Medicine Group. 2024 August 30;5(8):1012-1024. doi: 10.37871/jbres1983.
Preclinical Assessment of MicroMatrix® + Cytal® and Integra® DRT in a Porcine Third-Degree Burn Model
Adam M Jorgensen#, Ronald A Nelson#, Cara Clouse, Christie L Scott, Uma Gandhi, Anthony Atala and Sean V Murphy*
- Urinary bladder matrix
- Wound healing
- Porcine burn model
- Integra® DRT
- Epithelialization
- Dermal regeneration
Abstract
Third-degree burn wounds can cause devastating loss of functionality for patients. As such, platform technologies that allow effective wound management are needed. Herein, we describe the use of MatriStem™ Urinary Bladder Matrix (UBM)-based products (MicroMatrix® + Cytal®) and a collagen and glycosaminoglycan (GAG) product (Integra® Dermal Regeneration Template (DRT)) in a porcine third-degree burn wound model. Full-thickness third-degree burns were created on dorsal porcine skin, debrided using an excisional model, then treated with MicroMatrix® + Cytal® or Integra® DRT; treatment groups were compared against the untreated wound controls. MicroMatrix® + Cytal®-treated wounds appeared to have faster wound closure and epithelialization compared to untreated and Integra® DRT-treated wounds. Multi-factor wound healing analysis demonstrated that MicroMatrix® + Cytal® performed better than untreated and Integra® DRT-treated wounds based on the metrics of contraction, epithelialization, and wound closure rate. Histological analysis demonstrated that all treated wounds had complete epidermal closure at various levels of maturation and presented mature, thick, and organized collagen bundles. Integra® DRT -treated wounds appeared to have more similar histological resemblance to native tissue compared to MicroMatrix® + Cytal® and untreated control. Importantly, treatment with either MicroMatrix® + Cytal® products or Integra® DRT, resulted in an extracellular matrix (ECM) composition and cellular organization similar to healthy skin. MicroMatrix®+ Cytal®-treatment promoted rapid wound closure and neo-epithelialization, while Integra® DRT-treatment seemed superior in promoting a healthy and pro-regenerative dermal wound bed.
Introduction
Thermal burns are devastating to patients despite the current standard of care treatments. The current standard of care treatment utilizes an autologous split-thickness skin graft. These procedures require removing skin from a donor site on the patient, increasing the skin surface area through meshing, and applying the graft to the burned site [1-4]. While reasonable clinical outcomes are common with these treatments, extensive wounds or burns may limit the availability of patient graft samples [5]. Allografts have been suggested as an alternative graft source, however the requirement of immunosuppressive drugs to avert immune rejection limits this approach as a viable long-term treatment option for many patients [6,7].
Autograft and allograft limitations have led to the development of non-cellular dermal substitutes, including synthetic or natural scaffolds, such as silicone and collagen (e.g., Integra® DRT and Biobrane). Dermal substitutes improve wound healing outcomes compared with controls, but often require a secondary split-thickness skin graft procedure [8,9]. More complex biological skin equivalents are being developed by tissue engineers to produce additional wound treatment options for patients (e.g., Apligraft, Dermagraft, Stratagraft, and TransCyte). Tissue-engineered skin is often comprised of a scaffold seeded with human skin cells, primarily fibroblasts and keratinocytes. Multiple products have made it to market and have shown the ability to improve wound closure and healing compared to standard of care [10]. Others have attempted to engineer even more complex skin by bioprinting skin with multiple cell types to recapitulate further the structure and function of healthy human skin [11,12]. However, the use of some of these therapies may be limited by high production expenses, complex storage requirements, challenging application methodologies, and immune rejection risks. Thus, there is a significant need for a pro-regenerative wound healing product with 1) proven clinical efficacy, 2) non-living, room temperature stable components, and 3) retained bioactivity.
One of the products used to treat third-degree burns is Integra® DRT, an acellular bi-layer matrix made of an outer thin silicon layer and an inner complex cross-linked collagen and glycosaminoglycan. Integra® DRT promotes rapid vascularization and provides impressive coverage of wounds [13]. Notably, Integra use requires a two-procedure process requiring a secondary split-thickness skin graft to complete coverage [14]. The secondary procedures may limit product use due to the morbidity and scaring associated with skin donor sites [15].
However, given the disadvantages inherent to synthetic materials regarding wound remodeling and regeneration, decellularized grafts have been developed as biological alternatives. Decellularization of tissues and organs have yielded acellular matrices that promote site-appropriate remodeling of tissues and organs [16,17]. Extracellular Matrix (ECM) scaffolds have demonstrated the ability to direct site-specific remodeling of injured tissues in multiple preclinical and clinical applications [18]. Urinary Bladder Matrix (UBM) is an ECM derived from the inner layers of a porcine urinary bladder. Due to the minimal processing, UBM maintains its natural collagen structure, with an intact epithelial basement membrane that facilitates various tissue remodeling processes [19]. UBM has been extensively investigated in preclinical and clinical settings [20]. The results of those studies indicated the potential of UBM to promote neo-vascularization and neo-innervation, enhance cell adhesion and infiltration, modulate the immune response, and promote advantageous tissue remodeling responses [21]. In previous preclinical studies, UBM has been shown to simultaneously accelerate wound closure while promoting re-epithelialization in an ovine model [22]. In a randomized interim clinical trial of diabetic foot ulcer treatment, UBM accelerated wound closure with a lower recurrence rate [23]. Preliminary clinical studies using UBM to treat burns further confirmed that subsequent autografting was unnecessary due to expedited re-epithelialization with no subsequent contraction or postoperative complications [24]. These responses make UBM an attractive option as a scaffold for wound healing. While the specific mechanisms driving these results are still under investigation, the host immune response to UBM tends to include a robust immunomodulatory M2 macrophage phenotype, as opposed to the typical pro-inflammatory M1 phenotype [25].
Currently, UBM is not indicated for treatment of third-degree burns, unlike Integra® DRT, hence further assessment of UBM based products is required to determine their efficacy in burn injuries. In this study, we describe the use of a particulate UBM (MicroMatrix®) used concomitantly with a six-layer UBM device (Cytal®) to treat porcine burn wounds. This study’s primary objective was to characterize wound closure, wound contraction, and re-epithelialization of MicroMatrix® + Cytal®-treated, Integra® DRT-treated, and untreated full-thickness debrided skin burns. We also assessed the histological properties of the remodeled wounds to determine the effect of these products on epidermal maturation and extracellular matrix composition. These analyses demonstrate the efficacy of MicroMatrix® + Cytal® and Integra® DRT devices in full thickness debrided third-degree burns.
Results
Wound treatment and product application
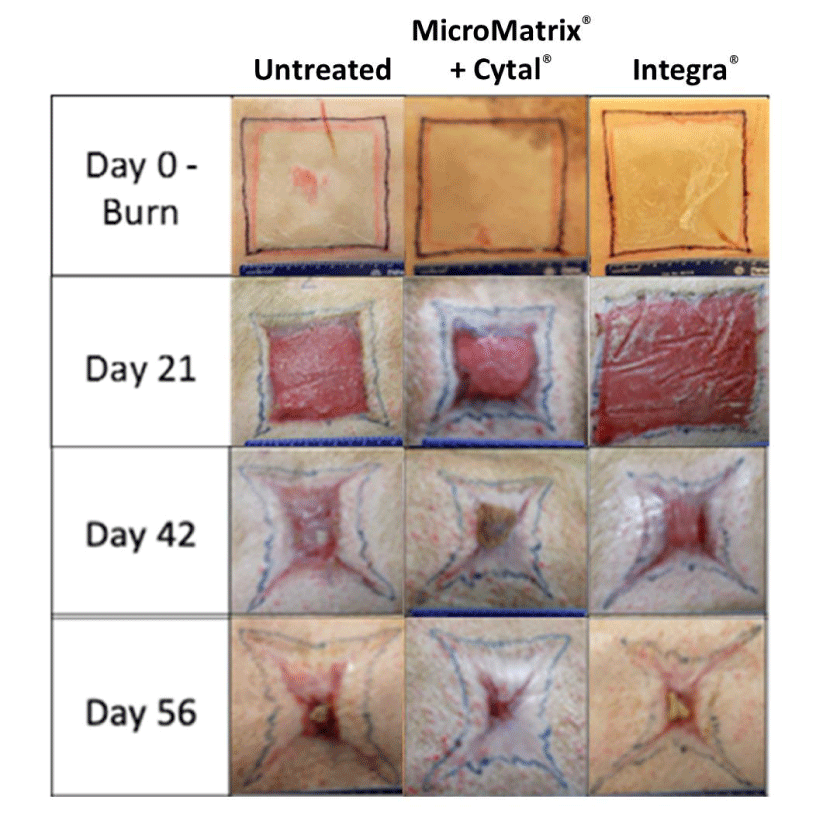
On Day 0, uniform 8 x 8 cm burn wounds were created. The next day wounds were debrided per standard protocol create a uniform depth (Figure 1). Representative images of wounds and treatments for Day 0, 21, 42, 56 time-points are shown in figure 2. The MicroMatrix® particulate (1000mg) was placed into a sterile bowl and mixed with 5mL sterile saline to form a thick paste. An even, thin coverage of the paste was applied to the wound bed. Following the application of MicroMatrix® paste, the Cytal® sheet was placed on the wound bed and sutured for additional security. Cytal® was simple to place and suture onto the wound, resulting in consistent and complete coverage of the wound area. Integra® DRT devices required minimal cutting and shaping to ensure exact fit into the wound as the product requires direct contact between the Integra® DRT device and the wound edge in a "butt joint" manner. Overall, all products were easy to hydrate and administer by a single surgeon with minimal preparation steps.
Wound closure
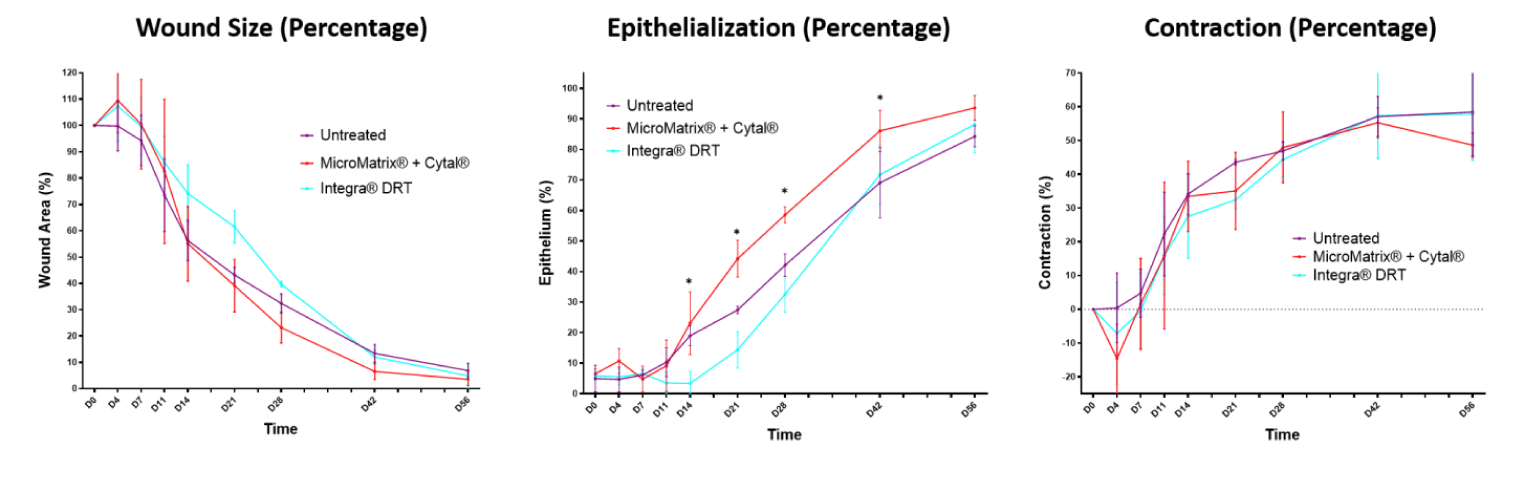
At each time point, the percentage of the open wound area relative to the original wound area was measured using digital photography to describe wound closure. Figure 3A and figure 4 show the combined and separated versions of these measurements, respectively. MicroMatrix® + Cytal® showed the greatest reduction in wound size over the time-course (as a percentage of wound area and total area) with maximal differences between days 14-28. Specifically, all MicroMatrix® + Cytal®-treated wounds had a significantly smaller wound area percentage than Integra® DRT-treated wounds at day 21 (39.1% ± 10.0 vs. 61.5% ± 6.1; p < 0.01). After removing the silicone backing on the Integra® DRT product by day 21, significant acceleration in wound closure was observed. By day 56, no significant difference between MicroMatrix® + Cytal® and Integra® DRT was observed (3.6% ± 2.4 vs. 4.9% ± 4.6).
Wound epithelialization
Epithelialization, visually presented as matte pink/white with an epithelial coating distinct from the wound area, was measured using digital photography and gross observations at all time-points. The total area of epithelialization was determined by subtracting the open wound area from the total area dictated by the preestablished tattoo border. Figure 3B and figure 4 show the combined and separated versions of these measurements, respectively. MicroMatrix® + Cytal®-treated wounds showed rapid re-epithelialization between days 11-42. MicroMatrix® + Cytal® wounds had significantly greater epithelialization compared with Integra® DRT-treated wounds at day 21 (44.2% ± 6.02 vs. 14.4% ± 5.9; p < 0.01). By day 28, MicroMatrix® + Cytal®-treated wounds also had significantly greater epithelialization than untreated wounds (58.6% ± 2.7 vs. 42.1% ± 3.7; p < 0.01). Improvements in epithelialization in MicroMatrix® + Cytal®-treated wounds compared with both Integra® DRT-treated and untreated wounds were present through day 42 (MicroMatrix® + Cytal®, 86.1% ± 6.7; Integra® DRT, 71.8% ± 9.6; Untreated, 69.1% ± 11.5; p < 0.05). By day 56, all groups had similar epithelialization (MicroMatrix® + Cytal®, 93.6% ± 4.1; Integra® DRT, 88.2% ± 9.4; Untreated, 84.3% ± 3.4).
Wound contraction
The area within the tattoo border at each time point, fractioned against the original tattoo, was measured, using digital photography, and expressed as a percentage to calculate percent wound contraction. Figure 3C and figure 4 show the combined and separated versions of these measurements, respectively. All groups showed similar initial contraction rates, with rapid contraction until day 21, at which stage wound stabilization had occurred for all wounds. Overall, no significant differences in wound contraction occurred between experimental groups despite untreated and Integra® DRT-treated wound groups having a higher percent contraction average by the completion of the study (MicroMatrix® + Cytal®, 48.7% ± 3.6; Integra® DRT, 57.9% ± 13.8; Untreated, 58.5% ± 13.0).
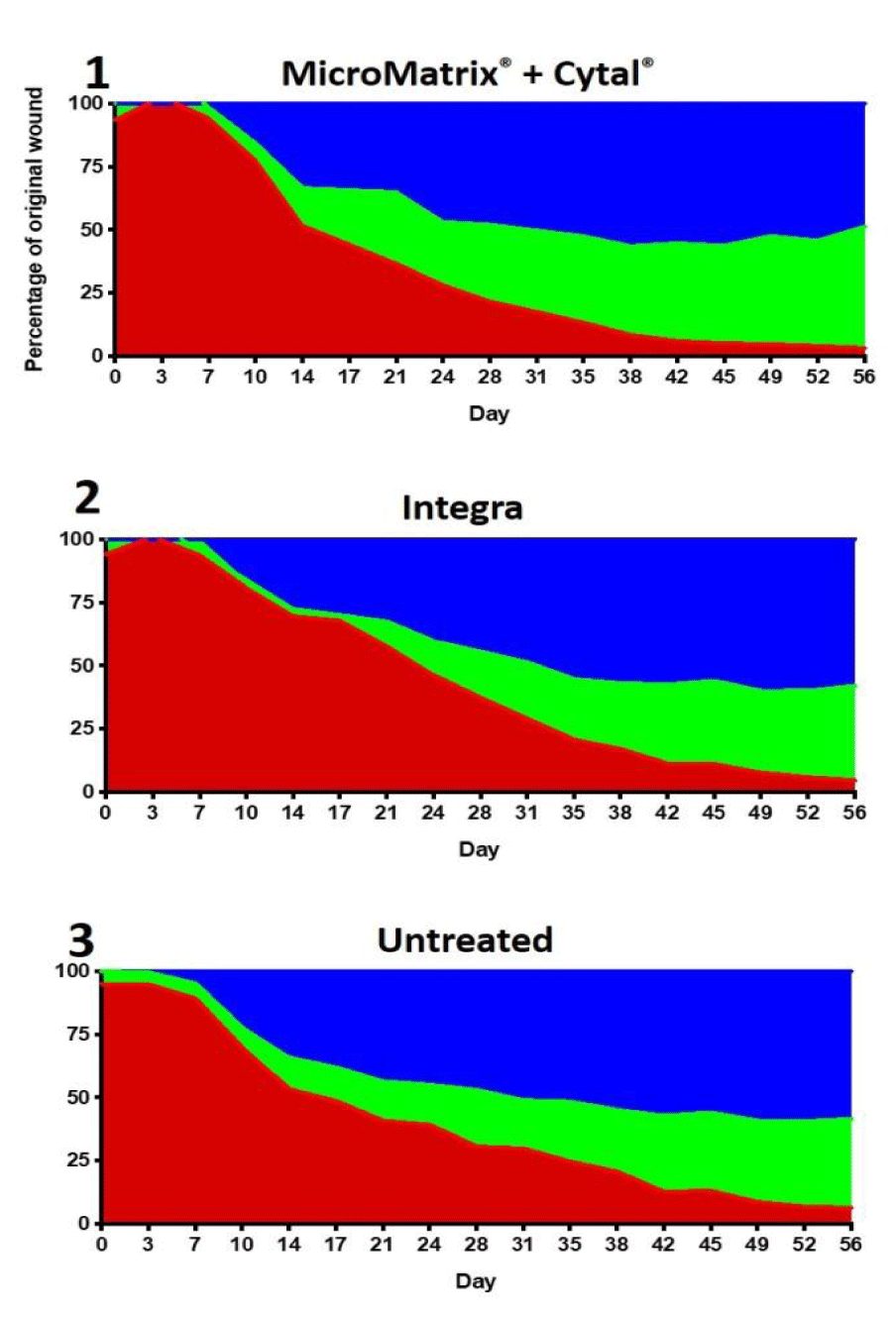
Combined analysis of wound closure, epithelializaiton, and contraction
While individual wound healing parameters such as wound closure, contraction, and epithelialization are necessary to fully assess wound management efficacy, combinatorial analysis is able to communicate a comprehensive view of the overall wound healing quality. To represent the total wound area at each time-point, we divided the wound into three categories: percentage of open wound area (red), percentage of contraction (green), and percentage of epithelialization (blue) (Figure 4). Using these analyses to describe the healing wound over time, the importance of the type and quality of wound healing was highlighted. The best-performing treatments were defined as having the following characteristics: 1) small wound area, 2) low wound contraction, and 3) high epithelialization throughout the study. Overall, MicroMatrix® + Cytal® showed superior wound closure and epithelialization during most of the experimental time-course, with Integra® DRT matching performance in these parameters at later time-points. All groups demonstrated similar wound contraction over the course of the study.
Histological analysis
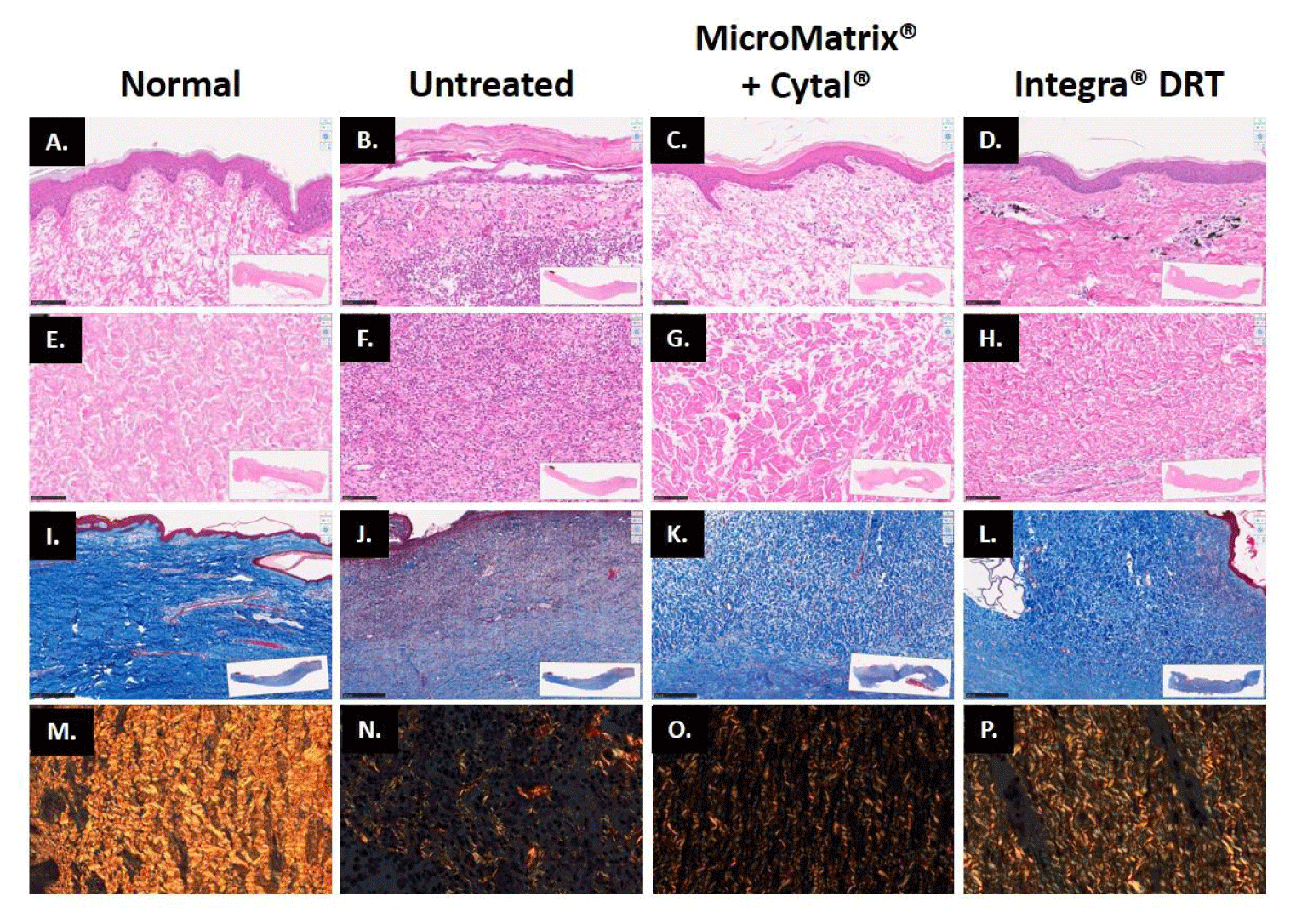
Slides were stained with hematoxylin and eosin (H&E) and used to compare the composition and general structure of each of the wounds; normal healthy pig skin was used as a control (Figure 5 A-H). The epidermis of healthy skin stained with H&E demonstrated stratification and rete ridges (Figure 5A). Analysis of the wound histology revealed that most wounds had some degree of epidermal covering (Figure 5A-D). All treated wounds showed a complete epidermis at various levels of maturation (Figure 5A-D). Specifically, the epidermis in wounds treated with MicroMatrix® + Cytal® appeared most similar to healthy skin, with epidermal coverage, thickness, and presence of rete ridges, confirming a mature epidermis [26] (Figure 5C). Wounds treated with Integra® DRT also displayed a epidermal coverage and thickness similr to healthy skin, although less rete ridge formation was observed (Figure 5D). Untreated wounds appeared to have inconsistent epidermis coverage, which was thinner where present and lacking noticeable epithelial rete peg protrusions (Figure 5B). The dermis of healthy skin stained with H&E consisted of organized light pink fibers with only minimal disorganized and thin purple fibers (Figure 5E-H). Integra® DRT-treated wounds showed the most similar dermis structure and organization to normal healthy skin with mature, thick and organized collagen bundles (Figure 5H). MicroMatrix® + Cytal®-treated wounds also showed appropriate dermis structure and organization, although less dense than healthy skin or Integra® DRT-treated wounds (Figure 5G). Finally, the untreated wounds showed a dense, highly cellularized dermis with signs of ongoing inflammatory infiltration (Figure 5F).
Masson’s Trichrome staining provides an overview of keratin and collagen composition for each wound, specifically, this staining aids in the identification of multiple ECM components simultaneously. Figure 5 (I-L) shows the representative images of the treatment groups and normal healthy skin. Normal healthy pig skin had a dermis consisting of intense collagen staining (blue) and keratin staining (red), specifically in the epidermis (Figure 5I). Additionally, high cellularity was visible in the epidermis and dermis (brown nuclei). MicroMatrix® + Cytal® and Integra® DRT-treated wounds had extracellular matrix composition, organization, and staining intensities similar to normal healthy pig skin (Figure 5K-L). Alternatively, untreated wounds showed a denser, red-stained dermis (Figure 5J). Keratin staining was as expected for all treatments, confirming epidermis evaluation by H&E.
Picrosirius red staining distinguishes between mature and immature collagen when imaged under polarized light. Figure 5 (M-P) shows the representative images of the treatment groups and normal healthy skin. Immature unorganized collagen stains green (Collagen Type III) and mature bundled and organized collagen fibers stain yellow/orange (collagen Type I). Healthy skin demonstrates intense staining of orange and green (ratio ~9:1), suggesting an organized network of large mature collagen fibers intermixed with smaller immature collagen (Figure 5M). Integra® DRT-treated wounds had collagen content and organization most similar to healthy skin (ratio ~7:1) (Figure 5P). Although MicroMatrix® + Cytal® had a similar collagen composition (ratio ~7:1), there were smaller collagen bundles (Figure 5O). Untreated wounds lacked any significant organized collagen structures (Figure 5N).
Discussion
This study utilized a well-developed and clinically relevant model for burn wound treatment. While it is difficult to fully recapitulate the complex and specific healing environment of burns, several models have been described in the literature [27]. In this study, we applied an 8 x 8 cm square metal brand to the center of the 10 x 10 cm tattooed area. The burn was left overnight untreated other than standard of care, before surgical debridement to the edge of the tattooed area was performed. This method recapitulates the normal surgical progression of clinical treatment in humans and allowed for the wound to take on the pathophysiology of a burn [28]. It has been shown that, although the rate of wound closure in pigs is slower than in humans, closure and contraction rates using porcine burn models are consistent throughout experiments, highlighting the clinical relevancy of this model [29].
MicroMatrix® + Cytal® treatment accelerated wound closure and improved epithelialization. Accelerated healing is attributed to the fast resorption profile of the MicroMatrix® device, which is thought to jump start the formation of granulation tissue and prepare the wound bed for vascularization and healing. This was particularly evident at day 21, wherein wound closure percentage and epithelialization was significantly improved in the MicroMatrix® + Cytal®-treatment group compared with other groups. It is important to note that this improvement was seen without an increase in contraction, suggesting wound healing driven by epithelialization. In Integra® DRT -treated wounds, the silicone bandage was removed 21 days after implantation per manufacturer’s instruction. Post the 21-day time-point, Integra® DRT-treated wounds healed rapidly with improved epithelialization and minimal contraction. As described in the results, the silicone membrane in Integra® DRT treated wounds had less epithelialization in the early time points. While in a clinical setting, this would resolved by the addition of a split-thickness autograft, it also does suggest that in our model the earlier removal of the silicone backing may have allowed for improved wound healing outcomes. By day 42, the MicroMatrix® + Cytal®-treatment group demonstrated a significant improvement in epithelialization compared to Integra® DRT and untreated wounds. These morphometric findings suggest that MicroMatrix® + Cytal®-treated wounds benefit from epidermal healing, both in the short and long term, without requiring a split-thickness autograft. Thus MicroMatrix® + Cytal® could be a candidate for use in patients where split-thickness autografting is not feasible; however, further clinical investigation is warranted.
Histological staining of the wound tissues further demonstrated the improved epithelialization seen in the MicroMatrix® + Cytal®-treated wounds. MicroMatrix® + Cytal®-treated wounds formed a thick epidermis with rete ridges. The epidermis formed in the Integra® DRT-treated wounds was thick and complete, but without observable rete ridges. Untreated wounds had a sparse epidermal covering, a thin epidermal layer where present, low cellularity, weak keratin staining, and a lack of rete ridges. These findings demonstrate that the in untreated burn wounds, the epidermis was less mature and likely developing at a slower rate than the MicroMatrix® + Cytal® or Integra® DRT-treated wounds. Dermis quality is also critical to the long-term success of healing wounds [30]. A healthy dermal ECM composition and organization facilitates normal mechanical and cellular properties, while an abnormal ECM composition and organization often indicates scarring and contraction; this is typically associated with diminished mechanical properties [31-33]. Histological staining demonstrated that the dermal ECM composition of Integra® DRT-treated wounds consisted of thick mature collagen fibers, consistent with healthy skin [34]. Picrosirius red staining demonstrated that these wounds contained a near-normal ratio of green and red-stained collagen fibers arranged in a basket-like weave pattern, resembling normal healthy skin. In contrast, MicroMatrix® + Cytal® treated wounds showed finer fibers in a lower orange to green ratio, which is a characteristic of immature healing [35,36]. This might be explained by MicroMatrix®+ Cytal® being more likely to be integrated and remodeled at early time-points due to its base material properties (powder and meshed patch, respectively) before inevitably being replaced by the native porcine ECM. The superior performance of the Integra® DRT regarding the observed dermis composition in this study may be due to an optimal ECM composition, mechanical properties, form factor, degradation rates as well as many other known and unknown factors. In this study we were unable to distinguish between remaining treatment-product ECM and native remodeled ECM. However, evaluation of ECM source, composition, density, and form factor (total matrix weight per wound, patch thickness and porosity, etc.) and how this impacts various wound healing characteristics could inform additional clinical insight in future studies.
It is also important to address the limitations of this study and communicate viable options to pursue in the future. First, this study utilized a single time-point (day 56) for wound histological analysis. This time-point is important for evaluating the resolution of the open wounds, however future studies will include further evaluation of the acute inflammatory response and early wound healing phases during the first days/weeks of treatment as well as evaluate long-term remodeling several months post-wound closure. These additional time-points utilizing "sacrificial" wounds for repeated biopsies and analysis would provide a better understanding of the overall healing process. Additionally, wound care management could be extended to capture longer time-points to evaluate scarring mechanisms and other complex wound healing processes. Finally, we are cautious to avoid over-interpretation of our data comparing a single application of Integra® DRT without application of a split-thickness graft, and two spaced applications of MicroMatrix®+ Cytal®. Integra® DRT is designed as a single application skin substitute followed up application of a split-thickness autograft after removal of the protective silicone layer. The lack of split-thickness autograft in our study, explains the differences seen in epidermal maturation, but provides supporting evidence that Integra® DRT is effective in preparing a healthy and pro-regenerative dermal wound bed. Alternatively, the manufacturer instructions for MicroMatrix® and Cytal® state that the wound should be inspected every 7 days, and new material can be applied to open wound areas as needed. Future studies could evaluate optimal timing for product re-application, timing and success rates for split-thickness autografting as well as potentially synergistic benefits of a combination device of Integra® DRT, MicroMatrix® and Cytal®. A combination product has the potential to allow for both improved dermal remodeling present in Integra® DRT treated wounds as well as mature epidermal remodeling seen in MicroMatrix® and Cytal® treated wounds without the need for autografts, making it an ideal candidate for full-thickness wounds.
Materials and Methods
Materials
Integra® DRT was purchased from Integra Lifesciences Corporation, PrSinceton, NJ. MicroMatrix® and Cytal® UBM products (MicroMatrix® and Cytal® 3-Layer) were generously provided by ACell Inc., Columbia, MD.
Animal study and regulations
This study was reviewed and approved by the Wake Forest University Institutional Animal Care and Use Committee (A18-012). Specific Pathogen Free (SPF) Yorkshire pigs were acclimatized for two weeks. A protective plastic saddle was placed on the animal for gradually increasing periods until the animal was comfortable wearing the saddle indefinitely. By the start of the study, the pigs fully tolerated the protective plastic saddle and weighed approximately 40-50kg (N = 3).
Full thickness burn
The animals were sedated and anesthetized with a combination of ketamine, xylazine, and acepromazine, and then maintained under anesthesia with inhaled isoflurane via an endotracheal tube with continuous monitoring. While immobilized in the dorsal position, the back and sides of the pigs were shaved and dried. Two 10 x 10 cm tattoos were stenciled on both sides of each pig (a total of four 10 x 10 cm tattoos per pig) to denote the area of the excisional wound (Figure 1A). The dorsal skin was sanitized with soap and water then sterilized with 70% alcohol and β-iodine. An 8 x 8 cm square metal brand heated to 90℃ was applied onto the center of the 10 x 10 tattooed area for 60 seconds to create the burn defect (Figure 1B). The wounds were covered with CoolMagic™ Hydrogel Sheets (MPM Medical, Inc.), bandaged, and covered with the protective saddle and jacket overnight (Figure 1C).
Full thickness debridement
Twenty-four hours after the initial burn wounds, the animals were again sedated and anesthetized with a combination of ketamine, xylazine, and acepromazine. They were maintained under anesthesia using inhaled isoflurane via an endotracheal tube with continuous monitoring. After removing the bandaging, the animals were immobilized and placed in a dorsal position. The burn defects were debrided to the depth of the bleeding edge and to the width of the 10 x 10 cm tattooed area using a scalpel blade, and digital photographs were taken (Figure 1D).
Wound treatments
Immediately after debridement, all wounds were treated according to the approved experimental groups. The three experimental groups were: 1) MicroMatrix® + Cytal® 3-Layer Wound Matrix, 2) Integra® DRT, and 3) untreated (Figure 1E). All treatments received standard of care bandaging on top of treatment application. First a topical antibiotic was applied to the wound area followed by (size) Tegaderm. The pigs were then held between two tables, and the wound areas were covered with a wrap of cast padding and a cohesive vet wrap bandaging. A custom cut stockinet was then place over the pig, with holes cut for the forelegs, and a custom protective plastic saddle was secured over the wound area using Velcro straps. A pig jacket was then placed over the pig to hold the saddle and underlying bandages in place. The three experimental treatments were distributed over the four skin defects to control for differences in wound locations, resulting in a total of at least three applications of each product per wound location. As per the treatment guidelines, MicroMatrix® + Cytal® was re-applied per manufacturer’s recommendations on day 14 and day 28. For Integra, the silicone backing of the product was removed from the integrated Integra® as per the product and clinical treatment protocol. During each 5 experimental rounds lasting 56 days, the wounds were inspected and imaged bi-weekly to 1) measure the wound size, re-epithelialization, and closure, 2) clean and administer antibiotics, and 3) apply fresh bandaging the wound bed.
Bandage changes
After the application of treatments and for each scheduled bandage change on days 4, 7, 11, 14, 18, 21, 28, 31, 35, 38, 42, 45, 49 and 52, each wound area was covered with standard wound dressing materials to provide protection. The materials included a topical antibiotic, Tegaderm, cast padding, cohesive bandaging, a stockinet, and a protective plastic saddle. First a topical antibiotic was applied to the wound area followed by (size) Tegaderm. The pigs were then held between two tables, and the wound areas were covered with a wrap of cast padding and a cohesive vet wrap bandaging. A custom cut stockinet was then place over the pig, with holes cut for the forelegs, and a custom protective plastic saddle was secured over the wound area. Prior to applying the protective plastic saddle, the edges were covered with porous surgical tape to protect against chaffing of the pig's neck. Two porous straps were positioned on the shoulders of the pig while an additional strap was positioned on the underside. These straps were secured using six Velcro strips applied directly to the protective plastic saddle. Lastly, a custom-designed jacket was donned to further secure the saddle and bandaging materials on the pig (Figure 1F).
Digital planimetry
Digital photographs were taken at each bandage change (days 0, 3, 7, 10, 14, 17, 21, 24, 28, 31, 35, 38, 42, 45, 49, 52, 56). Standardized digital planimetry was performed and quantified, as previously described [37]. Wound closure and epithelialization were measured using Image to determine the area of open wound and epithelialization based the color and texture of the healing wound (Supplementary Figure 1); open wounds were primarily shiny and dark red and shiny, while the epithelium was generally opaque, and ranged from pink to light red. Wound contraction was also measured using Image by measuring the area inside the healing wound’s tattoo square and comparing it against the original tattoo size.
Euthanasia
The study terminated after 56 days. The animals were sedated with ketamine, xylazine, and acepromazine. The wounds were harvested and divided for histology and immunohistochemistry analysis. Once completed, a lethal overdose of pentobarbital euthanasia solution was administered.
Histology and imaging
Wound sections were fixed for approximately 48 hours in 4% paraformaldehyde and then transferred to 70% ethanol before paraffin processing. A microtome (Leica) was used to generate 5 µm sectioned slides comprising the center and edge regions of the healed wounds. The slides were stained with hematoxylin and eosin, Masson's trichrome, and picrosirius red. A slide scanning system was used to capture the entire slide at 20x magnification to evaluate various regions of interest. NDP viewer (Hamamatsu Photonics K.K.) was used for assessing bright-field images for capturing hematoxylin and eosin and Masson's trichrome stained samples. OlyVIA software (Olympus Life Science) was used to evaluate the picrosirius red-stained sections under a polarized filter. Each slide was examined for epidermal formation and epithelialization, dermal organization, extracellular matrix composition, and organization. All experimental groups were compared to other treatment groups and controls, including normal undamaged healthy skin.
Statistical analysis
The data for each experimental group was presented as the mean difference. Statistical significance was analyzed using GraphPad Prism software (Graphpad Software Inc.). Mixed models analysis of variance was used to compare outcomes, with models including TREATMENT, DAY, and a TREATMENT x DAY interaction. Furthermore, within-animal variability was controlled by a compound symmetry structure. A confidence interval of at least 95% assumes significance.
Conclusion
Both MicroMatrix®+ Cytal® and Integra® DRT products were easy to apply to full thickness debrided third-degree burns and resulted in effective wound closure. Integra® DRT-treatment seemed superior in promoting a healthy and pro-regenerative dermal wound bed, while MicroMatrix®+ Cytal®-treatment promoted rapid wound closure and neo-epithelialization. Future studies will evaluate additional time-points for mechanistic and optimization studies, as well as explore potential synergistic benefits of each product by forming a combination device to facilitate faster wound closure.
Supplementary Materials
The following are available online at www.mdpi.com/xxx/s1, Figure S1: Digital Planimetry Method.
Author Contributions
For research articles with several authors, a short paragraph specifying their individual contributions must be provided. The following statements should be used Conceptualization, RAN, AA, and SVM; methodology, AMJ, RAN, CC, SLC UG, AA, and SVM; software, AMJ, AA, and SVM; validation, CC and SVM; formal analysis, AMJ, RAN, and SVM.; investigation, AMJ, RAN, CC, and SVM; resources, AA and SVM.; data curation, AMJ and SVM.; writing—original draft preparation, AMJ; writing—review and editing, AMJ, RAN, and SVM; visualization, AMJ, RAN, and SVM; supervision, AA and SVM; project administration, SVM; funding acquisition, AA and SVM. All authors have read and agreed to the published version of the manuscript.
Funding
This research was financially supported by ACell, Inc., which commercializes urinary bladder matrix as MatriStem UBM, MicroMatrix®, and Cytal® (S.V.M.) and NIH/NIAMS 1 F30 AR074866-01A1 (A.M.J).
Disclosure
This manuscript is a discussion of off-label use of devices that have not been cleared by the FDA for these indications.
Conflicts of Interest
A. Atala serves on the Scientific Advisory Board for ACell, Inc, and is a shareholder in the company.
References
- Blair VP, Brown JB. The use and uses of large split skin grafts of intermediate thickness. Plastic and Reconstructive Surgery. 1968;42(1):65-75.
- McDonald WS, Deitch EA. Hypertrophic skin grafts in burned patients: a prospective analysis of variables. J Trauma. 1987 Feb;27(2):147-50. doi: 10.1097/00005373-198702000-00008. PMID: 3546710.
- TANNER JC Jr, VANDEPUT J, OLLEY JF. THE MESH SKIN GRAFT. Plast Reconstr Surg. 1964 Sep;34:287-92. PMID: 14209177.
- Ratner D. Skin grafting. From here to there. Dermatol Clin. 1998 Jan;16(1):75-90. doi: 10.1016/s0733-8635(05)70488-5. PMID: 9460579.
- Alexander JW, MacMillan BG, Law E, Kittur DS. Treatment of severe burns with widely meshed skin autograft and meshed skin allograft overlay. J Trauma. 1981 Jun;21(6):433-8. PMID: 7230295.
- Vecin NM, Kirsner RS. Skin substitutes as treatment for chronic wounds: current and future directions. Front Med (Lausanne). 2023 Aug 29;10:1154567. doi: 10.3389/fmed.2023.1154567. PMID: 37711741; PMCID: PMC10498286.
- Safi AF, Kauke M, Nelms L, Palmer WJ, Tchiloemba B, Kollar B, Haug V, Pomahač B. Local immunosuppression in vascularized composite allotransplantation (VCA): A systematic review. J Plast Reconstr Aesthet Surg. 2021 Feb;74(2):327-335. doi: 10.1016/j.bjps.2020.10.003. Epub 2020 Oct 21. PMID: 33229219.
- Lesher AP, Curry RH, Evans J, Smith VA, Fitzgerald MT, Cina RA, Streck CJ, Hebra AV. Effectiveness of Biobrane for treatment of partial-thickness burns in children. J Pediatr Surg. 2011 Sep;46(9):1759-63. doi: 10.1016/j.jpedsurg.2011.03.070. PMID: 21929986; PMCID: PMC4257603.
- Rahmanian-Schwarz A, Beiderwieden A, Willkomm LM, Amr A, Schaller HE, Lotter O. A clinical evaluation of Biobrane(®) and Suprathel(®) in acute burns and reconstructive surgery. Burns. 2011 Dec;37(8):1343-8. doi: 10.1016/j.burns.2011.07.010. Epub 2011 Aug 17. PMID: 21852044.
- Przekora A. A Concise Review on Tissue Engineered Artificial Skin Grafts for Chronic Wound Treatment: Can We Reconstruct Functional Skin Tissue In Vitro? Cells. 2020 Jul 6;9(7):1622. doi: 10.3390/cells9071622. PMID: 32640572; PMCID: PMC7407512.
- Albanna M, Binder KW, Murphy SV, Kim J, Qasem SA, Zhao W, Tan J, El-Amin IB, Dice DD, Marco J, Green J, Xu T, Skardal A, Holmes JH, Jackson JD, Atala A, Yoo JJ. In Situ Bioprinting of Autologous Skin Cells Accelerates Wound Healing of Extensive Excisional Full-Thickness Wounds. Sci Rep. 2019 Feb 12;9(1):1856. doi: 10.1038/s41598-018-38366-w. PMID: 30755653; PMCID: PMC6372693.
- Jorgensen AM, Varkey M, Gorkun A, Clouse C, Xu L, Chou Z, Murphy SV, Molnar J, Lee SJ, Yoo JJ, Soker S, Atala A. Bioprinted Skin Recapitulates Normal Collagen Remodeling in Full-Thickness Wounds. Tissue Eng Part A. 2020 May;26(9-10):512-526. doi: 10.1089/ten.TEA.2019.0319. Epub 2020 Jan 28. PMID: 31861970; PMCID: PMC7249461.
- Heimbach DM, Warden GD, Luterman A, Jordan MH, Ozobia N, Ryan CM, Voigt DW, Hickerson WL, Saffle JR, DeClement FA, Sheridan RL, Dimick AR. Multicenter postapproval clinical trial of Integra dermal regeneration template for burn treatment. J Burn Care Rehabil. 2003 Jan-Feb;24(1):42-8. doi: 10.1097/00004630-200301000-00009. PMID: 12543990.
- Pham C, Greenwood J, Cleland H, Woodruff P, Maddern G. Bioengineered skin substitutes for the management of burns: a systematic review. Burns. 2007 Dec;33(8):946-57. doi: 10.1016/j.burns.2007.03.020. Epub 2007 Sep 7. PMID: 17825993.
- Dantzer E, Braye FM. Reconstructive surgery using an artificial dermis (Integra): results with 39 grafts. Br J Plast Surg. 2001 Dec;54(8):659-64. doi: 10.1054/bjps.2001.3684. PMID: 11728107.
- Badylak SF. The extracellular matrix as a scaffold for tissue reconstruction. Semin Cell Dev Biol. 2002 Oct;13(5):377-83. doi: 10.1016/s1084952102000940. PMID: 12324220.
- Jorgensen AM, Chou Z, Gillispie G, Lee SJ, Yoo JJ, Soker S, Atala A. Decellularized Skin Extracellular Matrix (dsECM) Improves the Physical and Biological Properties of Fibrinogen Hydrogel for Skin Bioprinting Applications. Nanomaterials (Basel). 2020 Jul 29;10(8):1484. doi: 10.3390/nano10081484. PMID: 32751101; PMCID: PMC7466410.
- Jorgensen AM, Yoo JJ, Atala A. Solid Organ Bioprinting: Strategies to Achieve Organ Function. Chem Rev. 2020 Oct 14;120(19):11093-11127. doi: 10.1021/acs.chemrev.0c00145. Epub 2020 Sep 4. PMID: 32885956; PMCID: PMC8459198.
- Brown B, Lindberg K, Reing J, Stolz DB, Badylak SF. The basement membrane component of biologic scaffolds derived from extracellular matrix. Tissue Eng. 2006 Mar;12(3):519-26. doi: 10.1089/ten.2006.12.519. PMID: 16579685.
- Sadtler K, Sommerfeld SD, Wolf MT, Wang X, Majumdar S, Chung L, Kelkar DS, Pandey A, Elisseeff JH. Proteomic composition and immunomodulatory properties of urinary bladder matrix scaffolds in homeostasis and injury. Semin Immunol. 2017 Feb;29:14-23. doi: 10.1016/j.smim.2017.05.002. Epub 2017 Jun 2. PMID: 28583764; PMCID: PMC8509637.
- Riganti JM, Ciotola F, Amenabar A, Craiem D, Graf S, Badaloni A, Gilbert TW, Nieponice A. Urinary bladder matrix scaffolds strengthen esophageal hiatus repair. J Surg Res. 2016 Aug;204(2):344-350. doi: 10.1016/j.jss.2016.04.053. Epub 2016 May 3. PMID: 27565070.
- Young DA, McGilvray KC, Ehrhart N, Gilbert TW. Comparison of in vivo remodeling of urinary bladder matrix and acellular dermal matrix in an ovine model. Regen Med. 2018 Oct;13(7):759-773. doi: 10.2217/rme-2018-0091. Epub 2018 Aug 31. PMID: 30182807.
- Alvarez OM, Smith T, Gilbert TW, Onumah NJ, Wendelken ME, Parker R, Markowitz L. Diabetic Foot Ulcers Treated With Porcine Urinary Bladder Extracellular Matrix and Total Contact Cast: Interim Analysis of a Randomized, Controlled Trial. Wounds. 2017 May;29(5):140-146. Epub 2017 Feb 27. PMID: 28267677.
- Kim JS, Kaminsky AJ, Summitt JB, Thayer WP. New Innovations for Deep Partial-Thickness Burn Treatment with ACell MatriStem Matrix. Adv Wound Care (New Rochelle). 2016 Dec 1;5(12):546-552. doi: 10.1089/wound.2015.0681. PMID: 28078188; PMCID: PMC5165666.
- Martinson M, Martinson N. A comparative analysis of skin substitutes used in the management of diabetic foot ulcers. J Wound Care. 2016 Oct 1;25(Sup10):S8-S17. doi: 10.12968/jowc.2016.25.Sup10.S8. PMID: 27681811.
- Urmacher C. Histology of normal skin. Am J Surg Pathol. 1990 Jul;14(7):671-86. doi: 10.1097/00000478-199007000-00008. PMID: 1694059.
- Murphy SV, Skardal A, Nelson RA Jr, Sunnon K, Reid T, Clouse C, Kock ND, Jackson J, Soker S, Atala A. Amnion membrane hydrogel and amnion membrane powder accelerate wound healing in a full thickness porcine skin wound model. Stem Cells Transl Med. 2020 Jan;9(1):80-92. doi: 10.1002/sctm.19-0101. Epub 2019 Jul 21. Erratum in: Stem Cells Transl Med. 2024 Aug 10:szae046. doi: 10.1093/stcltm/szae046. PMID: 31328435; PMCID: PMC6954699.
- Branski LK, Mittermayr R, Herndon DN, Norbury WB, Masters OE, Hofmann M, Traber DL, Redl H, Jeschke MG. A porcine model of full-thickness burn, excision and skin autografting. Burns. 2008 Dec;34(8):1119-27. doi: 10.1016/j.burns.2008.03.013. Epub 2008 Jul 10. PMID: 18617332; PMCID: PMC2637517.
- Blackstone BN, Kim JY, McFarland KL, Sen CK, Supp DM, Bailey JK, Powell HM. Scar formation following excisional and burn injuries in a red Duroc pig model. Wound Repair Regen. 2017 Aug;25(4):618-631. doi: 10.1111/wrr.12562. Epub 2017 Jul 31. PMID: 28727221; PMCID: PMC5690826.
- Branski LK, Mittermayr R, Herndon DN, Norbury WB, Masters OE, Hofmann M, Traber DL, Redl H, Jeschke MG. A porcine model of full-thickness burn, excision and skin autografting. Burns. 2008 Dec;34(8):1119-27. doi: 10.1016/j.burns.2008.03.013. Epub 2008 Jul 10. PMID: 18617332; PMCID: PMC2637517.
- Midwood KS, Williams LV, Schwarzbauer JE. Tissue repair and the dynamics of the extracellular matrix. Int J Biochem Cell Biol. 2004 Jun;36(6):1031-7. doi: 10.1016/j.biocel.2003.12.003. PMID: 15094118.
- Xue M, Jackson CJ. Extracellular Matrix Reorganization During Wound Healing and Its Impact on Abnormal Scarring. Adv Wound Care (New Rochelle). 2015 Mar 1;4(3):119-136. doi: 10.1089/wound.2013.0485. PMID: 25785236; PMCID: PMC4352699.
- Aarabi S, Bhatt KA, Shi Y, Paterno J, Chang EI, Loh SA, Holmes JW, Longaker MT, Yee H, Gurtner GC. Mechanical load initiates hypertrophic scar formation through decreased cellular apoptosis. FASEB J. 2007 Oct;21(12):3250-61. doi: 10.1096/fj.07-8218com. Epub 2007 May 15. Erratum in: FASEB J. 2011 Oct;25(10):3751. PMID: 17504973.
- Reilly GC, Engler AJ. Intrinsic extracellular matrix properties regulate stem cell differentiation. J Biomech. 2010 Jan 5;43(1):55-62. doi: 10.1016/j.jbiomech.2009.09.009. Epub 2009 Oct 2. PMID: 19800626..
- Fayzullin A, Ignatieva N, Zakharkina O, Tokarev M, Mudryak D, Khristidis Y, Balyasin M, Kurkov A, Churbanov S, Dyuzheva T, Timashev P. Modeling of old scars: histopathological, biochemical and thermal analysis of the scar tissue maturation. Biology. 2021 Feb 9;10(2):136 Reilly GC, Engler AJ. Intrinsic extracellular matrix properties regulate stem cell differentiation. J Biomech. 2010 Jan 5;43(1):55-62. doi: 10.1016/j.jbiomech.2009.09.009. Epub 2009 Oct 2. PMID: 19800626..
- Santoso CN, Widyantara KA, Kencana NB, Setyajati FD, Setiawati A. A comprehensive analysis of collagen fiber dynamics in wound healing. Indonesian Journal of Pharmacy. 2024.
- Molnar JA, Lew WK, Rapp DA, Gordon ES, Voignier D, Rushing S, Willner W. Use of standardized, quantitative digital photography in a multicenter Web-based study. Eplasty. 2009;9:e4. Epub 2009 Jan 12. PMID: 19212431; PMCID: PMC2627310.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.