Medicine Group. 2024 September 20;5(9):1132-1139. doi: 10.37871/jbres2001.
Reviewing Clinical Trials and Meta-Analyses to Assess the Efficacy and Safety of Huachansu in Cancer Treatment
Aline Genbauffe1, Laura Soumoy1,2, Florence Lefranc3 and Fabrice Journe1,4*
2National Institute of Health and Medical Research (INSERM) U981, Gustave Roussy Cancer Campus, Villejuif, France
3Department of Neurosurgery, University Hospital of Brussels (H.U.B), CUB Erasme Hospital, Free University of Brussels (ULB), Brussels, Belgium
4Laboratory of Clinical and Experimental Oncology, University Hospital of Brussels (H.U.B), Jules Bordet Institute, Free University of Brussels (ULB), Brussels, Belgium
Abstract
Traditional Chinese Medicine (TCM), including herbal remedies and animal secretions like HuaChansu, also called Cinobufacini, has gained attention for its therapeutic efficacy in cancer treatment. HuaChansu, extracted from the skin of toads from the genus Bufo, has been used traditionally for various diseases and recently explored for its anti-cancer properties attributed to bufadienolides like bufalin, resibufogenin, and cinobufagin. Clinical studies and meta-analyses have evaluated its efficacy and safety across various cancers. For hepatocellular carcinoma, non-small cell lung cancer, and pancreatic cancer, pilot studies and clinical trials showed HuaChansu's potential for disease stabilization with minimal toxicity. In breast cancer, a meta-analysis suggested that HuaChansu combined with chemotherapy improves response rates and quality of life. Studies on gallbladder and gastric cancers indicated similar benefits, enhancing therapeutic effects and reducing side effects. In lung cancer, HuaChansu improved survival rates and quality of life when combined with chemotherapy, though placebo-controlled trials are needed. For liver cancer, meta-analyses and randomized controlled trials demonstrated improved survival rates and reduced recurrence when HuaChansu was used with transarterial chemoembolization. These findings underscore HuaChansu potential as a complementary cancer treatment, warranting further research to optimize its dosage and administration for enhanced efficacy.
Introduction
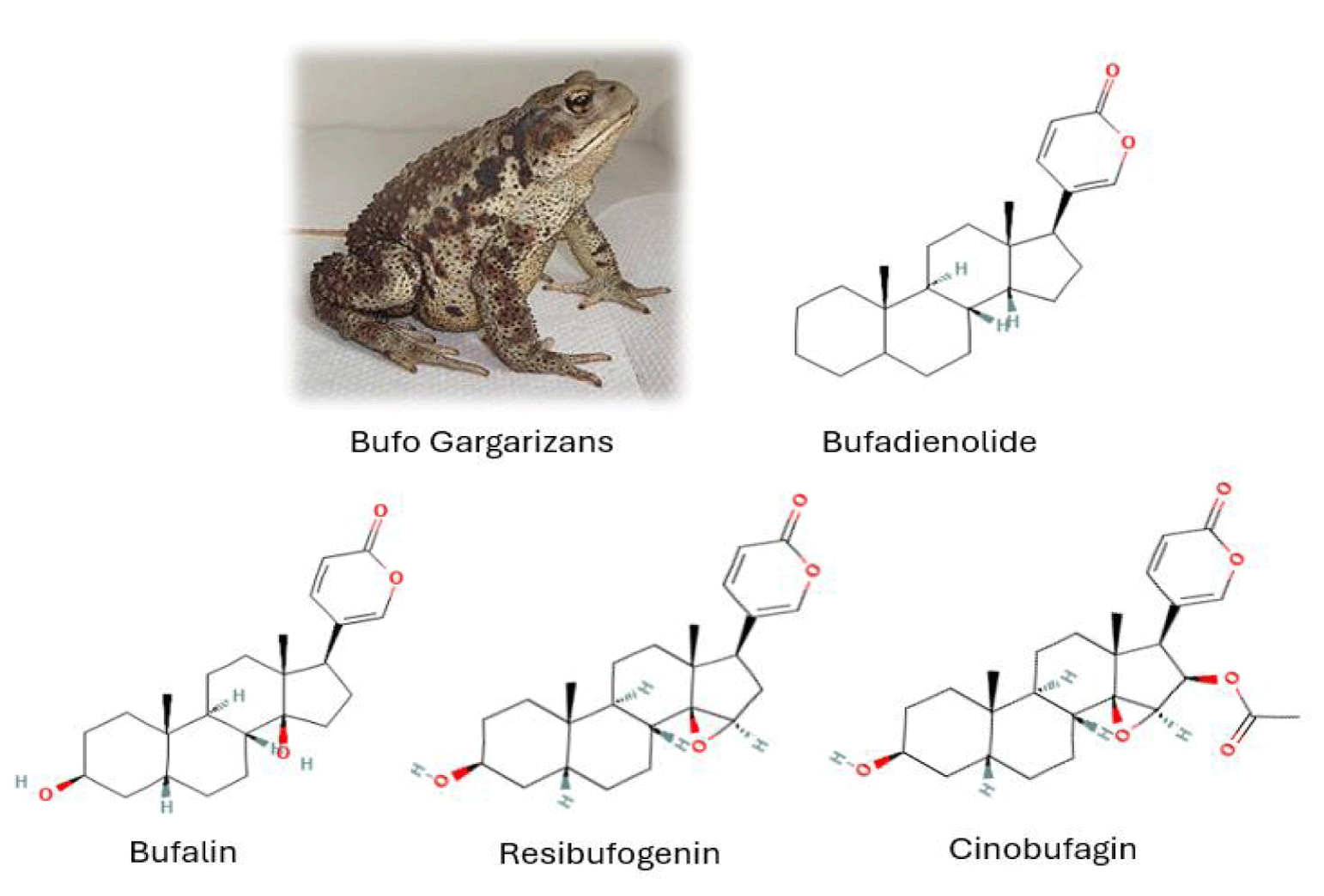
Traditional Chinese Medicine (TCM) is widely utilized in China and is based on plants and animal secretions. Its cost-effectiveness and therapeutic efficacy have drawn attention to this alternative medicine in Europe and the United States [1]. HuaChansu, also known as Cinobufacini, is the most common TCM and has been approved by Chinese NMPA since the 1990s. This water-soluble preparation, extracted from the skin and parotid venom glands of Bufo bufo gargarizans, a subspecies of the common toad found primarily in East Asia, is mainly composed of indole alkaloids (bufotenine, bufotenidine, cinobufotenine and serotonin) and steroidal cardiac glycosides [2], and was first used as an adjuvant for heart failure treatment [3]. Subsequently, it has been used as a detoxicant, analgesic, diuretic, hemostatic, anti-inflammatory, and cardiotonic agent. Recently, anti-cancer properties such as inhibition of cell proliferation, induction of cell differentiation, of apoptosis, and inhibition of cancer angiogenesis have been discovered and attributed to the action of bufadienolides [4,5] and cardiac glycosides, enabling them to be redirected towards cancer treatment [6]. Among these molecules, the cardiac glycosides bufalin, resibufogenin, and cinobufagin are the three main active components [7], demonstrating anti-cancer activities, as we previously reported in melanoma cell lines [5,8,9] (Figure 1).
Article Search Strategy
We conducted a search in PubMed and reviewed references from relevant articles to identify studies on the use of HuaChansu in cancer treatment. Only publications written in English between 2003 and 2024 were included, with a particular focus on the last five years. Clinical studies were screened based on the presence of a database entry, abstract, available full text, or title referring to these conditions. The following keywords were used: HuaChansu, Cinobufacini, cancer, clinical study, and trial. After a critical analysis of the publication content, the final selection of articles was included in this review. The study was conducted according to the PRISMA checklist for reviews and meta-analyses. Institutional review board approval was not required.
Initial study
A pilot study by the Fudan University Cancer Hospital (Shanghai, China) and the MD Anderson Cancer Centre (Houston, Texas, USA) explored the efficacy and side effects of HuaChansu in treating hepatocellular carcinoma, non-small cell lung cancer, and pancreatic cancer. The study enrolled 15 patients who received intravenous HuaChansu for 14 days followed by 7 days off. Six patients had stable disease (median duration = 6.0 months; range 3.5-11.1 months), and one hepatocellular carcinoma patient experienced a 20% reduction in tumor mass lasting over 11 months. In addition, 73% of patients had no toxicities greater than grade I, with no grades III and IV observed, despite using doses eight times higher than conventional doses in China. Grade I toxicities included hematological (leukopenia, thrombocytopenia), mucocutaneous (oral ulcer, rash), gastrointestinal (loss of appetite, constipation, diarrhoea), cardiovascular (premature ventricular contraction, hypertension), and respiratory (dyspnoea) effects. Electrocardiograms showed no cardiac dysfunction. This study highlighted that HuaChansu is well tolerated at higher concentrations and results in disease stabilization when used as a single agent [7].
Following this first observation, a phase II/III clinical trial (NCT00837239) was conducted to study the intravenous combination of gemcitabine and HuaChansu in pancreatic cancer. The study compared the effects of gemcitabine and HuaChansu versus gemcitabine and placebo in 76 patients with locally advanced or metastatic pancreatic adenocarcinoma (39 in the experimental group vs., 37 in the control group). No additional toxicity was induced by the combination, but no benefit was observed in terms of objective radiographical response rate, Progression-Free Survival (PFS), and time to progression. The lack of effectiveness could be due to several factors: the difficulty in treating advanced pancreatic cancer, a possibly too low dose of HuaChansu, potentially too short exposure time, and the type of administration [10].
Breast cancer
A meta-analysis of 16 studies involving 1331 patients evaluated the safety and efficacy of HuaChansu combined with chemotherapy. Most studies indicated that the combination improved the Objective Response Rate (ORR) and clinical benefit rate. The quality of life was enhanced by decreasing pain and side effects such as kidney damage. The tumor markers CA 15-3, CA 125 and CEA were also reduced with the combination compared to chemotherapy alone. However, attention must be paid to the quality of the meta-analysis, as patient pathological stages, HuaChansu dosages, and chemotherapy treatments varied across the studies [11].
Gallbladder cancer
Gallbladder carcinoma, often diagnosed at an advanced stage, is conventionally treated with surgery and chemotherapies [12]. The 5-year survival rate is less than 5%, with high risks of relapse [13]. A study analysed the efficacy and safety of combining Gemcitabine and Oxaliplatin (GEMOX) with HuaChansu, administered three days before chemotherapy cycles. Out of 23 patients, 8 (34.8%) showed a partial response, and 7 (30.4%) had stable disease. This combination was well tolerated, with myelosuppression being the main adverse effect, and improved patient quality of life with reduced pain [12].
Gastric cancer
China accounts for 43% of new gastric cancer cases worldwide, with a poor prognosis and a 5-year survival rate lower than 20% [14,15]. Two meta-analyses (12 studies, 853 patients, and 14 studies, 976 patients) investigated the combination of HuaChansu and chemotherapy versus chemotherapy alone. They found improved response rates and quality of life, with increased Karnofsky Performance Scale (KPS) scores and reduced cancer-related pain [16,17]. Another meta-analysis (27 studies, 1939 patients) showed that combining HuaChansu with chemotherapy improved therapeutic effects and alleviated side effects such as nausea, vomiting, leukopenia, anemia, and gastrointestinal effects [18].
A Bayesian network meta-analysis, comparing Oxaliplatin and Tegafur (SOX) chemotherapy regimens with the combination of SOX and TCM, indicates that HuaChansu is the TCM that demonstrates the highest clinical effectiveness (Surface Under the Cumulative Ranking curve (SUCRA): 78.71%) compared to other TCM by significantly decreasing leukopenia (SUCRA: 93.35%), thrombocytopenia (SUCRA: 80.19%), nausea and vomiting (SUCRA 95.15%) incidence induced by SOX chemotherapy [19].
Lung cancer
Lung cancer is the leading cause of cancer-related death in China, with rates projected to increase by 40% by 2030 [15]. A clinical trial involving 121 patients (65 in the treatment group vs., 56 in the control group) investigated the efficacy of HuaChansu combined with chemotherapy across different stages of lung cancer. Stage III patient survival significantly improved in the experimental group (19.3 months vs., 12.7 months) [20]. However, the absence of a placebo control warrants caution. Another study with 80 patients found that combining chemotherapy with HuaChansu injection reduced leukopenia incidence, however no differences were observed for other adverse effects like diarrhoea and vomiting [21]. Two additional clinical trials explored HuaChansu as maintenance therapy post-chemotherapy [19]. The first study indicated improved 1-year survival (78.1% vs., 53.1%) and quality of life, with reductions in fatigue, nausea, vomiting, appetite loss, and alopecia [22]. The second one showed a reduction in CTLA-4 concentration in serum after two HuaChansu cycle treatment [23].
Two meta-analyses of HuaChansu combined with platinum-based chemotherapy reported improvements in 1- and 2-year survival rates, objective tumor response rates, and quality of life by reducing side effects (27 studies, 2125 patients [24] and 19 studies, 1564 patients [25]). Another meta-analysis (32 studies, 2753 patients) found that administering HuaChansu before chemotherapy could enhance survival rates and quality of life by reducing alopecia, thrombocytopenia, and gastrointestinal effects [26].
Cinobufatolin was also assessed for the treatment of non-small cell lung cancer in meta-analyses [27,28]. One analysis indicates that intravenous infusion of Cinobufatolin improved the therapeutic effects of chemotherapy [27]. Indeed, the 1-2-3-year survival rates and quality of life were enhanced with Cinobufatolin injection [27]. The latest analysis (21 studies, 1735 patients) evaluated the safety and effectiveness of Cinobufatolin injection as an adjunctive treatment [28]. The findings suggest that the injection was safe, with 73% of patients experiencing no drug-related toxicity greater than grade I [28]. Moreover, chemotherapy efficacy was enhanced, as evidenced by improved ORR, disease control, and quality of life, achieved by reducing chemotherapy-related adverse effects [28].
Finally, a recent overview of systematic reviews and meta-analyses assessed the efficacy and safety of HuaChansu as an adjuvant therapy for non-small cell lung cancer based on Systematic Reviews (SRs) and Meta-Analyses (MAs) [29]. The results indicate generally low quality of the included SRs/MAs with significant shortcomings in reporting and risk of bias. In this context, while HuaChansu shows promise, higher quality SRs/MAs and randomized control trials are necessary to confirm its effectiveness and safety [29].
Liver cancer
Liver cancer, the sixth most common cancer and third leading cause of cancer-related death worldwide [15], is often treated with Transarterial Chemoembolization (TACE) at advanced stage [30]. This treatment reduces tumor blood supply by blocking hepatic arteria and gives high doses of chemotherapies to the tumor [30]. A meta-analysis (15 studies, 1225 patients) indicated that HuaChansu combined with TACE improved short-term effectiveness rates, quality of life, and 1- and 2-year survival rates compared to TACE alone [31]. This combination improves also the ORR and the 2-year survival rate in middle and advanced liver cancer stages [32]. A single-center randomized controlled trial (NCT01715532) compared HuaChansu combined with TACE versus TACE alone in unresectable hepatocellular carcinoma [33]. Results indicated significantly improved median PFS (6.8 months vs., 5.3 months; p = 0.029) and Overall Survival (OS) (14.8 months vs., 10.7 months; p = 0.025) with HuaChansu treatment [33]. Of note, the limitations of this trial include the non-blinded study design, data from a single center, and all patients having preserved liver functions [33].
Chemotherapeutic agents can also be replaced by HuaChansu in TACE [31]. Its effectiveness was reported in a meta-analysis (7 studies, 360 patients) where the authors found that HuaChansu could improve the quality of life and lower the grade I hepatotoxicity compared with traditional chemotherapeutic agent used during TACE [31]. A retrospective study (56 patients; 31 patients in HuaChansu TACE group vs., 25 patients in Epirubicin TACE group) indicates also that HuaChansu could improve the efficacy of TACE by improving ORR in large hepatocellular carcinoma, better than Epirubicin as chemotherapeutic agent [34].
HuaChansu role in preventing recurrence post-surgery and TACE was also studied [35]. A multicenter randomized controlled trial (ChiCTR-TRC-07000033) with 379 patients across five Chinese hospitals compared HuaChansu treatment (intravenous injection) and TACE after small hepatocellular carcinoma resection [35]. HuaChansu treatment reduced recurrence risk by 30.5%, prolonged median recurrence-free survival, and reduced adverse effects like fatigue, fever, upper abdominal pain, and nausea compared to TACE alone [35]. A retrospective case-controlled study found that intravenous HuaChansu for 15 days increased PFS rates (p < 0.01) and OS rates (p < 0.045), indicating longer times before recurrence and metastasis after surgery compared to TACE alone [36].
A retrospective study analysed also the benefits of using versus Cisplatin and 5-Fluorouracil (CF) with Hepatic Arterial Infusion (HAI). The latter one allows to directly deliver drugs in hepatic arteria and reduces systemic toxicities due to first-pass metabolism in liver. This study including 130 patients (69 patients in HuaChansu group vs., 61 patients in CF group) indicates that ORR increased by 17% while the OS and PFS decreased by 39% and 42%, respectively, for patients treated with HuaChansu [37].
Discussion
The impact of HuaChansu and its active components, bufadienolides, in cancer treatment, particularly through data from clinical trials and meta-analyses, has yielded promising results across various types of cancers, including hepatocellular carcinoma, non-small cell lung cancer, breast cancer, gallbladder cancer, gastric cancer, and liver cancer [7-34]. HuaChansu, as an additional treatment or used alone, has demonstrated potential in stabilizing disease, improving survival rates, enhancing quality of life, and reducing chemotherapy-related side effects. Table 1 reviews the effects of HuaChansu and HuaChansu in clinical studies.
| Table 1: Overview of the effects of HuaChansu treatments on clinical parameters across different cancer types in clinical trials. | |||||
| Cancer Type | Treatment | Overall Response Rate | Quality of Life | Side Effects | References |
| Hepatocellular carcinoma, Non-small cell lung cancer, Pancreatic cancer | HuaChansu (IV) | Disease stabilization (6 out of 15 patients), 20% reduction in tumor mass in 1 hepatocellular carcinoma patient | Improved (reduced toxicities greater than grade I) | Mainly Grade I toxicities observed (hematological, mucocutaneous, gastrointestinal, cardiovascular, respiratory effects) | [7] |
| Pancreatic cancer | HuaChansu (IV) + Gemcitabine | No benefit in ORR, PFS, and time to progression | Not reported | No additional toxicity induced by the combination | [10] |
| Breast cancer | HuaChansu (IV) + Chemotherapy (Docetaxel, Pirarubicin, Cyclophosphamide, Epirubicin, Vinorelbine, Capecitabine, 5-FU) | Improved ORR and clinical benefit rate | Enhanced (reduced tumor markers) | Not reported | [11] |
| Gallbladder cancer | HuaChansu (IV) + GEMOX | Partial response in 34.8% of patients, stable disease in 30.4% of patients | Improved (reduced pain, improved quality of life) | Mainly myelosuppression | [12] |
| Gastric cancer | HuaChansu (IV) + Chemotherapy (Oxaliplatin, Epirubicin, Floxuridine, Etoposide, Cisplatin, Capecitabine, Leucovorin, Paclitaxel) | Improved response rates, quality of life, KPS scores, and reduced cancer-related pain | Enhanced (reduced side effects such as nausea, vomiting, leukopenia, anemia, and gastrointestinal effects) | Not reported | [16-18] |
| Lung cancer | HuaChansu (PO and IV) + Chemotherapy (Docetaxel, Gemcitabine, Permetrexed, Cisplatin, Etoposide, Vinorelbine, Paclitaxel) | Improved survival in Stage III patients | Enhanced | Reduced leukopenia incidence, no differences observed for adverse effects like diarrhea and vomiting | [20-28] |
| Liver cancer | HuaChansu (PO or IV) + TACE | Improved short-term effectiveness rates, quality of life, 1-2 year survival rates, ORR | Not reported | Not reported | [31-35] |
| IV: Intravenous injection, PO: Per Os administration, RCT: Randomized Controlled Trial; [7]:Phase I; [10]: Phase II; [11,16-18,20-22,24,25,27,28,31-33,35]: Phase III. | |||||
The pilot study conducted in 2009 by the Fudan University Cancer Hospital and the MD Anderson Cancer Centre highlighted the tolerability and efficacy of HuaChansu at higher doses, showing significant disease stabilization and manageable toxicity levels in patients with hepatocellular carcinoma, non-small cell lung cancer, and pancreatic cancer [7]. This paved the way for a phase II clinical trial which, despite not showing significant improvement in objective response rates or PFS, pointed out areas for further research, including dose optimization and administration methods.
Meta-analyses on breast cancer and gastric cancer revealed that combining HuaChansu with chemotherapy improved ORR, clinical benefit rates, and quality of life, underscoring the potential synergistic effects of this component [11]. The studies on gallbladder and lung cancer further support the use of HuaChansu in combination with conventional chemotherapy, showing improved survival rates and reduced side effects, but the lack of placebo controls in some studies necessitates cautious interpretation [10,17-26].
The extensive research on liver cancer, involving combinations of HuaChansu with TACE and other treatments, has shown significant improvements in short-term effectiveness rates, survival rates, and quality of life [29-32]. These studies emphasize HuaChansu’s role in enhancing the efficacy of TACE and reducing adverse effects, highlighting its potential in managing advanced liver cancer stages and preventing post-surgery recurrence [33,34].
While these findings are encouraging, several limitations must be addressed. The variability in study designs, patient populations, dosages, and treatment protocols across different trials necessitates standardization for more definitive conclusions. Furthermore, the absence of placebo controls and the need for longer follow-up periods in some studies indicate areas for improvement in future research.
Conclusion
The clinical trials and meta-analysis studies reviewed demonstrate the promising role of HuaChansu and bufadienolides in cancer treatment (Figure 1). The combination of HuaChansu with conventional therapies has shown potential in improving survival rates, stabilizing disease progression, and enhancing the quality of life for patients with various types of cancer. However, further research is necessary to optimize dosages, administration methods, and treatment protocols. Standardizing study designs and including placebo controls will enhance the reliability of future findings. Overall, HuaChansu represents a valuable addition to the arsenal of cancer treatments, warranting continued investigation and integration into clinical practice.
Acknowledgement
The authors would like to thank the University of Mons (UMONS) for funding the PhD grant for Aline Genbauffe, as well as the “UMONS Research Institute for Health Sciences and Technology” and the “Association Jules Bordet” for their financial support. They also acknowledge Prof. Jean-Marie Colet (Laboratory of Human Biology and Toxicology, UMONS) for reading and commenting on the manuscript.
References
- Wang CY, Bai XY, Wang CH. Traditional Chinese medicine: a treasured natural resource of anticancer drug research and development. Am J Chin Med. 2014;42(3):543-59. doi: 10.1142/S0192415X14500359. PMID: 24871650.
- Zuo Q, Xu DQ, Yue SJ, Fu RJ, Tang YP. Chemical Composition, Pharmacological Effects and Clinical Applications of Cinobufacini. Chin J Integr Med. 2024 Apr;30(4):366-378. doi: 10.1007/s11655-024-3708-6. Epub 2024 Jan 12. PMID: 38212503.
- Qi F, Zhao L, Zhou A, Zhang B, Li A, Wang Z, Han J. The advantages of using traditional Chinese medicine as an adjunctive therapy in the whole course of cancer treatment instead of only terminal stage of cancer. Biosci Trends. 2015 Feb;9(1):16-34. doi: 10.5582/bst.2015.01019. PMID: 25787906.
- Zhan X, Wu H, Wu H, Wang R, Luo C, Gao B, Chen Z, Li Q. Metabolites from Bufo gargarizans (Cantor, 1842): A review of traditional uses, pharmacological activity, toxicity and quality control. J Ethnopharmacol. 2020 Jan 10;246:112178. doi: 10.1016/j.jep.2019.112178. Epub 2019 Aug 21. PMID: 31445132.
- Soumoy L, Ghanem GE, Saussez S, Journe F. Bufalin for an innovative therapeutic approach against cancer. Pharmacol Res. 2022 Oct;184:106442. doi: 10.1016/j.phrs.2022.106442. Epub 2022 Sep 9. PMID: 36096424.
- Nik Nabil WN, Dai R, Liu M, Xi Z, Xu H. Repurposing cardiac glycosides for anticancer treatment: a review of clinical studies. Drug Discov Today. 2024 Aug 3;29(10):104129. doi: 10.1016/j.drudis.2024.104129. Epub ahead of print. PMID: 39098384.
- Meng Z, Yang P, Shen Y, Bei W, Zhang Y, Ge Y, Newman RA, Cohen L, Liu L, Thornton B, Chang DZ, Liao Z, Kurzrock R. Pilot study of huachansu in patients with hepatocellular carcinoma, nonsmall-cell lung cancer, or pancreatic cancer. Cancer. 2009 Nov 15;115(22):5309-18. doi: 10.1002/cncr.24602. PMID: 19701908; PMCID: PMC2856335.
- Soumoy L, Genbauffe A, Mouchart L, Sperone A, Trelcat A, Mukeba-Harchies L, Wells M, Blankert B, Najem A, Ghanem G, Saussez S, Journe F. ATP1A1 is a promising new target for melanoma treatment and can be inhibited by its physiological ligand bufalin to restore targeted therapy efficacy. Cancer Cell Int. 2024 Jan 4;24(1):8. doi: 10.1186/s12935-023-03196-y. PMID: 38178183; PMCID: PMC10765859.
- Soumoy L, Wells M, Najem A, Krayem M, Ghanem G, Hambye S, Saussez S, Blankert B, Journe F. Toad Venom Antiproliferative Activities on Metastatic Melanoma: Bio-Guided Fractionation and Screening of the Compounds of Two Different Venoms. Biology (Basel). 2020 Aug 10;9(8):218. doi: 10.3390/biology9080218. PMID: 32785105; PMCID: PMC7464305.
- Meng Z, Garrett CR, Shen Y, Liu L, Yang P, Huo Y, Zhao Q, Spelman AR, Ng CS, Chang DZ, Cohen L. Prospective randomised evaluation of traditional Chinese medicine combined with chemotherapy: a randomised phase II study of wild toad extract plus gemcitabine in patients with advanced pancreatic adenocarcinomas. Br J Cancer. 2012 Jul 24;107(3):411-6. doi: 10.1038/bjc.2012.283. Epub 2012 Jul 10. PMID: 22782343; PMCID: PMC3405220.
- Xu J, Li D, Du K, Wang J. Efficacy and Safety of Cinobufacin Combined with Chemotherapy for Advanced Breast Cancer: A Systematic Review and Meta-Analysis. Evid Based Complement Alternat Med. 2020 Sep 19;2020:4953539. doi: 10.1155/2020/4953539. PMID: 33014106; PMCID: PMC7520696.
- Qin TJ, Zhao XH, Yun J, Zhang LX, Ruan ZP, Pan BR. Efficacy and safety of gemcitabine-oxaliplatin combined with huachansu in patients with advanced gallbladder carcinoma. World J Gastroenterol. 2008 Sep 7;14(33):5210-6. doi: 10.3748/wjg.14.5210. PMID: 18777599; PMCID: PMC2744012.
- Halaseh SA, Halaseh S, Shakman R. A Review of the Etiology and Epidemiology of Gallbladder Cancer: What You Need to Know. Cureus. 2022 Aug 22;14(8):e28260. doi: 10.7759/cureus.28260. PMID: 36158346; PMCID: PMC9491243.
- Yan X, Lei L, Li H, Cao M, Yang F, He S, Zhang S, Teng Y, Li Q, Xia C, Chen W. Stomach cancer burden in China: Epidemiology and prevention. Chin J Cancer Res. 2023 Apr 30;35(2):81-91. doi: 10.21147/j.issn.1000-9604.2023.02.01. PMID: 37180831; PMCID: PMC10167608.
- Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024 May-Jun;74(3):229-263. doi: 10.3322/caac.21834. Epub 2024 Apr 4. PMID: 38572751.
- Zhang X, Yuan Y, Xi Y, Xu X, Guo Q, Zheng H, Hua B. Cinobufacini Injection Improves the Efficacy of Chemotherapy on Advanced Stage Gastric Cancer: A Systemic Review and Meta-Analysis. Evid Based Complement Alternat Med. 2018 Sep 4;2018:7362340. doi: 10.1155/2018/7362340. PMID: 30254688; PMCID: PMC6142757.
- Wu J, Zhang D, Ni M, Xue J, Wang K, Duan X, Liu S. Effectiveness of Huachansu injection combined with chemotherapy for treatment of gastric cancer in China: a systematic review and Meta-analysis. J Tradit Chin Med. 2020 Oct;40(5):749-757. doi: 10.19852/j.cnki.jtcm.2020.05.004. PMID: 33000575.
- Sun H, Wang W, Bai M, Liu D. Cinobufotalin as an effective adjuvant therapy for advanced gastric cancer: a meta-analysis of randomized controlled trials. Onco Targets Ther. 2019 Apr 26;12:3139-3160. doi: 10.2147/OTT.S196684. PMID: 31118669; PMCID: PMC6507077.
- Bu ZJ, Wan SR, Steinmann P, Yin ZT, Tan JP, Li WX, Tang ZY, Jiang S, Ye MM, Xu JY, Zheng YY, Wang XH, Liu JP, Liu ZL. Effectiveness and Safety of Chinese Herbal Injections Combined with SOX Chemotherapy Regimens for Advanced Gastric Cancer: a Bayesian Network Meta-Analysis. J Cancer. 2024 Jan 1;15(4):889-907. doi: 10.7150/jca.91301. PMID: 38230219; PMCID: PMC10788720.
- Xu ZY, Jin CJ, Zhou CC, Wang ZQ, Zhou WD, Deng HB, Zhang M, Su W, Cai XY. Treatment of advanced non-small-cell lung cancer with Chinese herbal medicine by stages combined with chemotherapy. J Cancer Res Clin Oncol. 2011 Jul;137(7):1117-22. doi: 10.1007/s00432-011-0975-3. Epub 2011 Feb 5. PMID: 21298288.
- Jie C, Jian Z, Di Y, Jin C. Clinical curative effect on non-small cell lung cancer patients by cinobufacini injection combined first line chemotherapy. Journal of International Oncology. 2016;43(10):741.
- Jiang Y, Liu LS, Shen LP, Han ZF, Jian H, Liu JX, Xu L, Li HG, Tian JH, Mao ZJ. Traditional Chinese Medicine treatment as maintenance therapy in advanced non-small-cell lung cancer: A randomized controlled trial. Complement Ther Med. 2016 Feb;24:55-62. doi: 10.1016/j.ctim.2015.12.006. Epub 2015 Dec 17. PMID: 26860802.
- Jiang Y, Wu LB, Shen LP, Zhang P, Jiang WJ, Tian JH, Liu LS. [Effect of traditional Chinese medicine treatment as maintenance therapy on regulating the serum concentration of sCTLA-4 in patients with advanced non-small-cell lung cancer and its relationship with prognosis]. Zhonghua Zhong Liu Za Zhi. 2016 Oct 23;38(10):757-762. Chinese. doi: 10.3760/cma.j.issn.0253-3766.2016.10.009. PMID: 27784460.
- Xu Y, Han D, Feng FC, Wang ZC, Gu C, Peng WP, He HL, Zhou XM. [Meta-analysis of Cinobufacini Injection combined with platinum-contained first-line chemotherapy in treatment of non-small cell lung cancer]. Zhongguo Zhong Yao Za Zhi. 2019 Nov;44(21):4728-4737. Chinese. doi: 10.19540/j.cnki.cjcmm.20190505.501. PMID: 31872671.
- Peng W, Xu Y, Feng F, Gu C, Wang Z, Han D, Zhou X, He H. Meta-Analysis of Therapy of Cinobufacini Capsule Adjunct with First-Line Platinum-Based Chemotherapy for the Treatment of Advanced NSCLC. Evid Based Complement Alternat Med. 2021 Aug 21;2021:5596415. doi: 10.1155/2021/5596415. PMID: 34471417; PMCID: PMC8405304.
- Tan X, Liang X, Xi J, Guo S, Meng M, Chen X, Li Y. Clinical efficacy and safety of Huachansu injection combination with platinum-based chemotherapy for advanced non-small cell lung cancer: A systematic review and meta-analysis of randomized controlled trials. Medicine (Baltimore). 2021 Sep 10;100(36):e27161. doi: 10.1097/MD.0000000000027161. PMID: 34516509; PMCID: PMC8428704.
- Zhang F, Yin Y, Xu T. Cinobufotalin injection combined with chemotherapy for the treatment of advanced NSCLC in China: A PRISMA-compliant meta-analysis of 29 randomized controlled trials. Medicine (Baltimore). 2019 Aug;98(35):e16969. doi: 10.1097/MD.0000000000016969. PMID: 31464940; PMCID: PMC6736171.
- Li LL, Su YX, Mao Y, Jiang PY, Chu XL, Xue P, Jia BH, Zhu SJ. The Effectiveness and Safety of Cinobufotalin Injection as an Adjunctive Treatment for Lung Cancer: A Meta-Analysis of Randomized Controlled Trials. Evid Based Complement Alternat Med. 2021 Feb 5;2021:8852261. doi: 10.1155/2021/8852261. PMID: 33628320; PMCID: PMC7881937.
- Yang T, Wu C, Li P, Zhong Y, Wu W, Wang S, Yang X. Efficacy and Safety of Huachansu as an Adjuvant Therapy for Non-Small Cell Lung Cancer: An Overview of Systematic Reviews and Meta-Analyses. Integr Cancer Ther. 2024 Jan-Dec;23:15347354241237234. doi: 10.1177/15347354241237234. PMID: 38469799; PMCID: PMC10935755.
- Raoul JL, Forner A, Bolondi L, Cheung TT, Kloeckner R, de Baere T. Updated use of TACE for hepatocellular carcinoma treatment: How and when to use it based on clinical evidence. Cancer Treat Rev. 2019 Jan;72:28-36. doi: 10.1016/j.ctrv.2018.11.002. Epub 2018 Nov 12. PMID: 30447470.
- Ling CQ, Fan J, Lin HS, Shen F, Xu ZY, Lin LZ, Qin SK, Zhou WP, Zhai XF, Li B, Zhou QH; Chinese Integrative Therapy of Primary Liver Cancer Working Group. Clinical practice guidelines for the treatment of primary liver cancer with integrative traditional Chinese and Western medicine. J Integr Med. 2018 Jul;16(4):236-248. doi: 10.1016/j.joim.2018.05.002. Epub 2018 May 17. PMID: 29891180.
- Wu T, Sun R, Wang Z, Yang W, Shen S, Zhao Z. A meta-analysis of Cinobufacini combined with transcatheterarterial chemoembolization in the treatment of advanced hepatocellular carcinoma. J Cancer Res Ther. 2014 Aug;10 Suppl 1:60-4. doi: 10.4103/0973-1482.139763. PMID: 25207894.
- Gao H, He J, Cheng CS, Zhuang L, Chen H, Meng Z. Unresectable hepatocellular carcinoma: transarterial chemoembolisation plus Huachansu - a single-center randomised controlled trial. BMJ Support Palliat Care. 2023 Jul 3:spcare-2022-003870. doi: 10.1136/spcare-2022-003870. Epub ahead of print. PMID: 37400162.
- Dong J, Zhai X, Chen Z, Liu Q, Ye H, Chen W, Ling C. Treatment of Huge Hepatocellular Carcinoma Using Cinobufacini Injection in Transarterial Chemoembolization: A Retrospective Study. Evid Based Complement Alternat Med. 2016;2016:2754542. doi: 10.1155/2016/2754542. Epub 2016 May 17. PMID: 27293455; PMCID: PMC4886065.
- Zhai XF, Chen Z, Li B, Shen F, Fan J, Zhou WP, Yang YK, Xu J, Qin X, Li LQ, Ling CQ. Traditional herbal medicine in preventing recurrence after resection of small hepatocellular carcinoma: a multicenter randomized controlled trial. J Integr Med. 2013 Mar;11(2):90-100. doi: 10.3736/jintegrmed2013021. PMID: 23506690.
- Chen Z, Chen HY, Lang QB, Li B, Zhai XF, Guo YY, Yue XQ, Ling CQ. Preventive effects of jiedu granules combined with cinobufacini injection versus transcatheter arterial chemoembolization in post-surgical patients with hepatocellular carcinoma: a case-control trial. Chin J Integr Med. 2012 May;18(5):339-44. doi: 10.1007/s11655-012-1083-1. Epub 2012 May 2. PMID: 22549390.
- Xue T, Yu H, Feng W, Wang Y, Wu S, Wang L, Zhu P, Guan J, Sun Q. Efficacy and Safety of Hepatic Arterial Infusion Therapy with Cinobufacini in Advanced Hepatocellular Carcinoma with Macrovascular Invasion: A Retrospective Cohort Study. Cancer Manag Res. 2024 Mar 14;16:163-175. doi: 10.2147/CMAR.S440017. PMID: 38505102; PMCID: PMC10948332.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.