2025 October 18;6(10):1444-1456. doi: 10.37871/jbres2201.
HIF-1α Promotes Osteogenic-Angiogenic Coupling Response of BMSCs Cell Sheets
Dan Zhang*, Yonghui Teng, Wei Liu, Chang Han and Hanwen Zhang
- Hypoxia-inducible factor-1α
- Bone marrow mesenchymal stem cells
- Cell sheets
- Bone engineering
- Osteogenic-angiogenic coupling response
Abstract
Background: There is a critical need for management of vascularization in bone tissue engineering. The purpose of this study was to use Hypoxia-Inducible Factor-1α (HIF-1α) transduced Bone Marrow Mesenchymal Stem Cells(BMSCs) to fabricate prevascularized osteogenic cell sheets In vitro and explore HIF-1α promoted osteogenic-angiogenic coupling response of BMSCs cell sheets.
Methods: HIF-1α was over-expressed by using a lentiviral vector which encoded Green Fluorescent Protein(GFP) and transduced steadily in Wistar rats BMSCs to form HIF-1α/BMSCs. While empty lentiviral transduced BMSCs(Empty/BMSCs) were as a negative control group and BMSCs without transduced were as a blank control group. Fluorescence microscope was used to detect GFP expressions, meanwhile Real-time quantitative and western blot were performed to assess the expression level of HIF-1α. Next, HIF-1α/BMSCs were cultured to form Osteogenic Cell Sheets (OCTs) with a density of 1 × 105/cm2 under osteogenesis medium. Then, Alkaline Phosphatase staining (ALP) at day 14 and Alizarin-red staining at day 21 were performed to check the characteristic of osteogenesis. Empty/BMSCs formed OCTs served as control. Simultaneously, HIF-1α/BMSCs were induced to differentiate into endothelial-like cells(i-ECs) for 14 days, and the conversion rates were performed by Flow Cytometry assay. At the same time, we used Transwell assay to detect whether HIF-1α could migrate i-ECs In vitro. Finally, i-ECs were seeded onto OCTs with a density of 5 × 104/cm2 to fabricate Prevascularized- Osteogenic Cell Sheets (P-OCTs). In order to detect the role of HIF-1α involved in osteogenic-angiogenic coupling response of P-OCTs, Immunofluorescent staining for CD31 was performed at 1,3,7,14 days to check i-ECs migration and networks formation, and western blot of Osteopontin (OPN) and Ssteocalcin (OCN) at 1,7,14 days to check bone formation. Meanwhile, Empty/BMSCs formed P-OCTs were as control.
Conclusion: BMSCs were transduced by Lenti-HIF-1α with optimal multiplicity of infection was 30(MOI = 30) and the GFP expression over 90%. At the same time, qPCR and western blot confirmed a high HIF-1α expression in HIF-1α/BMSCs group, which had a statistic significance among BMSCs group and Empty/BMSCs group (p < 0.05). Next, Flow Cytometric analysis results showed the conversion rate of HIF-1α/BMSCs to i-ECs was 92.43% in experimental group (p < 0.05), which indicated BMSCs transduced by HIF-1α had a great superiority to differentiate into endothelial cells under experimental conditions. Transwell assay showed that HIF-1α could recruit i-ECs in vitro, with an average of over 400 i-ECs migrating per field of view in HIF-1α/BMSCs group. Meanwhile no migrating i-ECs in Empty/BMSCs group (p < 0.05). On the other hand, ALP at day 14 and Alizarin-red staining at day 21 of OCTs in HIF-1α/BMSCs group showed an obviously osteogenic differentiation characteristic with more deep stained calcium nodules deposits than Empty/BMSCs group (p < 0.05). Finally, we fabricated P-OCTs and detected angiogenesis by Immunofluorescent staining for CD31 at day 1,3,7,14, which showed i-ECs migrated reticulated fast and formed a large number of lumens and networks in HIF-1α/BMSCs group. While i-ECs migrated slowly and the lumens and networks formation was limited in Empty/BMSCs group (p < 0.05). At the same time, the expressions of OCN, OPN at day 1,7,14 showed that HIF-1α could promote osteogenic response in P-OCTs significantly in vitro (p < 0.05). All in all, the over-expressed HIF-1α of BMSCs cell sheets strategy can provide a new promising method for bone engineering, which could promote osteogenic-angiogenic coupling response In vitro.
Background
Large bone defects mainly caused by trauma, tumor, inflammation are still a significant challenge in regeneration medicine due to the insufficient vascularization in engineered bone grafts [1,2]. Angiogenesis and osteogenesis are tightly coupled during bone development and regeneration. Insufficient vascularization induces lack of nutrients and oxygen which supplies a hypoxic-ischemic metabolic microenvironment for bone grafts causes low cell survival rate, slow growth of new bone, and bone grafts failure even more [3-5]. Recently, prevscularization cell sheet technology has been provided a promising strategy for bone engineering. Endothelial Cells (ECs) were seeded on the 3D cell sheets with a certain density, then they migrated into cell sheets and formed blood vessels gradually. Many studies [6-8] have demonstrated that cell sheets-based prevascularizing system was beneficial for vascular network formation and enhanced bone regeneration in vitro. However, the enhancement In vivo was still limited and sluggish resulting in precarious and insufficient prevascularized networks and poor connection with the host in hypoxic-ischemic environment [9-11]. Therefore, it is still a critical need for the management of vascularization in bone engineering under a hypoxic-ischemic metabolic microenvironment.
Hypoxia-Inducible factor-1a(HIF-1a) is an active transcription factor that upregulates numerous genes at low oxygen conditions which involve in cell proliferation and differentiation, angiogenesis, glycolysis, and wound healing [12]. Angiogenic and osteogenic coupling is a complex and precise regulation process in bone healing, and a large number of studies show that HIF-1a plays an important role in this process. However, the expression of HIF-1a is restricted by the host while is overexpressed in cancers. With the development of biomedicine engineering, gene therapy has been investigated as a targeted therapy for bone healing. Sun J, et al. [13] demonstrated HIF-1a-overexpressed exosomes could rescue the impaired angiogenic ability, migration, and proliferation by hypoxia-pretreated HUVECs In vitro and mediate cardioprotection by upregulating proangiogenic factors and enchancing neovessel formation. Rachel F, et al. [14] found that gene therapy for peripheral arterial disease by increasing revascularisation was safe and efficacy. Among genome engineering tools, Clustered Regularly Interspaced Short Palindromic Repeats(CRISPR) technology for ocular angiogenesis showed a huge potential by up-regulating the expression of HIF-1a [15]. So, the usage of HIF- 1a in biomedcine engineering is safe and efficient, which gives us some inspirations in bone engineering.
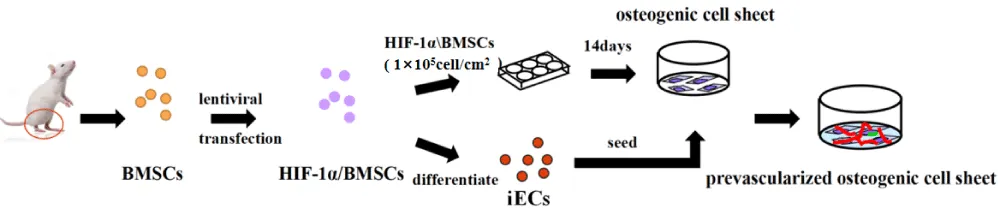
Therefore, we assumed that HIF-1a-overexpressed in BMSCs and used HIF-1a/BMSCs to construct Osteogenic Cell Sheets (OCTs). Then, we induced HIF-1a/BMSCs differentiate into Endothelial Cells(i-ECs) and seeded them onto OCTs to fabricate Prevascularized Osteogenesis Cell Sheets(P-OCTs). Finally, we detected vascular networks formation and bone regeneration in vitro, in order to provide evidence for participation of HIF-1a in angiogenic and osteogenic coupling effects.
Materials and Methods
rBMSCs harvest and culture
Three or four-week-old Wistar rats of 60-100g in weight were purchased from Jiangxi Zhonghongboyuan Biotechnology Company (Jiangxi province, China). The Wistar rats were anesthetized with diethyl ether and then killed by cervical dislocation. Rat bone marrow was aspirated from femoral medullary canal and supercentrifuged at 1500 r/min, then cultured in low-glucose Dulbecco’s modified Eagle’s medium (L-DMEM, Hyclone, USA) supplement with 10% fetal bovine serum (FBS, Hyclone, USA) and 2% antibiotics (100U/ml penicillin and streptomycin, Hyclone, USA), wihch was called L-DMED complete medium. BMSCs were cultured in a humidified incubator (Heraeus, Germany) of 5% CO2 at 37°C and the culture medium was replaced every 2 or 3 days. BMSCs were passaged with 0.25% trypsin/EDTA when adherent and confluent completely.
HIF-1α transduction into BMSCs
In order to construct a stable transfection BMSCs lines, the backbone vector pHBLV-CMV-MCS-EF1-ZsGreen-T2A-puro (Hanbio Biotechnology, Shanghai, China) was used for the reconstruction of a lentiviral vector containing HIF-1α. The Lenti-HIF-1α was made to overexpress using replication defective lentivirus that encoded Green Fluorescent Protein (GFP), and BMSCs were transduced according to the product instruction. Then, a specific form of HIF-1α RNA was extracted according to the protocol provided in the HiScript II Q RT SuperMix for qPCR (+gDNA wiper) (R223-01, Vazyme, China) instructions. Real-time quantitative PCR was performed by using the SuperStar Universal SYBR Master Mix (CW3360M, CWBIO, China) in the CFX Connect™ real-time system (Bio-rad, USA). The relative quantity of mRNA was calculated using the 2-△△CT method. The sequences of primers designed by Generalbiol CO. Ltd. (Anhui, China) were listed in table 1. The BMSCs group was as a blank control group, an empty lentivirus transduced BMSCs(Empty/BMSCs) was employed as a negative control group.
| Table 1: Primers for RT-PCR analysis of HIF-1α. | |||
| Gene | Primer sequence (5'-3') | Product length(bp) | Annealing temperature(℃) |
| β-actin F | GCCATGTACGTAGCCATCCA | 375 | 58 |
| β-actin R | GAACCGCTCATTGCCGATAG | ||
| HIF-1α F | CTAAAGGACAAGTCACCACAGG | 314 | 58.7 |
| HIF-1α R | TAGCAGAGTCAGGGCATCG | ||
Western blot analysis
Cells were washed twice with cold PBS and lysed in 200μL RIPA cell lysis buffer (C1053, Applygen, China) for 10-20 minutes, and then scraped into microfuge tubes and centrifuged for 10 minutes at 12000 r/min. Collected the supernatant and the protein concentration was measured by BCA Protein Assay Kit (E-BC-K318-M, Elabscience, China). Subsequently, equal amounts of cell lysates were separated by SDS-PAGE gel (A1010, Solarbio, China) and transferred to polyvinylidene fluoride membranes (PVDF, IPVH00010, Millipore). The membranes were blocked with 3% skim milk (P1622, Applygen, China) in Tween Tris-buffered saline(TTBS) (0.5%, Tween-20) for 1 hour. The blocked membranes were incubated with primary antibodies (Rabbit Anti Rat HIF-1α, Bioss, China) overnight. After washing in TTBS, membranes were then incubated with secondary antibodies (HRP conjugated Goat Anti-Rabbit IgG (H+L), GB23303, Servicebio, China) for 2 hours at room temperature. Images for western blot analysis were visualizes by automatic chemiluminescence image analysis system (Tanon-5200, Tanon Life Science, China). Relative protein levels were calculated as ratio level of protein of interest to α-actin in each sample.
Differentiation of HIF-1α/BMSCs into endothelial-like cells
The second generations of HIF-1α/BMSCs were seeded into 6-well plates at a density of 3 × 104 cell/cm2 and cultured with L-DMEM complete medium in a humidified atmosphere of 5% CO2 at 37°C. When HIF-1α/BMSCs reached confluence, the culture medium was replaced by M199 medium (Hyclone, USA) plus 10% FBS, vascular endothelial growth factor (VEGF, 10μg/L, Peproech, USA) and basic fibroblast growth factor (bFGF, 2μg/L, Peproech, USA), which referred as M199 complete medium. HIF-1α/BMSCs were differentiated for 14 days, while HIF-1α/BMSCs cultured without VEGF and bFGF were as a control.
Flow cytometric analysis of endothelial-like cells
Flow cytometric analysis was used to detect whether the i-ECs had the endothelia specific phenotype PECAM-1(CD31). After differentiated culture for 14 days, HIF-1α/BMSCs were trypsinized (0.25% trypsin/EDTA), and added into an Eppendorf tube with a density of 1 × 105/mL, then incubated with the monoclonal antibody CD31(25-0310-82, Thermo Scientific, China) for 30 minutes at 4°C While non-differentiated HIF-1α/ BMSCs group served as a control. Afterwards, the samples were performed on the FACSVerse flow cytometer (BD, USA) and analyzed the CD31-positive expressions.
Transwell assay of endothelial-like cells
HIF-1α/BMSCs were seeded into 24-well plate with a density of 1 × 105/cm2 for continuous culture to form cell sheet. i-ECs were seeded in the upper chamber with a density of 1 × 105/mL for 100 μL, and 600 μL of H-DMEM complete medium (containing 10% FBS) was added to the lower chamber. Next, the plate was incubated at 37°C in a humidified air and 5% CO2 environment for 24 hours. Then, the unmigrated i-ECs in the upper chamber were removed, and the chamber was fixed with 4% polyformaldehyde for 30 minutes. The migrated cells in the lower chamber were stained with 1% methylene blue solution diluted in PBS (1:40) for 15 minutes. The cells were rinsed three times with PBS, and the number of migrated cells was counted under microscope. Empty/BMSCs were seeded at the same density in a 24-well plate as a control group.
Fabrication of osteogenesis cell sheets
HIF-1α/BMSCs were seeded into the 6-well plate with a density of 1 × 105/cm2 and cultured in osteogenesis medium which contained 12% FBS, 50 mg/L ascorbic acid, 10nmol/L dexamethasone, and 10 mmol/L α-glycerophosphate for 21days to fabricate Osteogenesis Cell Sheets (OCTs). Alkaline Phosphatase (ALP) at day 14 and Alizarin Red-S staining at day 21 were performed to characterize the osteogenic properties of OCTs. At the same time, Empty/BMSCs formed OCTs served as a control.
Fabrication of prevascularized osteogenesis cell sheet
To fabricate Prevascularized Osteogenesis Cell Sheets (P-OCTs), i-ECs were seeded onto the osteogenesis cell sheets surface with a density of 5 × 104/cm2 and cultured with a mixed medium which concluded M199 complete medium and osteogenesis medium (1:1, v/v). The culture medium was replaced every 2-3 days. At the same time, i-ECs were seeded onto Empty/BMSCs formed OCTs served as a control.
Immunofluorescent staining of prevascularized osteogenesis cell sheet
The fluorescent microscope was used to detect the i-ECs migration in the OCTs. After cultured for 1,3,7,14 days, the P-OCTs was washed with Phosphate-Buffered Saline (PBS), fixed in 4% paraformaldehyde for 15 minutes, and then blocked in 5% goat serum-PBS buffer solution for 1 hour at room temperature. A primary antibody Rabbit Anti Rat CD31 (GB11053-2, Servicebio, China, dilution 1/200) in 1% Bovine Serum Albumin (BSA)-PBS was added to the samples and incubated overnight at 4%. After washing with PBS, a secondary antibody Cy3 Goat Anti-Rabbit IgG(H + L) (AS007αABclonal, China, dilution 1/200) in 1% BSA-PBS buffer was added and incubated in the dark for 1 hour at room temperature. Finally, the cell nuclei were counterstained with DAPI (KGA215-50, keygentec. Jiangsu, China) for 5 minutes and rinsed three times with PBS in the dark. The fluorescent staining images were captured by confocal microscopy (Research invested system microscope, IX71, Olympus).
Osteogenesis proteins expression of prevascularized osteogenesis cell sheet
Osteogenesis proteins were extracted and electroblotted as above-mentioned after P-OCT cultured for 1,7,14 days. The same procedure was followed on reaction products of the following antibodies, including to Osteocalcin (OCN) (Rabbit Anti Rat OCN, A20800, Abclonal, China), Osteopontin (OPN)(Rabbit Anti Rat OPN, YT3467, Immunoway, China).
Statistical Analysis
All data were express as mean ± standard deviation. The variance(ANOVA) and Tukey post hoc tests were performed to analyze the results by using SPSS20.0 software, where a significant difference was considered if the p value was less than 0.05 (Figure 1).
Results
HIF-1α transduction
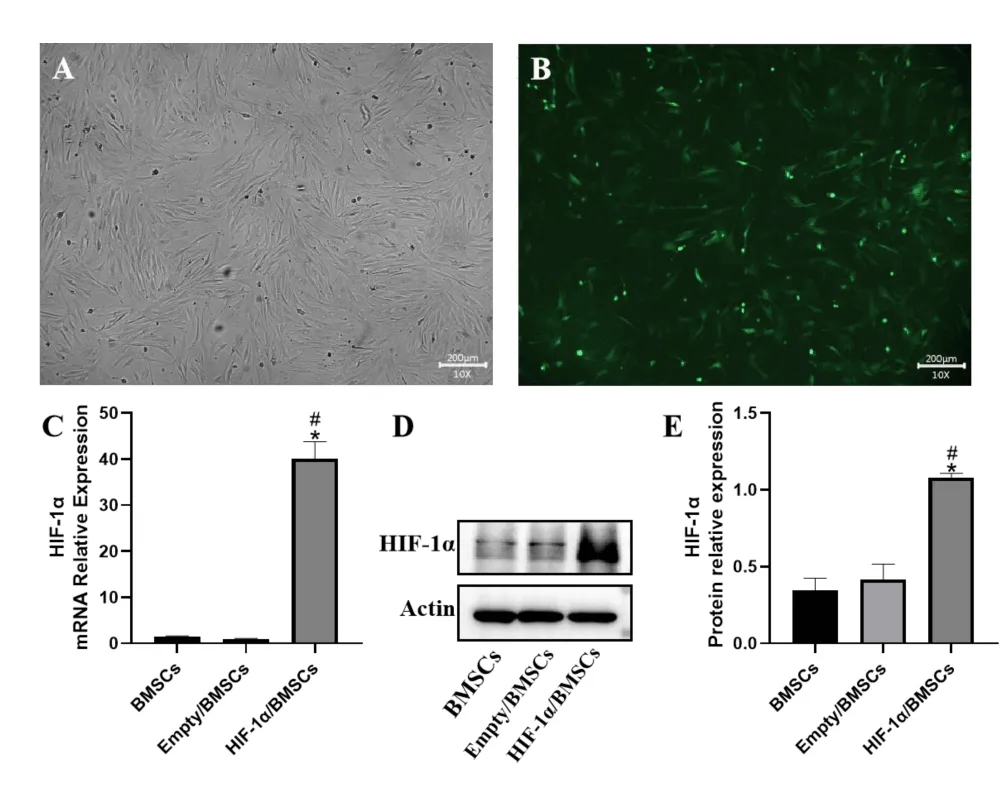
Through a set of preliminary experiments using various doses of lentivirus, we found the optimal multiplicity of infection was 30(MOI = 30). After transduction, BMSCs fluoresced green under inverted fluorescence microscopy, showing efficiency of transduction of approximately 90% (Figures 2A,B). HIF-1α mRNA and protein expression were prominent in HIF-1α/BMSCs groups compared with BMSCs group and Empty/BMSCs group by qPCR and western blot (Figures 2C-E). The quantitative results showed a significant HIF-1α mRNA and HIF-1α protein expression in HIF-1α/BMSCs group between those two groups (*: BMSCs group vs HIF-1α/BMSCs group; #: Empty/BMSCs vs HIF-1α/BMSCs group), while BMSCs group and Empty/BMSCs group had no statistical significance. (p < 0.05).
Morphology and characterization of i-ECs
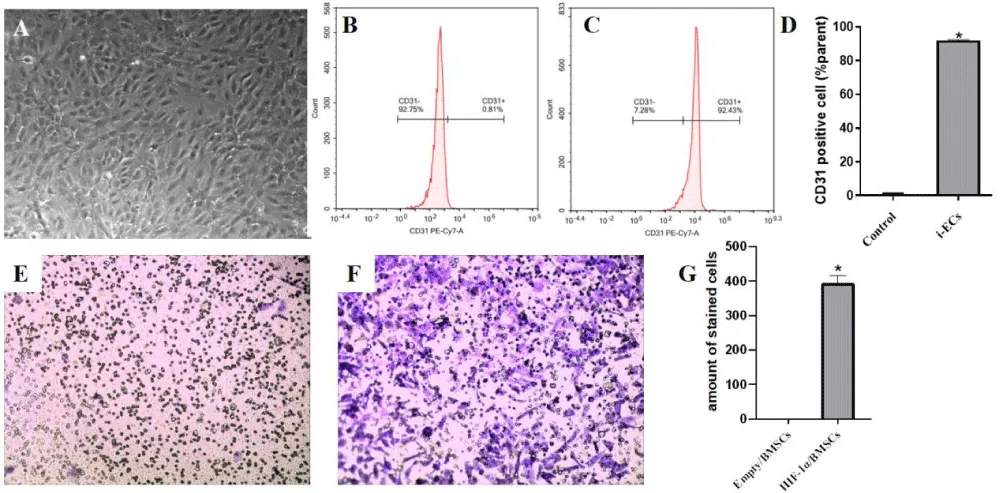
Figure 3 showed cell morphology and flow cytometry results of i-ECs. It seemed that i-ECs were more oval, short and swirling grew (Figure 3A), presenting a cobblestone-like, while HIF-1α/BMSCs were spindle-shaped spreading and long (Figure 2A). Flow cytometry results showed that HIF-1α/BMSCs without induced had only 0.81% CD31 positive expression (Figure 3B), but after differentiated into i-ECs by endothelial culture medium which expressed 92.43% (Figure 3C). Quantitative results (Figure 3D) showed a significant difference in differentiated group (i-ECs group) and non-differentiated group by counting CD31 positive expression (p < 0.05), which indicated BMSCs transduced by HIF-1α had a great superiority to differentiated into endothelial cells under experimental condition. Transwell assay showed that HIF-1α could recruit i-ECs in vitro, with an average of over 400 i-ECs migrating per field of view in HIF-1α/BMSCs group (Figure 3E). Meanwhile no migrating i-ECs in control group (Figure 3F), and quantitative results (Figure 3G) showed a significant difference between two groups (p < 0.05).
Characteristics of osteogenesis cell sheets
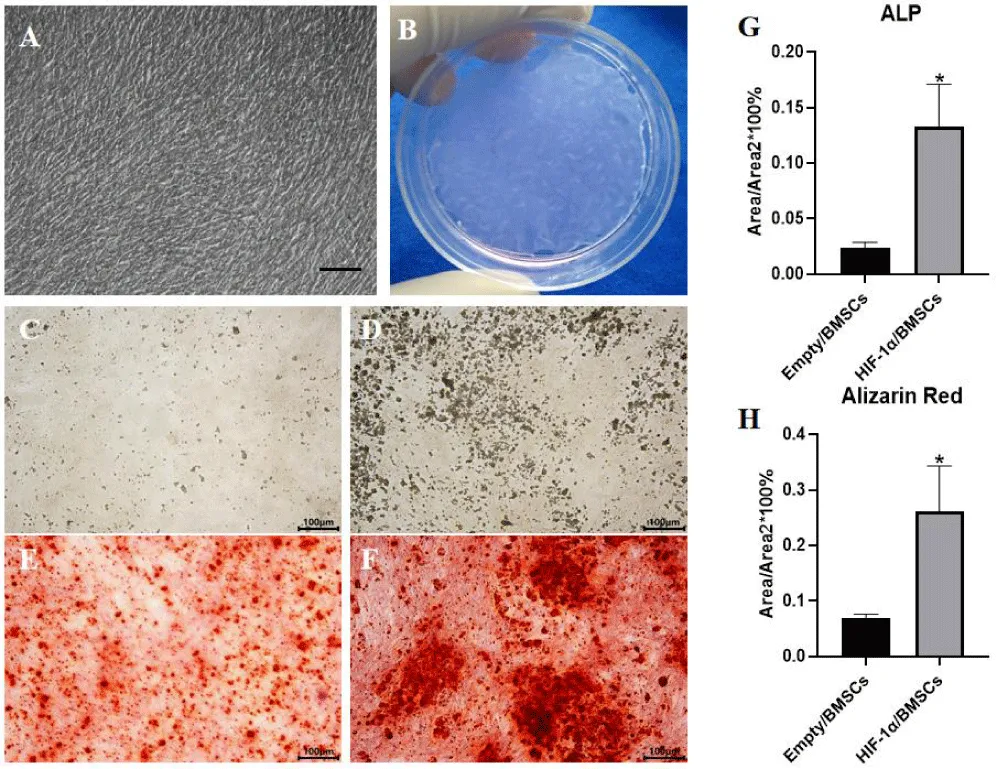
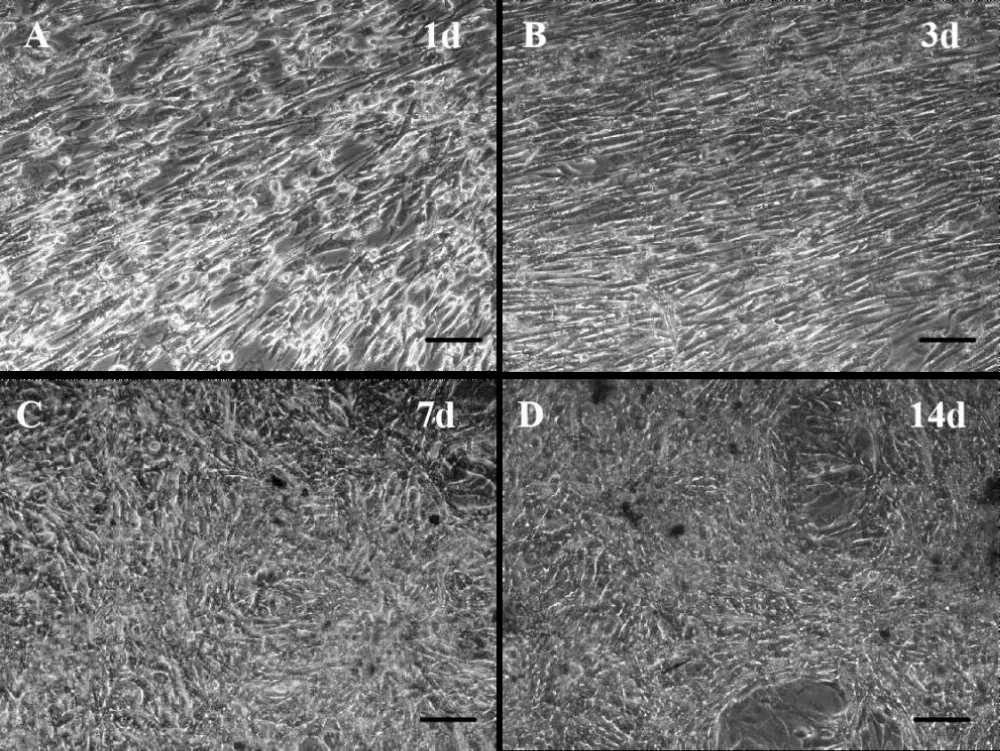
HIF-1α/BMSCs were cultured in osteogenesis medium for 21 days to fabricate Osteogenesis Cell Sheets (OCTs) (Figure 4A) which performed a dense and turbid film with the edges curled inward with time increased over 21 days (Figure 4B), and sometimes it could be lifted up by point forceps. ALP (Figures 4C,D) at day 14 and Alizarin red-S staining (Figures 4E,F) at day 21 showed both two kinds of OCTs had osteogenic differentiation under the same condition, but HIF-1α/BMSCs formed OCTs (Figures 4D,F) had an obvious and tremendous osteogenic differentiation characteristic with massive stained calcium nodules deposited than Empty/BMSCs group (Figures 4C,E). Meanwhile, the quantitative results (Figures 4G,H) showed a significant osteogenic characteristic deviation between HIF-1α/BMSCs group and Empty/BMSCs group, which indicated HIF-1α could promote the potential for osteogenic differentiation.
Immunofluorescent staining for CD31 of pre-osteogenesis cell sheets
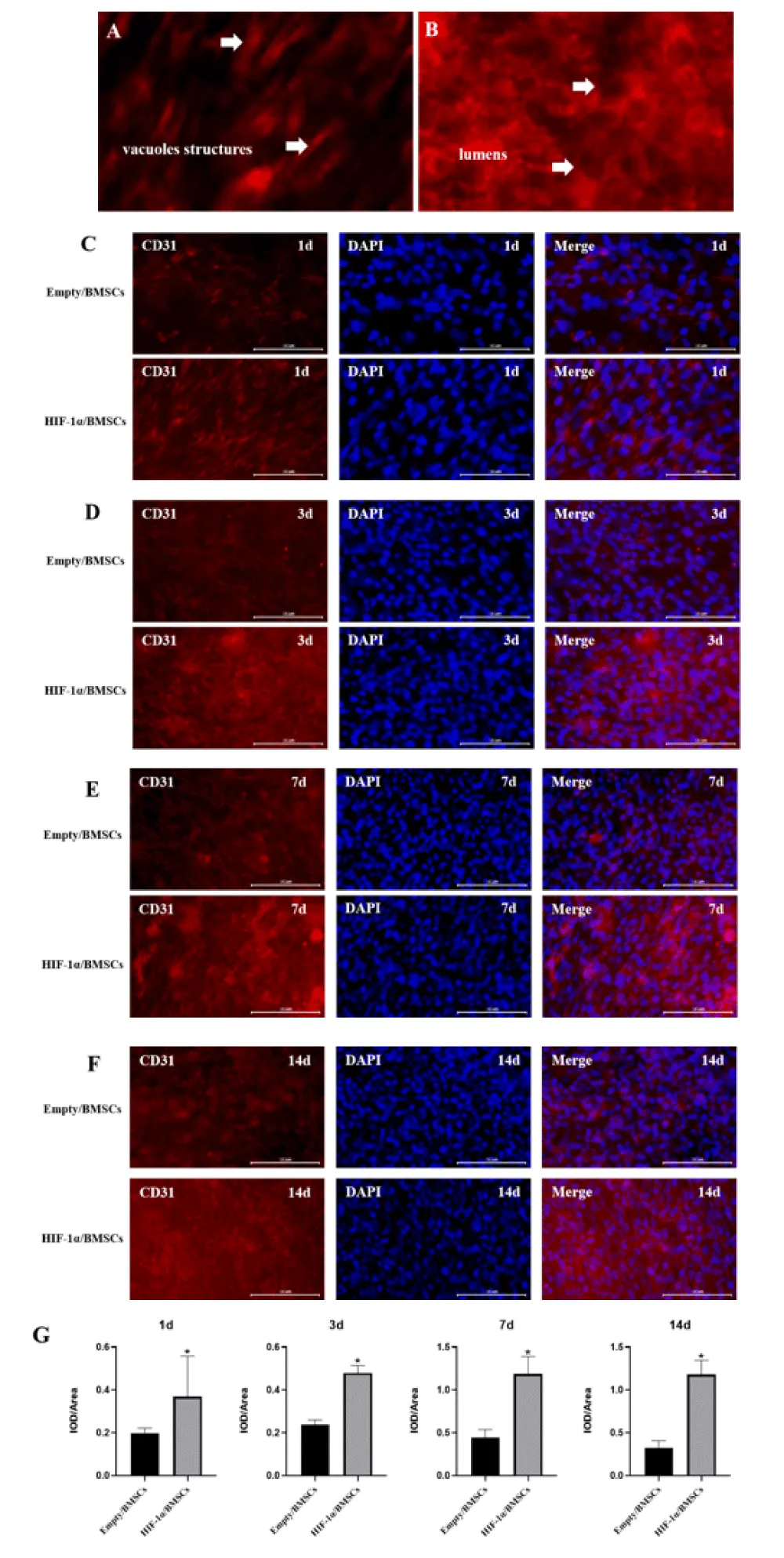
When i-ECs seeded onto OCTs to construct P-OCTs, the microscopic morphologies became so different than before. With time increased, P-OCTs showed grid multilayers and became more and more compact (Figure 5). In order to detect the role of HIF-1α involved in osteogenic-angiogenic coupling response of P-OCTs, Immunofluorescent staining for CD31 was performed to detect the networks formation of i-ECs on OCTs. The closed loop or branch network formed by continuous CD31+ signal was defined as a vacuoles structure (Figure 6A), while the hollow structure was defined as a lumen-like structure (Figure 6B). At day 1, i-ECs adhered and proliferated massively on OCTs, then they migrated fused into OCTs which showed a random arrangement and continued to form a large number of alignment with each other. The HIF-1α/BMSCs group showed a large number of i-ECs proliferation than Empty/BMSCs group, with a more and quick cell-to-cell alignment. And the vacuoles structures have been formed in HIF-1α/BMSCs group at day 1 (Figure 6C). At day 3, with the multiplication of i-ECs, they migrated reticulated and formed a large number of lumens in HIF-1α/BMSCs group (Figure 6D), while Empty/BMSCs group began to formed the vacuoles structures. At day 7, the lumens began to connect and formed stable tubulose vascular networks in HIF-1α/BMSCs group (Figure 6E). on the other hand, Empty/BMSCs group began to form the lumens. At day 14, the lumens became thicker and compact (Figure 6F). HIF-1α/BMSCs group formed a more stable and a large quantity of networks in the OCTs, while formed some sparse lumens in Empty/BMSCs group. Quantitative reselts (Figure 6G) showed a significant difference in vascular networks formation between two groups by counting CD31 positive expressing lumens at day 1,3,7,14 (p < 0.05). The networks formation was increasing by time which was highest at day 7, and it seemed that HIF-1α/BMSCs group was more than twice as Empty/BMSCs group. With time increased, the lumens had a sharp decrease in Empty/BMSCs group, but still kept a relative stability in HIF-1α/BMSCs group. At last time, the quantities of lumens in HIF-1α/BMSCs group were more than three times as Empty/BMSCs group.
Analysis of osteogenesis proteins expression
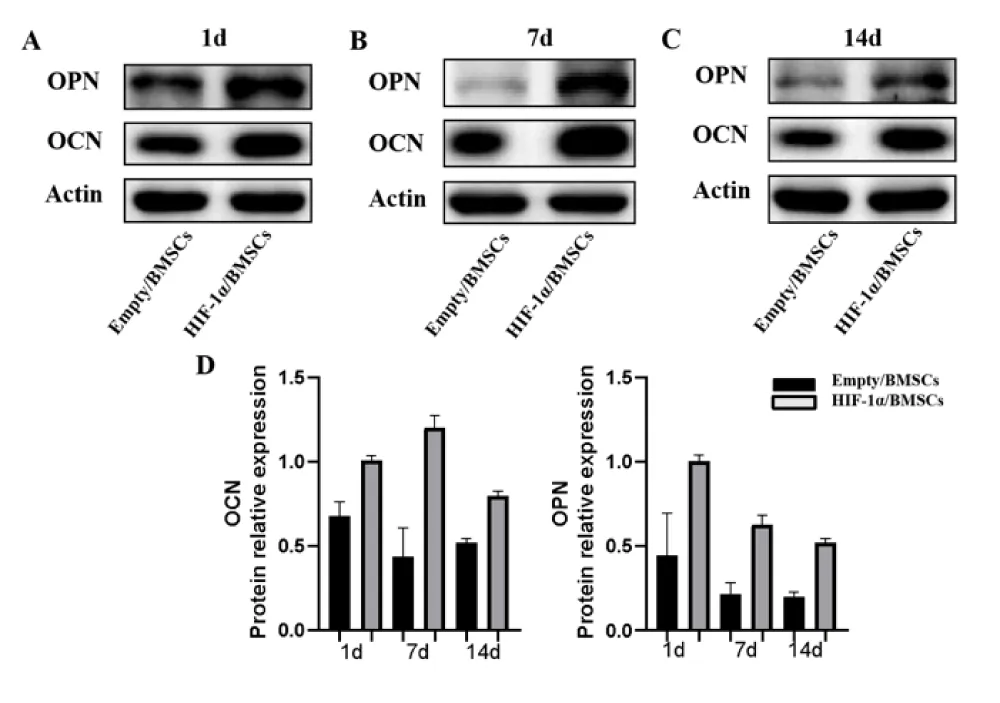
HIF-1α has profound effects on osteogenesis in OCTs. To further explore the role of HIF-1α involved in osteogenic-angiogenic coupling response of P-OCTs, we analyzed osteogenic proteins expressions of OCN, OPN by western blot (Figure 7). At day 1, the expression of OCN in Empty/BMSCs group was highest. With time increased, OCN had a down-regulated expression and stayed at a level at day 14. Meanwhile OCN in HIF-1α/BMSCs group was up-regulated and highest at day 7, then down-regulated slowly at day 14. All in all, the expressions of OCN in HIF-1α/BMSCs group were higher than Empty/BMSCs group at any time point (p < 0.05). On the other hand, the expressions of OPN were highest at day 1, and had a quick down-regulation at day 7. With time increased, the expression leveled off at day 14. On the whole, the expressions of OPN in the HIF-1α/BMSCs group were higher than Empty/BMSCs group at any time point (p < 0.05).
Discussion
Sufficient and stable vascularization plays an important role in tissue-engineered bone constructs for optimal cell survival and implants integration, but which is limited and complicated in hypoxic-ischemic conditions [16]. At this critical point, HIF-1a is an active transcription factor which plays an important role in angiogenesis and osteogenesis by up-regulating numerous genes and relevant factors [17]. Therefore, we assumed that HIF-1a-overexpressed in BMSCs to construct prevascularized osteogenic cell sheets In vitro, and we detected the effect of HIF-1a in angiogenic and osteogenic coupling response.
In order to fabricate angiogenesis, we used endothelial culture medium contained VEGF and bFGF to induce HIF-1a/BMSCs to differentiate for 14 days, then Flow Cytometric analysis was detected that over 92.43% CD31 positive expressions. However, in our previous study [18], the conversion rate was just 35.1%, which indicated that BMSCs transduced by HIF-1a had a great superiority to differentiate into endothelial cells under experimental conditions and showed a great potential for constructing angiogenesis. Then we seeded i-ECs onto Osteogenesis Cell Sheets (OCTs) to fabricate prevascularization. We observed i-ECs migrated reticulated fast and formed a large number of lumens and networks in HIF-1a/BMSCs groups (p < 0.05). On the other hand, we examined ALP at day 14 and Alizarin Red-S staining at day 21 of OCTs to detect whether HIF-1a could improve the ability of osteogenesis In vitro. The results showed an obviously osteogenic differentiation characteristic with more deep stained calcium nodules than Empty/BMSCs groups (p < 0.05). Furthermore, we checked the expressions of OCN, OPN by western blot, which showed statistical significance with Empty/BMSCs groups at any time points (p < 0.05). All in all, we concluded that HIF-1a-overexpressed in BMSCs could promote osteogenic-angiogenic coupling response by constructing prevascularized osteogenic cell sheets In vitro.
HIF signaling pathways induced by hypoxia were involved in angiogenesis and osteogenesis by modulating vital genes, such as VEGF and EPO, and was a crucial regulator that influenced final fate of bone regeneration [19,20]. Studies have been demonstrated that knockdown HIF-1a In vivo or In vitro significantly impaired bone generation and osteogenesis of periosteum-derived stem cells, indicating the indispensability of HIF-1a in bone regeneration under hypoxia [21]. In our study, we adopted 10 μg/L VEGF and 2 μg/L bFGF in M199 media to culture HIF-1a/BMSCs and construct prevascularization which showed a positive function under HIF-1a’s regulation. It has been widely reported that HIF-1a/VEGF signaling pathway plays a key role in cell proliferation, migration, angiogenesis, and osteogenesis in anoxic environment [22-24]. And recent studies have investigated the coupling of HIF-1a-driven bone formation with angiogenesis induced by VEGF upregulation in vivo [25,26], and studies have reported that activation of VEGF/AKT/mTOR signaling pathway could promote angiogenesis and differentiation of BMSCs [22]. Cobalt Chloride (CoCl2), a hypoxia-inducing agent, promoted tissue regrowth and would healing by stimulating angiogenesis through the HIF-1a/VEGF signaling pathway [27]. Meanwhile, bFGF played a major role in tissue repair and regeneration with a recognized role in epithelial and mesenchymal cell proliferation as well as a putative function in angiogenesis [28]. Furthermore, studies showed that FGF signaling could inhibit apoptosis and promote angiogenesis via HIF1a-meidated mechanism [29].
On the other hand, we fabricated OCTs by confluent culturing HIF-1a/BMSCs for 21days. As we all known, cell sheet technology takes advantages of its densely populated microenvironment which determines cell fate and function [30]. OCTs with cell sheets’ superiority that retain deposited Extracellular Matrix (ECM), cell adhesive proteins and cell-cell and cell-matrix interactions generated during confluent culture [31,32]. Studies indicated the retention of active components in ECM, such as TGF-a1, BMP-2, collagen type a, tenascin, Emilin, which had high osteoconductive capacity [33,34]. In our study, we found when i-ECs seeded onto OCTs formed by HIF-1a/BMSCs, they migrated, rearranged and formed vascular networks on OCTs efficiently. The microenvironment and crosstalk between endothelial cells and bone-forming cells are essential for successful bone regeneration, due to the endothelial cell matrix facilitates osseointegration and mineralization, in turm, osteogenic cells secrete paracrine factors and stimulate angiogenesis [35,36]. Studies found the activation of TGF-a and Notch signaling pathway in the endothelial cells during both contact and indirect co-cultivation with osteoblasts, which promoted bone formation [37].
Conclusion
HIF-1α over-expressed in BMSCs shows a great potential in angiogenic and osteogenic differentiation. On the one hand, HIF-1α/BMSCs had a great superiority to differentiated into endothelial cells under experimental condition, which could provide a steady flow of endothelial precursor cells for constructing prevascularization. In addition, i-ECs seeded onto osteogenic cell sheets showed a quick and extensive connection to each other and formed more stable tubulose vascular networks. On the other hand, HIF-1α/BMSCs showed a significant osteogenic differentiation characteristic, which had more deep stained calcium nodules deposits than the control groups. All in all, HIF-1α plays an important role in angiogenic and osteogenic coupling response of BMSCs sheets in vitro, which provides a promising strategy for bone engineering.
Acknowledgments
The authors declare that that they have not use AI-generated work in this manuscript.
Funding
This work was supported by the Nature Science Foundation of Ningxia Province (No.2023AAC03880).
Availability of Data and Meterials
Not applicable.
Authors’ Contributions
DZ, YT, WLiu, ChH and HWZ participated in the acquisition of data, analysis and interpretation of data, and writing of the manuscript. DZ participated in the study design, analysis and interpretation of data, and writing of the manuscript. All authors read and approved the final manuscript for publication.
Competing Interests
The authors declare that they have no competing interests.
Ethics Approval and Consent to Participate
The project named the Effect and Mechanism of HIF-1α on osteogenic and angiogenic coupling reaction of prevascularized BMSCs cell sheets was approved by the Nature Science Foundation of Ningxia Province (No.2023AAC03880). The Animal Ethics Committee of Yinchuan Stomatology Hospital approved all the experimental animal procedures and sample collections at September 9, 2022 and the approval nomber was YCKQLL2022018.
References
- McGovern JA, Griffin M, Hutmacher DW. Animal models for bone tissue engineering and modelling disease. Dis Model Mech. 2018 Apr 23;11(4):dmm033084. doi: 10.1242/dmm.033084. PMID: 29685995; PMCID: PMC5963860.
- Simunovic F, Finkenzeller G. Vascularization Strategies in Bone Tissue Engineering. Cells. 2021 Jul 11;10(7):1749. doi: 10.3390/cells10071749. PMID: 34359919; PMCID: PMC8306064.
- Ren L, Ma D, Liu B, Li J, Chen J, Yang D, Gao P. Preparation of three-dimensional vascularized MSC cell sheet constructs for tissue regeneration. Biomed Res Int. 2014;2014:301279. doi: 10.1155/2014/301279. Epub 2014 Jul 8. PMID: 25110570; PMCID: PMC4119697.
- Kang Y, Ren L, Yang Y. Engineering vascularized bone grafts by integrating a biomimetic periosteum and β-TCP scaffold. ACS Appl Mater Interfaces. 2014 Jun 25;6(12):9622-33. doi: 10.1021/am502056q. Epub 2014 Jun 6. PMID: 24858072; PMCID: PMC4075998.
- Mercado-Pagán ÁE, Stahl AM, Shanjani Y, Yang Y. Vascularization in bone tissue engineering constructs. Ann Biomed Eng. 2015 Mar;43(3):718-29. doi: 10.1007/s10439-015-1253-3. Epub 2015 Jan 24. PMID: 25616591; PMCID: PMC4979539.
- Ren L, Ma D, Liu B, Li J, Chen J, Yang D, Gao P. Preparation of three-dimensional vascularized MSC cell sheet constructs for tissue regeneration. Biomed Res Int. 2014;2014:301279. doi: 10.1155/2014/301279. Epub 2014 Jul 8. PMID: 25110570; PMCID: PMC4119697.
- Chen J, Zhang D, Li Q, Yang D, Fan Z, Ma D, Ren L. Effect of different cell sheet ECM microenvironment on the formation of vascular network. Tissue Cell. 2016 Oct;48(5):442-51. doi: 10.1016/j.tice.2016.08.002. Epub 2016 Aug 9. PMID: 27561623.
- Xu M, Li J, Liu X, Long S, Shen Y, Li Q, Ren L, Ma D. Fabrication of vascularized and scaffold-free bone tissue using endothelial and osteogenic cells differentiated from bone marrow derived mesenchymal stem cells. Tissue Cell. 2019 Dec;61:21-29. doi: 10.1016/j.tice.2019.08.003. Epub 2019 Aug 7. PMID: 31759403.
- de Silva L, Bernal PN, Rosenberg A, Malda J, Levato R, Gawlitta D. Biofabricating the vascular tree in engineered bone tissue. Acta Biomater. 2023 Jan 15;156:250-268. doi: 10.1016/j.actbio.2022.08.051. Epub 2022 Aug 28. PMID: 36041651.
- Rücker C, Kirch H, Pullig O, Walles H. Strategies and First Advances in the Development of Prevascularized Bone Implants. Curr Mol Biol Rep. 2016;2(3):149-157. doi: 10.1007/s40610-016-0046-2. Epub 2016 Aug 15. PMID: 27617188; PMCID: PMC4996880.
- Tsiklin IL, Shabunin AV, Kolsanov AV, Volova LT. In Vivo Bone Tissue Engineering Strategies: Advances and Prospects. Polymers (Basel). 2022 Aug 8;14(15):3222. doi: 10.3390/polym14153222. PMID: 35956735; PMCID: PMC9370883.
- Zou D, Han W, You S, Ye D, Wang L, Wang S, Zhao J, Zhang W, Jiang X, Zhang X, Huang Y. In vitro study of enhanced osteogenesis induced by HIF-1α-transduced bone marrow stem cells. Cell Prolif. 2011 Jun;44(3):234-43. doi: 10.1111/j.1365-2184.2011.00747.x. Erratum in: Cell Prolif. 2021 Jan;54(1):e12952. doi: 10.1111/cpr.12952. PMID: 21535264; PMCID: PMC6496451.
- Sun J, Shen H, Shao L, Teng X, Chen Y, Liu X, Yang Z, Shen Z. HIF-1α overexpression in mesenchymal stem cell-derived exosomes mediates cardioprotection in myocardial infarction by enhanced angiogenesis. Stem Cell Res Ther. 2020 Aug 28;11(1):373. doi: 10.1186/s13287-020-01881-7. PMID: 32859268; PMCID: PMC7455909.
- Forster R, Liew A, Bhattacharya V, Shaw J, Stansby G. Gene therapy for peripheral arterial disease. Cochrane Database Syst Rev. 2018 Oct 31;10(10):CD012058. doi: 10.1002/14651858.CD012058.pub2. PMID: 30380135; PMCID: PMC6517203.
- Chung SH, Sin TN, Ngo T, Yiu G. CRISPR Technology for Ocular Angiogenesis. Front Genome Ed. 2020 Dec 22;2:594984. doi: 10.3389/fgeed.2020.594984. PMID: 34713223; PMCID: PMC8525361.
- de Silva L, Bernal PN, Rosenberg A, Malda J, Levato R, Gawlitta D. Biofabricating the vascular tree in engineered bone tissue. Acta Biomater. 2023 Jan 15;156:250-268. doi: 10.1016/j.actbio.2022.08.051. Epub 2022 Aug 28. PMID: 36041651.
- You J, Liu M, Li M, Zhai S, Quni S, Zhang L, Liu X, Jia K, Zhang Y, Zhou Y. The Role of HIF-1α in Bone Regeneration: A New Direction and Challenge in Bone Tissue Engineering. Int J Mol Sci. 2023 Apr 28;24(9):8029. doi: 10.3390/ijms24098029. PMID: 37175732; PMCID: PMC10179302.
- Zhang D, Gao P, Li Q, Li J, Li X, Liu X, Kang Y, Ren L. Engineering biomimetic periosteum with β-TCP scaffolds to promote bone formation in calvarial defects of rats. Stem Cell Res Ther. 2017 Jun 5;8(1):134. doi: 10.1186/s13287-017-0592-4. PMID: 28583167; PMCID: PMC5460346.
- Bai H, Wang Y, Zhao Y, Chen X, Xiao Y, Bao C. HIF signaling: A new propellant in bone regeneration. Biomater Adv. 2022 Jul;138:212874. doi: 10.1016/j.bioadv.2022.212874. Epub 2022 May 18. PMID: 35913258.
- Tao J, Miao R, Liu G, Qiu X, Yang B, Tan X, Liu L, Long J, Tang W, Jing W. Spatiotemporal correlation between HIF-1α and bone regeneration. FASEB J. 2022 Oct;36(10):e22520. doi: 10.1096/fj.202200329RR. PMID: 36065633.
- Zhuang Y, Zhao Z, Cheng M, Li M, Si J, Lin K, Yu H. HIF-1α Regulates Osteogenesis of Periosteum-Derived Stem Cells Under Hypoxia Conditions via Modulating POSTN Expression. Front Cell Dev Biol. 2022 Feb 17;10:836285. doi: 10.3389/fcell.2022.836285. PMID: 35252198; PMCID: PMC8891937.
- Song S, Zhang G, Chen X, Zheng J, Liu X, Wang Y, Chen Z, Wang Y, Song Y, Zhou Q. HIF-1α increases the osteogenic capacity of ADSCs by coupling angiogenesis and osteogenesis via the HIF-1α/VEGF/AKT/mTOR signaling pathway. J Nanobiotechnology. 2023 Aug 7;21(1):257. doi: 10.1186/s12951-023-02020-z. PMID: 37550736; PMCID: PMC10405507.
- Guo Q, Yang J, Chen Y, Jin X, Li Z, Wen X, Xia Q, Wang Y. Salidroside improves angiogenesis-osteogenesis coupling by regulating the HIF-1α/VEGF signalling pathway in the bone environment. Eur J Pharmacol. 2020 Oct 5;884:173394. doi: 10.1016/j.ejphar.2020.173394. Epub 2020 Jul 27. PMID: 32730833.
- Mao J, Liu J, Zhou M, Wang G, Xiong X, Deng Y. Hypoxia-induced interstitial transformation of microvascular endothelial cells by mediating HIF-1α/VEGF signaling in systemic sclerosis. PLoS One. 2022 Mar 1;17(3):e0263369. doi: 10.1371/journal.pone.0263369. PMID: 35231032; PMCID: PMC8887755.
- Maes C, Araldi E, Haigh K, Khatri R, Van Looveren R, Giaccia AJ, Haigh JJ, Carmeliet G, Schipani E. VEGF-independent cell-autonomous functions of HIF-1α regulating oxygen consumption in fetal cartilage are critical for chondrocyte survival. J Bone Miner Res. 2012 Mar;27(3):596-609. doi: 10.1002/jbmr.1487. PMID: 22162090.
- Liu K, Shi L, Wang S, Yalikun A, Hamiti Y, Yusufu A. [Effect of accordion technique and deferoxamine on promoting bone regeneration in distraction osteogenesis]. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2024 Aug 15;38(8):1001-1009. Chinese. doi: 10.7507/1002-1892.202404073. PMID: 39175324; PMCID: PMC11335587.
- Rathinasamy VS, Paneerselvan N, Jagadeeshan S, Erusan RR, Manjunathan R. Cobalt Chloride-Induced Tissue Regeneration and Wound Healing Depend on HIF-1α and VEGF-A-Mediated Neovascularization. Zebrafish. 2025 Oct 15. doi: 10.1177/15458547251383496. Epub ahead of print. PMID: 41091550.
- Farooq M, Khan AW, Kim MS, Choi S. The Role of Fibroblast Growth Factor (FGF) Signaling in Tissue Repair and Regeneration. Cells. 2021 Nov 19;10(11):3242. doi: 10.3390/cells10113242. PMID: 34831463; PMCID: PMC8622657.
- Chen K, Rao Z, Dong S, Chen Y, Wang X, Luo Y, Gong F, Li X. Roles of the fibroblast growth factor signal transduction system in tissue injury repair. Burns Trauma. 2022 Mar 23;10:tkac005. doi: 10.1093/burnst/tkac005. PMID: 35350443; PMCID: PMC8946634.
- De Pieri A, Rochev Y, Zeugolis DI. Scaffold-free cell-based tissue engineering therapies: advances, shortfalls and forecast. NPJ Regen Med. 2021 Mar 29;6(1):18. doi: 10.1038/s41536-021-00133-3. PMID: 33782415; PMCID: PMC8007731.
- Bou-Ghannam S, Kim K, Grainger DW, Okano T. 3D cell sheet structure augments mesenchymal stem cell cytokine production. Sci Rep. 2021 Apr 14;11(1):8170. doi: 10.1038/s41598-021-87571-7. PMID: 33854167; PMCID: PMC8046983.
- Thummarati P, Laiwattanapaisal W, Nitta R, Fukuda M, Hassametto A, Kino-Oka M. Recent Advances in Cell Sheet Engineering: From Fabrication to Clinical Translation. Bioengineering (Basel). 2023 Feb 6;10(2):211. doi: 10.3390/bioengineering10020211. PMID: 36829705; PMCID: PMC9952256.
- Miar S, Pearson J, Montelongo S, Zamilpa R, Betancourt AM, Ram B, Navara C, Appleford MR, Ong JL, Griffey S, Guda T. Regeneration enhanced in critical-sized bone defects using bone-specific extracellular matrix protein. J Biomed Mater Res B Appl Biomater. 2021 Apr;109(4):538-547. doi: 10.1002/jbm.b.34722. Epub 2020 Sep 11. PMID: 32915522; PMCID: PMC8740960.
- Onishi T, Shimizu T, Akahane M, Omokawa S, Okuda A, Kira T, Inagak Y, Tanaka Y. Osteogenic extracellular matrix sheet for bone tissue regeneration. Eur Cell Mater. 2018 Aug 2;36:68-80. doi: 10.22203/eCM.v036a06. PMID: 30069865.
- He Y, Wang W, Lin S, Yang Y, Song L, Jing Y, Chen L, He Z, Li W, Xiong A, Yeung KWK, Zhao Q, Jiang Y, Li Z, Pei G, Zhang ZY. Fabrication of a bio-instructive scaffold conferred with a favorable microenvironment allowing for superior implant osseointegration and accelerated in situ vascularized bone regeneration via type H vessel formation. Bioact Mater. 2021 Aug 12;9:491-507. doi: 10.1016/j.bioactmat.2021.07.030. Erratum in: Bioact Mater. 2022 May 27;20:164. doi: 10.1016/j.bioactmat.2022.04.010. PMID: 34820585; PMCID: PMC8586756.
- Rao RR, Peterson AW, Ceccarelli J, Putnam AJ, Stegemann JP. Matrix composition regulates three-dimensional network formation by endothelial cells and mesenchymal stem cells in collagen/fibrin materials. Angiogenesis. 2012 Jun;15(2):253-64. doi: 10.1007/s10456-012-9257-1. Epub 2012 Mar 2. PMID: 22382584; PMCID: PMC3756314.
- Perepletchikova D, Kuchur P, Basovich L, Khvorova I, Lobov A, Azarkina K, Aksenov N, Bozhkova S, Karelkin V, Malashicheva A. Endothelial-mesenchymal crosstalk drives osteogenic differentiation of human osteoblasts through Notch signaling. Cell Commun Signal. 2025 Feb 19;23(1):100. doi: 10.1186/s12964-025-02096-0. Erratum in: Cell Commun Signal. 2025 Mar 12;23(1):133. doi: 10.1186/s12964-025-02118-x. PMID: 39972367; PMCID: PMC11841332.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.