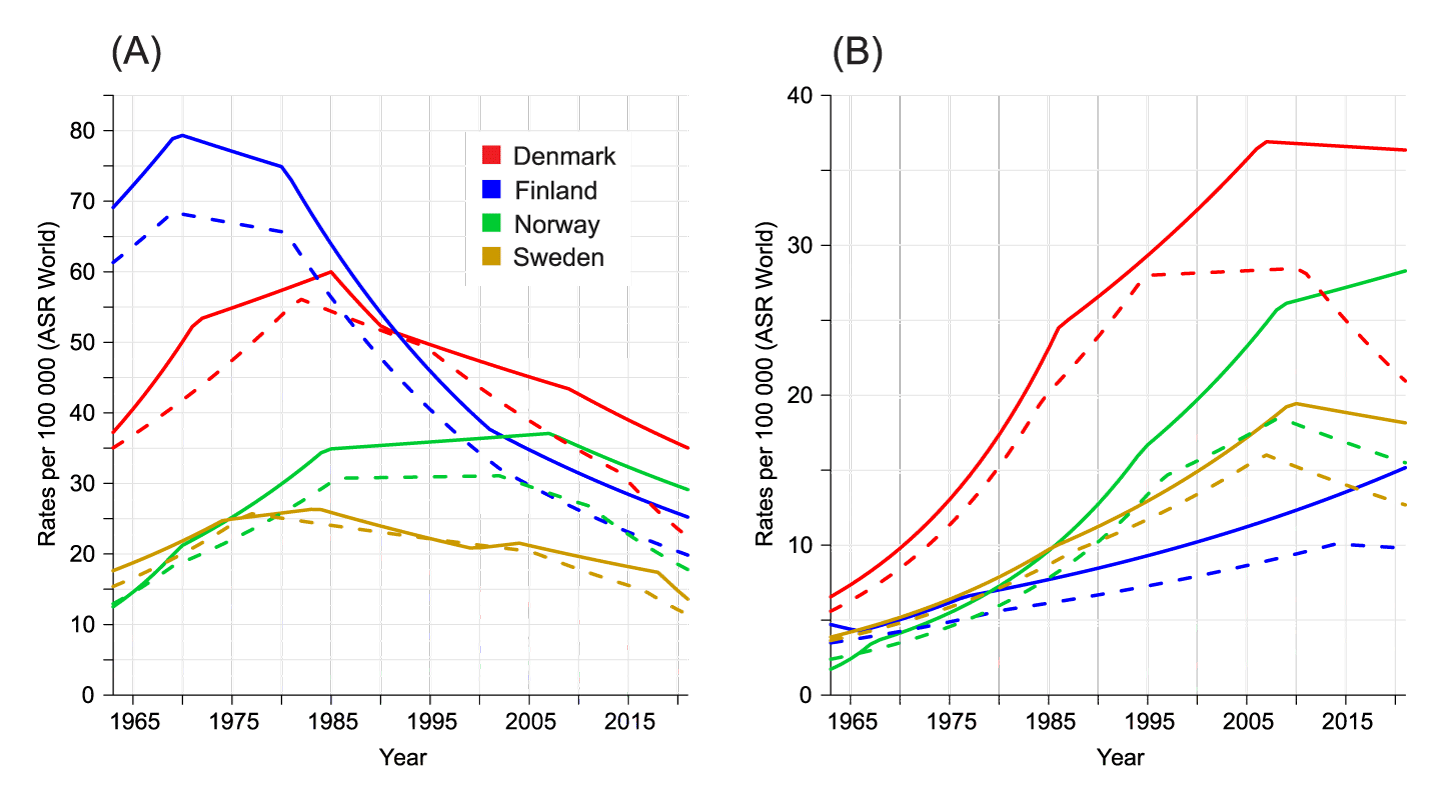
Age-standardized (world) male and female incidence and mortality trends for LC for period 1963 to 2021 are described in figure 1. Male peak incidence rates were highest for FI at 80/100,000 and lowest for SE at 25/100,000 (Figure 1A). The peak incidence was in 1970 in FI, in 1985 in DK and in 2007 in SE and NO. Mortality rates followed the incidence rates at a lower level. The female incidence rate was by far the highest for DK, reaching a peak of 37/100,000 in 2007 (Figure 1B). The NO incidence rate climbed until 2021, reaching 28/100,000. The lowest but still increasing incidence of 15/100,000 was for FI women in 2021.
Relative age-specific survival in the Nordic countries and USA
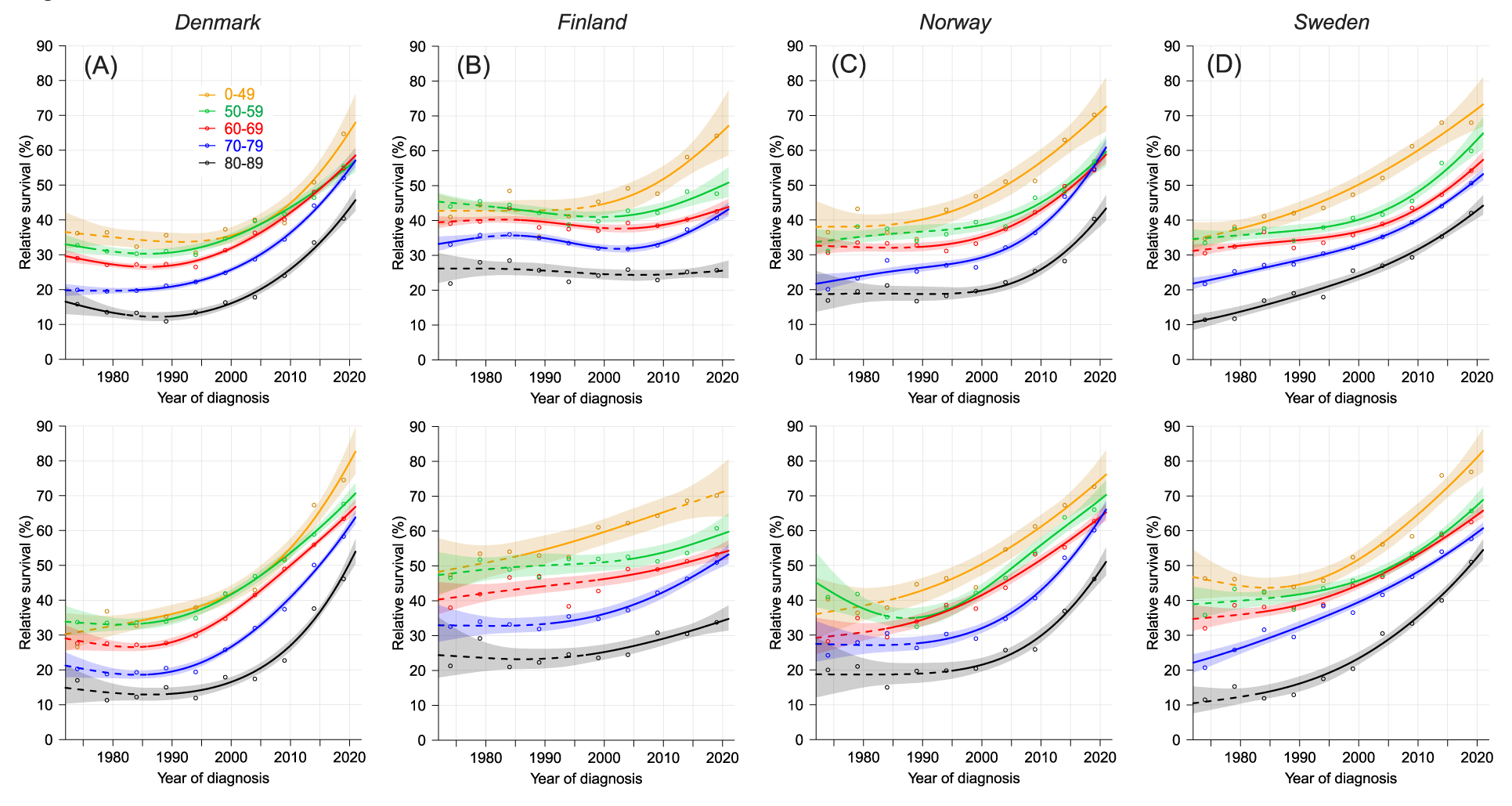
Figure 2 shows 1-year relative survival in the four countries, male data on top and female data underneath. Survival started to increase after year 2000, except for SE men some increase took place from the beginning on. Men diagnosed at age below 50 years reached a survival of 70%, some 30 % units better than for the 80-89-year-old men; survival for FI men at that age did not increase in the 50-year period. Female survival started at the same level as male survival but developed to some 10 % units higher level.
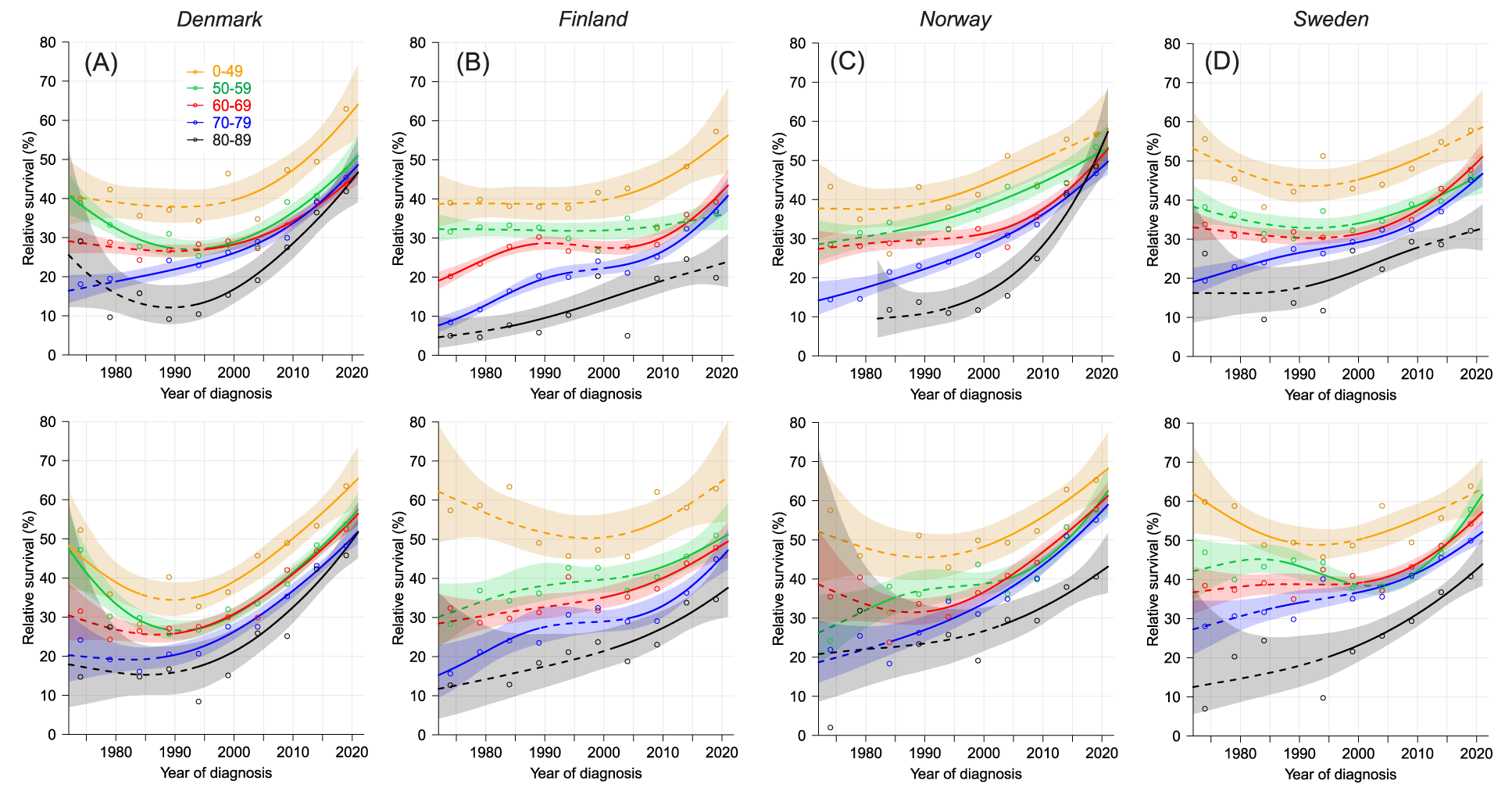
Conditional 5/1-relative survival is shown in figure 3. The shapes of the curves are like those in figure 2 but they run at some 20 % units lower level.
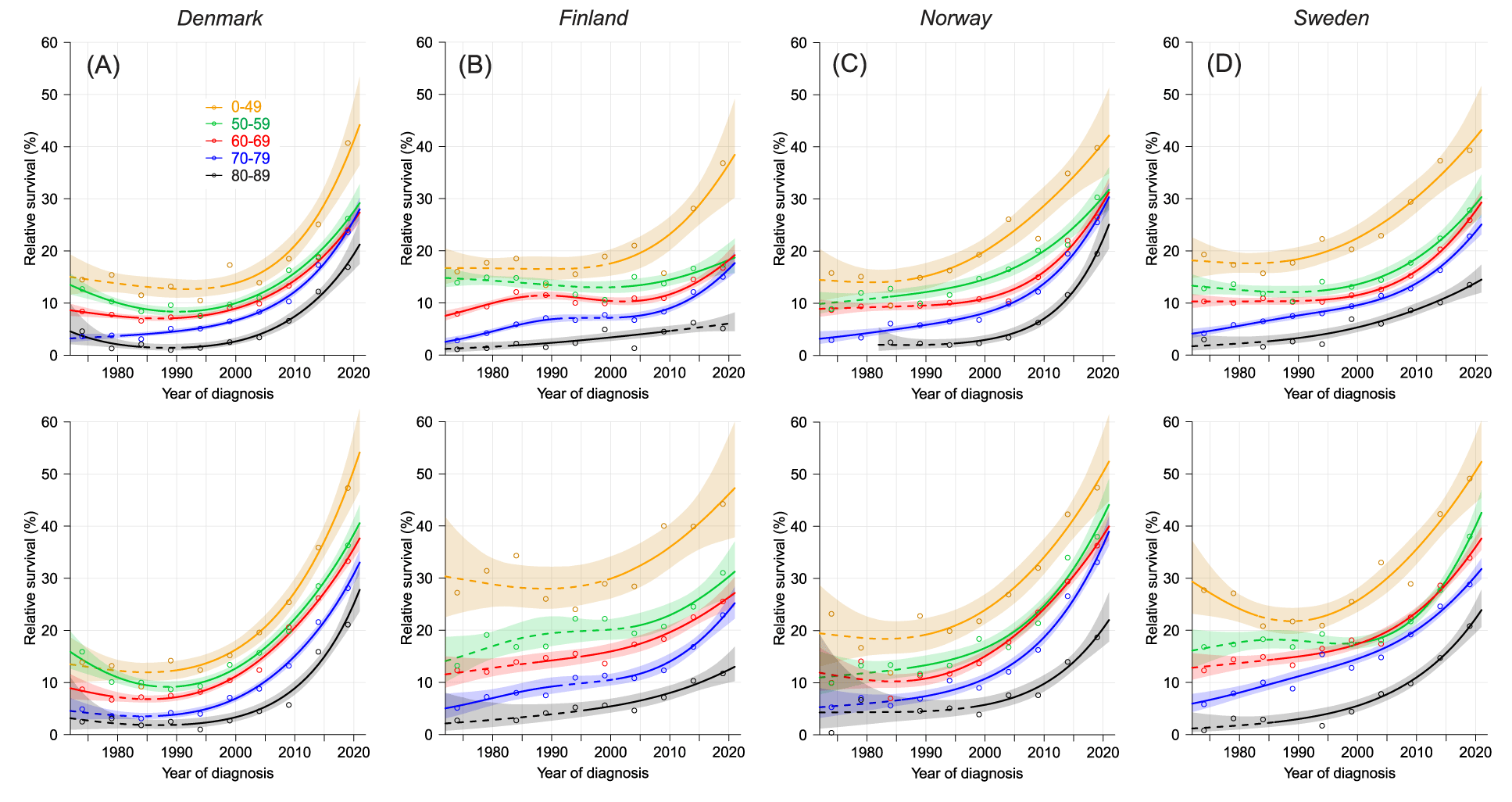
Survival for 5 years is a composite of 1- and 5/1-year survival (Figure 4). The final survival levels for the age-groups were equal for DK, NO and SE, but considering the low starting levels, the survival improvement was largest in DK, as is also confirmed by the increasingly steep curves towards year 2021. For DK, FI and SE every female age-group showed a better survival that the male age-groups. This was also true for NO, except for the oldest age-group. For men below age 50 years survival was over 40% (FI somewhat less) and for women of this age it was over 50% in countries other than FI. Survival reached over 20% for the oldest male and female patients but it was 7% for men and 12% for women in FI. DK survival curves stayed well parallel to each other but for FI and SE men the curves diverged showing increasing age gaps.
According to the US SEER database 5-year survival in 2016-20 for all LC was 22.1% for men and 30.3% for women. In the available age groups < 50, 50-64 and 65+ male survival was 33.3, 23.5 and 20.8% compared to female survival of 44.7, 33.8 and 28.1% (all age-group- and sex-differences were significant, non-overlapping CIs).
Discussion
We could document for all Nordic countries a period from 1972 to 2000 when hardly any change in relative survival in LC took place, followed by a steep positive development that was observed for all countries and most age-groups in both 1- and 5-year survival. However there were marked sex- and country-specific differences. Female survival exceeded male survival in almost all age-groups which became most prominent in the last decade and was evident already in 1-year survival which exceeded male survival by 10 % units in many age-groups. Sex differences have been reported earlier for LC without clear explanation [13,15,16]. Although NORDCAN lacks histological data, the SE national LC register for year 2008 to 2022 showed that women had a larger proportion of therapeutically amenable adenocarcinoma (the proportion increased from 50 to 60%, compared to men from 40 to 54%); therapeutically more refractory SCC decreased in the same period from 14 to 13% in women and 25 to 21% in men; small cell carcinoma declined for both sexes from 14 to 10% (20230921_nlcr_nationell_rapport2022.pdf (cancercentrum.se). The above studies also reported the low LC survival in FI, suggesting lower investment in health care as a contributing factor [13,15,16]. OECD statistics on numbers of CT scanners per 1 000 000 inhabitants in 2021/22 are telling on this point (FI 17, SE 23, NO 30, DK 44) (https://data.oecd.org/healtheqt/computed-tomography-ct-scanners.htm?appsule=4); maybe this could be a surrogate for early LC detection.
The most telling document of the development of age-group specific survival is provided by figure 4 describing 5-year relative survival. Firstly, analyzing absolute survival gaps between the youngest and the oldest patients over the 50-year period shows strong improvement and constant gaps for DK and NO men, a small widening of gaps for NO women and SE, and a larger widening for FI. Secondly, we need to observe that survival for the 80-89-year old patients was practically nil in the early period. Thus survival improvements have not equally benefitted all age-groups in the Nordic countries but, excluding FI, a quarter of the oldest patients has now reached 5-year survival.
Age-group-specific 5-year survival was better in the Nordic countries than in USA. Even looking at the exact last data points in figure 3, 5-year survival for men diagnosed before age 50 years was 40% (FI 37%) compared to US survival of 33.3%; for female the data points were at 48% (FI 45%) compared to US survival of 44.7%. Averaging the Nordic data points for 50-59 and 60-69 male survival was 27% (FI 17%) compared to US men of 50-64 years of 23.5%; Nordic female survival was 37% (FI 28%) compared to US of 33.8%. A relevant difference between the countries was in the histological distribution of LC, with US having a higher proportion of relatively fatal forms. In SE, SCC histology accounted for some 10 % units lower proportion compared to US, and even SE proportion of small cell carcinoma was a few % units lower compared to US.
The first question arising from these results is how the survival boost in year 2000 was brought about. Previous clinical and survival studies on LC from the Nordic countries describe some periodic changes in diagnostics and treatment of LC [15,16,35,36]. These and the national LC quality register for SE (2022), report that a half of patients were diagnosed with metastasis and over 70% were in age group 70+ years (20230921_nlcr_nationell_rapport2022.pdf (cancercentrum.se). Facilitated care pathways together with centralized treatment and consultation by multidisciplinary teams have been the cornerstones of the national cancer plans that were first instituted in DK in 2000, and in NO and SE shortly thereafter [37]. These three countries were part of an international study on time intervals from symptoms to treatment in LC among 10 jurisdictions [38]. The shortest times were found for DK, NO was on the second place but SE landed on the seventh place.
Other improvements since year 2000, according to the above SE report, include wide use of sensitive CT in LC diagnostics and PET to determine the extent of tumor spread. Diagnostics are increasingly based on histological (PAD) samples which can be used for additional laboratory testing, including Epidermal Growth Factor Receptor (EGFR) mutation and Anaplastic Lymphoma Kinase (ALK) rearrangement analyses; targeted therapies are available for patients testing positive. Minimal invasive surgery has become the main therapeutic modality for early-stage lung cancer, while for stage II-III patients radio- (recently stereotactic radiotherapy), chemo- and recently immunotherapy are offered [11]. For stage IV patients surgery, chemotherapy (platinum is often used), targeted therapy or immunotherapy are treatment options; immunotherapy is indicated for PDL1 positive patients or if tumor mutation burden is high; it is often combined first line with chemotherapy or chemoimmunotherapy [10,11,39,40]. Periodic changes were reported in a NO LC study as both early diagnostic stages and curative treatment have increased, the latter from 22.9% 2001 to 37.9% in 2016; these constituted 20.6% surgery and 17.3% radiotherapy, conventional and stereotactic radiotherapy in equal proportions [36].
Stage presentation of LC has been relatively constant in SE since 2002 and stage IV has remained dominant (20140210-nlcr-rapport.pdf (cancercentrum.se). Although the importance of stage for survival is well known, the modification of this by the performance status is less well known [41]. Median survival in stage IV non-small cell LC diagnosed in SE in 2016-22 was over 1.5 years for WHO 0, below 1 years for WHO 1, about 4 months for WHO 2 and less than 2 months for WHO 3 and 4 (20230921_nlcr_nationell_rapport2022.pdf. (cancercentrum.se)). Few published studies control both stage and performance status although some include comorbidities [17,18,20,41]. Time-dependent changes in LC histology have also contributed to the positive survival development, as the share of the relatively fatal small cell and SCC LC have decreased; these cancer types are more related to smoking than adenocarcinoma which could contribute to small country-specific differences in histological types [16].We reported also demographic and incidence changes in LC which have been quite large in the Nordic countries. The incidence difference (male/female) decreased from 5/1 to 5/5. The median age of onset increased by 10 years for men (66 to 76 years) and by 4 years for women (69 to 73 years). Our data do not tell what caused these changes which could be related to changes is cigarettes and general increase in longevity [42]. However, at least the major increase in age of onset for men past the female age of onset is likely to be related to the early declining in smoking frequency in men [3].
The second question is the survival of the oldest patients and the age-differences overall. Even though the fair goal of oncology is to offer equal survival chances to patients of all ages the current state is far from equal survival, and worse, the survival gaps may be increasing. A Dutch study compared 5-year survival in all cancer patients diagnosed in 1990-94 to those diagnosed in 2015-9 [19]. The survival difference between < 80 year and 80+ year old increased from 26 to 37% points. Our recent survival studies from NORDCAN showed vastly widening age gaps for multiple myeloma and acute myeloid leukemia [43,44]. In USA concerns have also been raised on how to narrow the age-related survival gap, particularly in cancers with relatively large age-related differences in survival, including those at the stomach, esophagus, liver and pancreas [14]. The present results were encouraging in showing increases in survival in all age groups, and in DK, with the largest survival increases, patients of all ages reaped the benefits. Most likely this indicates that effective treatment is offered to many patients irrespective of age as long as the performance status this allows. The largest age differences were in FI because of the poor improvement of survival among the 80-89-old. A further observation was that in 5/1-year conditional survival the age-group differences narrowed in time, however with country-specific differences. This is further evidence that effective treatment was offered also to the oldest patients to help them survive from year 1 to year 5. The limitation of NORDCAN data is the lack of clinic-pathological and treatment data, which we however complemented from other sources, yet lacking information on individual patients. Although data in the Nordic cancer registries are largely comparable, the SE cancer registry does not complement its cancer notification from the cause of death register, alike the other registries [30]. This is known to lead to a small underreporting of fatal cancers [30,45]. Nevertheless the Nordic cancer registries are the source of the oldest available national cancer data which we were able to use here.
In conclusion, the results are reassuring that survival for LC that did not improve in 30 years until year 2000, subsequently improved with increasing tempo towards 2017-21. Even more reassuring is that the increase in survival supported all age groups which suggests that even the oldest patients are offered effective treatment. As LC remains a fatal disease primary prevention is the first target. For further gains in survival in LC it would be necessary to diagnose more patients in early stages. Population screening strategies with CT are being planned but the costs may be prohibitive [11]. However there is a continued debate on this topic which is outside the scope of the present paper [46]. Predictive biomarkers would be useful as for any cancer. Improved treatment has already played an important role but the present study is not able to catch the most recent trends, because the last informative periods are the combination of the 5-year periods of 2012-16 and 2017-21.
Data Availability
A public database freely available to anyone was used at website https://nordcan.iarc.fr/en
Ethics
Anonymous data from a publically available database were used posing no ethical issues.
Author Contributions
Design: Kari Hemminki
Acquisition of data: Frantisek Zitricky, Kari Hemminki
Statistical analysis and interpretation: Kari Hemminki, Frantisek Zitricky, Asta Försti, Otto Hemminki and Akseli Hemminki
Manuscript writing: Kari Hemminki and all other authors.
Approval of the final text: All authors
Conflict of Interest
A.H. is shareholder in Circio Holdings ASA. A.H. is employee and shareholder in TILT Biotherapeutics Ltd. Other authors declared no conflict of interest.
Funding Sources
The Cooperatio Program, research area SURG and National Institute for Cancer Research – NICR (Programme EXCELES, ID Project No. LX22NPO5102), funded by the European Union - Next Generation EU and the SALVAGE project, reg.no: CZ.02.01.01/00/22_008/0004644, Jane and Aatos Erkko Foundation, Sigrid Juselius Foundation, Finnish Cancer Organizations, University of Helsinki, Helsinki University Central Hospital, Novo Nordisk Foundation, Päivikki and Sakari Sohlberg Foundation and Finnish Red Cross Blood Service.
References
- Islami F, Torre LA, Jemal A. Global trends of lung cancer mortality and smoking prevalence. Transl Lung Cancer Res. 2015 Aug;4(4):327-38. doi: 10.3978/j.issn.2218-6751.2015.08.04. PMID: 26380174; PMCID: PMC4549470.
- Thun M, Peto R, Boreham J, Lopez AD. Stages of the cigarette epidemic on entering its second century. Tob Control. 2012 Mar;21(2):96-101. doi: 10.1136/tobaccocontrol-2011-050294. PMID: 22345230.
- Lipfert FW, Wyzga RE. Longitudinal relationships between lung cancer mortality rates, smoking, and ambient air quality: a comprehensive review and analysis. Crit Rev Toxicol. 2019 Oct;49(9):790-818. doi: 10.1080/10408444.2019.1700210. Epub 2020 Jan 27. PMID: 31985340.
- Hemminki K, Försti A, Hemminki A, Ljungberg B, Hemminki O. Incidence trends in lung and bladder cancers in the Nordic Countries before and after the smoking epidemic. Eur J Cancer Prev. 2022 May 1;31(3):228-234. doi: 10.1097/CEJ.0000000000000694. PMID: 34074862.
- Turner MC, Andersen ZJ, Baccarelli A, Diver WR, Gapstur SM, Pope CA 3rd, Prada D, Samet J, Thurston G, Cohen A. Outdoor air pollution and cancer: An overview of the current evidence and public health recommendations. CA Cancer J Clin. 2020 Aug 25:10.3322/caac.21632. doi: 10.3322/caac.21632. Epub ahead of print. PMID: 32964460; PMCID: PMC7904962.
- Frank C, Fallah M, Ji J, Sundquist J, Hemminki K. The population impact of familial cancer, a major cause of cancer. Int J Cancer. 2014 Apr 15;134(8):1899-906. doi: 10.1002/ijc.28510. Epub 2013 Oct 31. PMID: 24590453.
- Liu X, Hemminki K, Försti A, Sundquist K, Sundquist J, Ji J. Cancer risk in patients with type 2 diabetes mellitus and their relatives. Int J Cancer. 2015 Aug 15;137(4):903-10. doi: 10.1002/ijc.29440. Epub 2015 Feb 5. PMID: 25604005.
- Cumberbatch MGK, Jubber I, Black PC, Esperto F, Figueroa JD, Kamat AM, Kiemeney L, Lotan Y, Pang K, Silverman DT, Znaor A, Catto JWF. Epidemiology of Bladder Cancer: A Systematic Review and Contemporary Update of Risk Factors in 2018. Eur Urol. 2018 Dec;74(6):784-795. doi: 10.1016/j.eururo.2018.09.001. Epub 2018 Sep 26. PMID: 30268659.
- Brown KF, Rumgay H, Dunlop C, Ryan M, Quartly F, Cox A, Deas A, Elliss-Brookes L, Gavin A, Hounsome L, Huws D, Ormiston-Smith N, Shelton J, White C, Parkin DM. The fraction of cancer attributable to modifiable risk factors in England, Wales, Scotland, Northern Ireland, and the United Kingdom in 2015. Br J Cancer. 2018 Apr;118(8):1130-1141. doi: 10.1038/s41416-018-0029-6. Epub 2018 Mar 23. PMID: 29567982; PMCID: PMC5931106.
- Löfling L, Bahmanyar S, Kieler H, Lambe M, Wagenius G. Temporal trends in lung cancer survival: a population-based study. Acta Oncol. 2022 May;61(5):625-631. doi: 10.1080/0284186X.2021.2013529. Epub 2021 Dec 10. PMID: 34889167.
- Maconachie R, Mercer T, Navani N, McVeigh G; Guideline Committee. Lung cancer: diagnosis and management: summary of updated NICE guidance. BMJ. 2019 Mar 28;364:l1049. doi: 10.1136/bmj.l1049. Erratum in: BMJ. 2019 Apr 1;365:l1514. doi: 10.1136/bmj.l1514. PMID: 30923038.
- Allemani C, Matsuda T, Di Carlo V, Harewood R, Matz M, Nikšić M, Bonaventure A, Valkov M, Johnson CJ, Estève J, Ogunbiyi OJ, Azevedo E Silva G, Chen WQ, Eser S, Engholm G, Stiller CA, Monnereau A, Woods RR, Visser O, Lim GH, Aitken J, Weir HK, Coleman MP; CONCORD Working Group. Global surveillance of trends in cancer survival 2000-14 (CONCORD-3): analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet. 2018 Mar 17;391(10125):1023-1075. doi: 10.1016/S0140-6736(17)33326-3. Epub 2018 Jan 31. PMID: 29395269; PMCID: PMC5879496.
- Hemminki J, Försti A, Hemminki A, Hemminki K. Survival trends in solid cancers in the Nordic countries through 50 years. Eur J Cancer. 2022 Nov;175:77-85. doi: 10.1016/j.ejca.2022.08.015. Epub 2022 Sep 9. PMID: 36096040.
- Withrow DR, Nicholson BD, Morris EJA, Wong ML, Pilleron S. Age-related differences in cancer relative survival in the United States: A SEER-18 analysis. Int J Cancer. 2023 Jun 1;152(11):2283-2291. doi: 10.1002/ijc.34463. Epub 2023 Feb 23. PMID: 36752633.
- Tichanek F, Försti A, Hemminki O, Hemminki A, Hemminki K. Survival in Lung Cancer in the Nordic Countries Through A Half Century. Clin Epidemiol. 2023 May 1;15:503-510. doi: 10.2147/CLEP.S406606. PMID: 37153073; PMCID: PMC10162394.
- Lundberg FE, Ekman S, Johansson ALV, Engholm G, Birgisson H, Ólafsdóttir EJ, Mørch LS, Johannesen TB, Andersson TM, Pettersson D, Seppä K, Virtanen A, Lambe M, Lambert PC. Trends in lung cancer survival in the Nordic countries 1990-2016: The NORDCAN survival studies. Lung Cancer. 2024 Jun;192:107826. doi: 10.1016/j.lungcan.2024.107826. Epub 2024 May 22. PMID: 38795460.
- Francisci S, Minicozzi P, Pierannunzio D, Ardanaz E, Eberle A, Grimsrud TK, Knijn A, Pastorino U, Salmerón D, Trama A, Sant M; EUROCARE-5 Working Group:. Survival patterns in lung and pleural cancer in Europe 1999-2007: Results from the EUROCARE-5 study. Eur J Cancer. 2015 Oct;51(15):2242-2253. doi: 10.1016/j.ejca.2015.07.033. Epub 2015 Sep 26. PMID: 26421826.
- Bi JH, Tuo JY, Xiao YX, Tang DD, Zhou XH, Jiang YF, Ji XW, Tan YT, Yuan HY, Xiang YB. Observed and relative survival trends of lung cancer: A systematic review of population-based cancer registration data. Thorac Cancer. 2024 Jan;15(2):142-151. doi: 10.1111/1759-7714.15170. Epub 2023 Nov 20. PMID: 37986711; PMCID: PMC10788469.
- Schuurman MS, Lemmens VEPP, Portielje JEA, van der Aa MA, Visser O, Dinmohamed AG. The cancer burden in the oldest-old: Increasing numbers and disparities-A nationwide study in the Netherlands, 1990 to 2019. Int J Cancer. 2024 Jan 15;154(2):261-272. doi: 10.1002/ijc.34705. Epub 2023 Sep 4. PMID: 37664984.
- Schuurman MS, Lemmens VEPP, Portielje JEA, van der Aa MA, Visser O, Dinmohamed AG. The cancer burden in the oldest-old: Increasing numbers and disparities-A nationwide study in the Netherlands, 1990 to 2019. Int J Cancer. 2024 Jan 15;154(2):261-272. doi: 10.1002/ijc.34705. Epub 2023 Sep 4. PMID: 37664984.
- Park SY, Lee JG, Kim J, Bae MK, Lee CY, Kim DJ, Chung KY. The influence of smoking intensity on the clinicopathologic features and survival of patients with surgically treated non-small cell lung cancer. Lung Cancer. 2013 Sep;81(3):480-486. doi: 10.1016/j.lungcan.2013.07.002. Epub 2013 Jul 26. PMID: 23896023.
- Okamoto T, Suzuki Y, Fujishita T, Kitahara H, Shimamatsu S, Kohno M, Morodomi Y, Kawano D, Maehara Y. The prognostic impact of the amount of tobacco smoking in non-small cell lung cancer--differences between adenocarcinoma and squamous cell carcinoma. Lung Cancer. 2014 Aug;85(2):125-30. doi: 10.1016/j.lungcan.2014.06.006. Epub 2014 Jun 16. PMID: 24976334.
- Bergman M, Fountoukidis G, Smith D, Ahlgren J, Lambe M, Valachis A. Effect of Smoking on Treatment Efficacy and Toxicity in Patients with Cancer: A Systematic Review and Meta-Analysis. Cancers (Basel). 2022 Aug 25;14(17):4117. doi: 10.3390/cancers14174117. PMID: 36077654; PMCID: PMC9454993.
- IARC. Cancer incidence in five continents. Editor. Lyon: IARC; 2002.
- Hemminki K, Försti A, Hemminki A, Ljungberg B, Hemminki O. Survival in bladder and upper urinary tract cancers in Finland and Sweden through 50 years. PLoS One. 2022 Jan 4;17(1):e0261124. doi: 10.1371/journal.pone.0261124. PMID: 34982793; PMCID: PMC8726478.
- Patja K, Hakala SM, Boström G, Nordgren P, Haglund M. Trends of tobacco use in Sweden and Finland: do differences in tobacco policy relate to tobacco use? Scand J Public Health. 2009 Mar;37(2):153-60. doi: 10.1177/1403494808100277. Epub 2009 Jan 22. PMID: 19164430.
- GBD 2015 Tobacco Collaborators. Smoking prevalence and attributable disease burden in 195 countries and territories, 1990-2015: a systematic analysis from the Global Burden of Disease Study 2015. Lancet. 2017 May 13;389(10082):1885-1906. doi: 10.1016/S0140-6736(17)30819-X. Epub 2017 Apr 5. Erratum in: Lancet. 2017 Oct 7;390(10103):1644. doi: 10.1016/S0140-6736(17)32559-X. PMID: 28390697; PMCID: PMC5439023.
- Koskinen A, Hemminki O, Försti A, Hemminki K. Incidence and survival in laryngeal and lung cancers in Finland and Sweden through a half century. PLoS One. 2022 May 27;17(5):e0268922. doi: 10.1371/journal.pone.0268922. PMID: 35622857; PMCID: PMC9140270.
- Engholm G, Ferlay J, Christensen N, Bray F, Gjerstorff ML, Klint A, Køtlum JE, Olafsdóttir E, Pukkala E, Storm HH. NORDCAN--a Nordic tool for cancer information, planning, quality control and research. Acta Oncol. 2010 Jun;49(5):725-36. doi: 10.3109/02841861003782017. PMID: 20491528.
- Pukkala E, Engholm G, Højsgaard Schmidt LK, Storm H, Khan S, Lambe M, Pettersson D, Ólafsdóttir E, Tryggvadóttir L, Hakanen T, Malila N, Virtanen A, Johannesen TB, Larønningen S, Ursin G. Nordic Cancer Registries - an overview of their procedures and data comparability. Acta Oncol. 2018 Apr;57(4):440-455. doi: 10.1080/0284186X.2017.1407039. Epub 2017 Dec 11. Erratum in: Acta Oncol. 2018 Apr;57(4):565. doi: 10.1080/0284186X.2018.1425193. PMID: 29226751.
- Larønningen S, Ferlay J, Beydogan H, Bray F, Engholm G, Ervik M. NORDCAN: Cancer incidence, mortality, prevalence and survival in the nordic countries. Association of the nordic cancer registries. Cancer Registry of Norway. 2022.
- Muggeo VM. Estimating regression models with unknown break-points. Stat Med. 2003 Oct 15;22(19):3055-71. doi: 10.1002/sim.1545. PMID: 12973787.
- Muggeo VMR. Segmented: An r package to fit regression models with broken-line relationships. R News. 2008;8:20-25.
- Zitricky F, Koskinen AI, Hemminki O, Försti A, Hemminki A, Hemminki K. Survival in oral and pharyngeal cancers is catching up with laryngeal cancer in the NORDIC countries through a half century. Cancer Med. 2024 Jan;13(1):e6867. doi: 10.1002/cam4.6867. Epub 2024 Jan 2. PMID: 38164108; PMCID: PMC10807619.
- Sachs E, Sartipy U, Jackson V. Sex and Survival After Surgery for Lung Cancer: A Swedish Nationwide Cohort. Chest. 2021 May;159(5):2029-2039. doi: 10.1016/j.chest.2020.11.010. Epub 2020 Nov 17. PMID: 33217414; PMCID: PMC8129733.
- Solberg S, Nilssen Y, Brustugun OT, Grimsrud TK, Haram PM, Helbekkmo N, Helland Å, Hjelde HH, Jakobsen B, Møller B, Petersen M, Strand TE, Wahl SGF, Aanerud M, Fjellbirkeland L. Increase in curative treatment and survival of lung cancer in Norway 2001-2016. Eur J Epidemiol. 2019 Oct;34(10):951-955. doi: 10.1007/s10654-019-00536-z. Epub 2019 Jul 16. PMID: 31313073.
- Probst HB, Hussain ZB, Andersen O. Cancer patient pathways in Denmark as a joint effort between bureaucrats, health professionals and politicians--a national Danish project. Health Policy. 2012 Apr;105(1):65-70. doi: 10.1016/j.healthpol.2011.11.001. Epub 2011 Dec 1. PMID: 22136810.
- Menon U, Vedsted P, Zalounina Falborg A, Jensen H, Harrison S, Reguilon I, Barisic A, Bergin RJ, Brewster DH, Butler J, Brustugun OT, Bucher O, Cairnduff V, Gavin A, Grunfeld E, Harland E, Kalsi J, Knudsen AK, Lambe M, Law RJ, Lin Y, Malmberg M, Turner D, Neal RD, White V, Weller D. Time intervals and routes to diagnosis for lung cancer in 10 jurisdictions: cross-sectional study findings from the International Cancer Benchmarking Partnership (ICBP). BMJ Open. 2019 Nov 27;9(11):e025895. doi: 10.1136/bmjopen-2018-025895. PMID: 31776134; PMCID: PMC6886977.
- Hendriks LE, Kerr KM, Menis J, Mok TS, Nestle U, Passaro A, Peters S, Planchard D, Smit EF, Solomon BJ, Veronesi G, Reck M; ESMO Guidelines Committee. Electronic address: [email protected]. Non-oncogene-addicted metastatic non-small-cell lung cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2023 Apr;34(4):358-376. doi: 10.1016/j.annonc.2022.12.013. Epub 2023 Jan 17. PMID: 36669645.
- Hendriks LE, Kerr KM, Menis J, Mok TS, Nestle U, Passaro A, Peters S, Planchard D, Smit EF, Solomon BJ, Veronesi G, Reck M; ESMO Guidelines Committee. Electronic address: [email protected]. Oncogene-addicted metastatic non-small-cell lung cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2023 Apr;34(4):339-357. doi: 10.1016/j.annonc.2022.12.009. Epub 2023 Jan 23. PMID: 36872130.
- Jakobsen E, Rasmussen TR, Green A. Mortality and survival of lung cancer in Denmark: Results from the Danish Lung Cancer Group 2000-2012. Acta Oncol. 2016 Jun;55 Suppl 2:2-9. doi: 10.3109/0284186X.2016.1150608. Epub 2016 Apr 8. PMID: 27056247.
- Song MA, Benowitz NL, Berman M, Brasky TM, Cummings KM, Hatsukami DK, Marian C, O'Connor R, Rees VW, Woroszylo C, Shields PG. Cigarette Filter Ventilation and its Relationship to Increasing Rates of Lung Adenocarcinoma. J Natl Cancer Inst. 2017 Dec 1;109(12):djx075. doi: 10.1093/jnci/djx075. PMID: 28525914; PMCID: PMC6059254.
- Hemminki K, Zitricky F, Försti A, Kontro M, Gjertsen BT, Severinsen MT, Juliusson G. Age-specific survival in acute myeloid leukemia in the Nordic countries through a half century. Blood Cancer J. 2024 Mar 14;14(1):44. doi: 10.1038/s41408-024-01033-7. PMID: 38480693; PMCID: PMC10937905.
- Hemminki K, Zitricky F, Försti A, Silvennoinen R, Vangsted A, Hansson M. Large differencies in age-specific survival in multiple myeloma in the nordic countries. Blood Cancer J. 2024 Mar 11;14(1):43. doi: 10.1038/s41408-024-01026-6. PMID: 38467614; PMCID: PMC10928156.
- Ji J, Sundquist K, Sundquist J, Hemminki K. Comparability of cancer identification among Death Registry, Cancer Registry and Hospital Discharge Registry. Int J Cancer. 2012 Nov 1;131(9):2085-93. doi: 10.1002/ijc.27462. Epub 2012 Mar 15. PMID: 22307919.
- O'Dowd EL, Lee RW, Akram AR, Bartlett EC, Bradley SH, Brain K, Callister MEJ, Chen Y, Devaraj A, Eccles SR, Field JK, Fox J, Grundy S, Janes SM, Ledson M, MacKean M, Mackie A, McManus KG, Murray RL, Nair A, Quaife SL, Rintoul R, Stevenson A, Summers Y, Wilkinson LS, Booton R, Baldwin DR, Crosbie P. Defining the road map to a UK national lung cancer screening programme. Lancet Oncol. 2023 May;24(5):e207-e218. doi: 10.1016/S1470-2045(23)00104-3. PMID: 37142382.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.