Medicine Group. 2024 July 24;5(7):859-865. doi: 10.37871/jbres1964.
Postural Orthostatic Tachycardia Syndrome (POTS) of Autoimmune Origin - Case and Review
Zora Lazúrová1,2*, Peter Mitro1, Ivica Lazúrová2, Monika Lukáčová1, Harald Heidecke3, Ján Lepej4, Igor Marin4 and Yehuda Shoenfeld5,6
2Department of Internal medicine, Medical Faculty, P.J. Šafárik University, University Hospital Košice, Slovakia
3CellTrend Laboratory, Luckenwalde, Germany
4Institute of nuclear and molecular medicine, Košice, Slovakia
5Zabludowicz centre for autoimmune diseases, Sheba Medical Center, Tel Aviv, Israel
6Reichman University, Herzolia, Israel
- Postural Orthostatic Tachycardia Syndrome (POTS)
- Anti-adrenergic receptor antibodies
- Antimuscarinic receptor antibodies
- Angiotensin II receptor antibodies
- Autoimmune dysautonomia
- Autoimmunity
Abstract
Introduction: Postural Orthostatic Tachycardia Syndrome (POTS) belongs to a group of orthostatic intolerance syndromes. It is defined by the increase in heart rate ≥30 bpm or heart rate higher than 120/min during first 10 minutes after standing, without significant decrease in blood pressure. It is a rare disease, typically affecting younger females. The dysregulation of autonomic nervous system plays a key role in the pathogenesis. The autonomic dysbalance can be caused by various neurohumoral mechanisms, receptor hypersensitivity, hypovolemia, mast cell dysregulation and last but not least, autoimmunity. The presence of autoantibodies targeted against various adrenergic and muscarinic receptors, as well, as angiotensin or endothelin receptors, has been identified in some but not all patients with POTS.
Case Description: We present a case of a young patient with an overlap syndrome of POTS and vasovagal vasodepressoric syncope diagnosed with the head up tilt test, without any identified trigger. The laboratory investigation documented the presence of anti-alpha-1, beta-1 and beta-2 adrenergic receptor antibodies, antimuscarinic receptor 3 antibodies and antibodies against angiotensin II type 1 receptor. MIBG (metaiodobenzylguanidine) scan has not revealed any changes in the sympathetic inervation of the heart. The initial treatment by betablockers, as well as by ivabradine failed. On midodrine medication, the symptoms of postural tachycardia and the reflex syncope improved.
Conclusion: The role of autoimmunity in POTS is currently studied, though the role of autoantibodies against autonomic nervous system in the pathogenesis of vasovagal syncope remains unclear.
Introduction
Postural Orthostatic Tachycardia Syndrome (POTS), originally described in 1993 by Schondorf and Low, is characterized by autonomic dysfunction commonly presenting with symptoms typical for orthostatic intolerance [1]. According to HRS (Heart Rhythm Society) Expert Consensus Statement, the POTS is defined as an increase in heart rate ≥ 30 bpm (≥ 40 bpm for those aged 12-19 years) or exceeding 120 bpm during 10 minutes of upright position, in the absence of an orthostatic hypotension. Symptoms duration should be at least 3 months and other causes of tachycardia should be excluded (anemia, fever, hyperthyroidism, medication, physical deconditioning, etc) [2,3]. Although POTS criteria do not include a significant orthostatic decrease in systolic Blood Pressure (BP), an “overlap“ syndromes, such as POTS in combination with orthostatic hypotension or vasovagal syncope may occur in some patients [3,4].
Postural orthostatic tachycardia syndrome affects predominantly women in the fertile age with the prevalence of 70-80% of all patients. The etiology of primary POTS is still unrecognized and probably it is multifactorial. Several precipitating factors were described to be related to manifestation of the syndrome, i.e. viral infections, vaccines, head trauma, surgery or pregnancy [4,5].
Pathophysiology of POTS remains poorly understood, though idiopathic hypovolemia, alpha receptor hypersensitivity, as well as autoimmune mechanisms due to the presence of autoantibodies against adrenergic and cholinergic receptors were proposed. Also, various humoral mechanisms have been studied, however, none of them has been identified as the primary cause of the syndrome [4,5].
Pathophysiological mechanisms may differ in various patients. In post viral POTS, autoimmunity is suspected as the main mechanism. In hyperadrenergic POTS increased sympathetic activity and excess of circulating catecholamines play a role. In a subset of patients small fiber neuropathy in lower limbs is present, leading to decreased venous return and compensatory heart rate increase during orthostasis. Moreover, relative or absolute hypovolemia is observed in many patients with POTS [6,7].
In last two decades there is a growing body of evidence that immune mediated mechanisms may play an important role in the etiopathogenesis of POTS [5-9]. Earlier studies demonstrated the presence of autoantibodies against ganglionic acetylcholine receptors in patients with orthostatic intolerance [10]. More recently researchers focused on the presence of adrenergic and muscarinic acetylcholine receptor antibodies resulting to hypothesis of novel autoimmune mechanisms in the etiology of POTS [11]. First, the presence of autoantibodies against angiotensin II, alpha-1 and beta-1/2 adrenergic receptors was demonstrated by Li H, et al. [13] and subsequently confirmed by few other studies [12]. Similarly, some studies also confirmed the presence of muscarinic acetylcholine receptor antibodies in approximately 50% of POTS patients. Integrating these data, the hypothesis of autoimmune complex was spawned. Autoimmune POTS together with chronic fatigue syndrome, complex regional pain syndrome or silicone implant incompatibility syndrome, could be incorporated in the new concept of autoimmune neurosensory dysautonomia [14].
Herein we refer to a patient with an autoimmune POTS in a combination with vasovagal syncope and the presence of autoantibodies against autonomic nervous system.
Case Report
A 34-year-old man was referred to the Syncopal Unit of the Institute of Cardiovascular Diseases in Košice, because of presyncopal and syncopal episodes lasting about one year. The family history was insignificant. In a previous medical history there was bicuspid aortic valve with a mild aortic regurgitation and a bipolar affective disorder.
The patient´s complaints consisted of repeating presyncopal/syncopal episodes with feeling of warmth, palpitations, tinnitus and vertigo, which were alleviated by sitting or lying down. He was not able to perform any physical activity, reported loss of appetite, weightloss and consequently developed depressive episodes. In addition, the patient complained of a chest pain and palpitations in an upright position with no relation to the syncopal episodes.
At the time of the first admission he did not take any medication. No viral infection, operation or vaccination preceded the onset of the symptoms.
On physical examination, the patient was eupnoic and well hydrated. Thyroid gland was not enlarged. Auscultation finding on the lung and heart were normal. His blood pressure at the time of examination was 110/80 mmHg and heart rate was 88 bpm.
Because of the chest pain he underwent repeated cardiological examinations including coronary CT angiography and invasive coronary angiography with normal interpretation. On the Holter ECG monitor (seven days) there were episodes of symptomatic sinus tachycardia with heart rate up to 190/min. Echocardiography revealed a bicuspid aortic valve with mild regurgitation and no stenosis? Left ventricular ejection function was normal (LVEF 60%). Blood laboratory parameters including thyroidal hormones were unremarkable.
He underwent Head- Up-Tilt (HUT) test with an increase in the heart rate from resting 85bpm to 115-137 bpm during the passive phase, while the BP remained unchanged 100-110/60-70 mmHg in the upright position. No symptoms occurred in a passive phase of Head Up Tilt test (HUT). Two minutes after nitroglycerine administration (400ug of nitroglycerin sublingually) his blood pressure dropped to 80/60 mmHg, with a marked increase in heart rate up to 170 – 180 bpm. This was accompanied with nausea, blurred vision, warmth, sweating, palpitations, and lightheadedness (identical with spontaneous presyncope). Test was finished when the presyncope ocurred, and the syncope was considered as inevitable. Since sinus tachycardia in first ten minutes was asymptomatic, but the symptoms occurred at the moment when the blood pressure dropped, the diagnosis of vasodepressor vasovagal syncope was estabilished in accordance with VASIS classification [15].
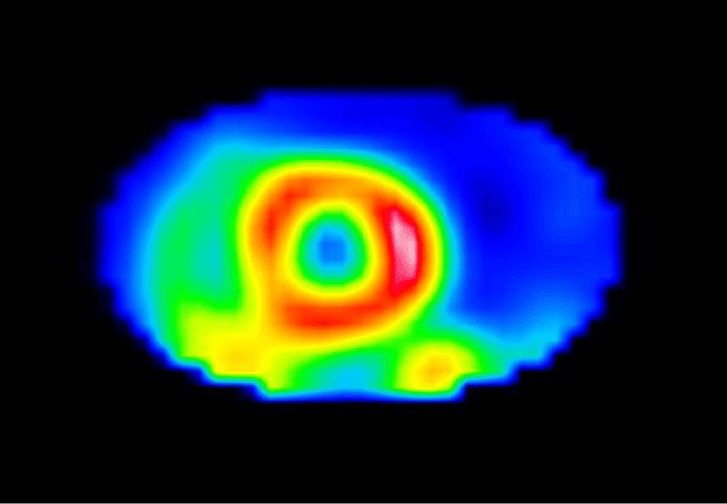
The MIBG scan was performed to assess the sympathetic innervation of the heart. Normal cardiac accumulation of isotope was described, with a normal SMI index 2.2 (normal more then 1.33) (Figure 1).
The diagnosis of POTS together with vasodepressor type of vasovagal syncope (VASIS 3) was determined in accordance with these findings after exclusion of other causes of postural tachycardia.
The patient was further tested for the presence of autoantibodies against angiotensin II type 1 receptor, endothelin receptor, adrenergic and muscarinic acetylcholine receptors. Blood samples were analyzed by ELISA method in CellTrend Laboratory, Germany.
The results of the antibody testing are depicted in the table 1. The autoantibody evaluation demonstrated presence of anti AT1R, anti alpha1-adrenergic and anti-beta-1, as well as anti-beta-2 adrenergic autoantibodies. Among the muscarinic cholinergic receptor autoantibodies, high titer of antimuscarinic cholinergic receptor-3 antibodies was detected. Based on these results we assumed the diagnosis of autoimmune POTS.
| Table 1: Results of the autoantibodies against adrenergic and cholinergic receptors in the patient with POTS. | ||
| Parameter | Ab levels (U/ml) | Normal range (U/ml) |
| Anti AT1R Ab | 22.9 Positive | 0-17 U/ml |
| Anti ETAR Ab | 15.4 At risk | 0-10 U/ml |
| Anti α1-adrenergic receptor Ab | 25.6 Positive | 0-7 U/ml |
| Anti α2-adrenergic receptor Ab | 7.1 Negative | 0-15 U/ml |
| Anti ß1-adrenergic receptor Ab | 50.0 Positive | 0-15 U/ml |
| Anti ß2- adrenergic receptor Ab | 39.1 Positive | 0-14 U/ml |
| Anti Muscarinic receptor-1 Ab | 2.9 Negative | 0-9 U/ml |
| Anti Muscarinic receptor-2 Ab | 3.0 Negative | 0-9 U/ml |
| Anti Muscarinic receptor-3 Ab | 42.9 Positive | 0-10 U/ml |
| Anti Muscarinic receptor-4 Ab | 3.6 Negative | 0-10.7 U/ml |
| Anti Muscarinic receptor-5 Ab | 5.8 Negative | 0-14.2 U/ml |
| Abbreviations: ATIR- Angiotensin II Receptor Type 1; ETAR- Endothelin A Receptor; AB- Antibodies | ||
The treatment with low dose betablockers – metoprolol 25 mg twice daily was initiated. However, this therapy lead to symptomatic arterial hypotension and was later switched to ivabradine in initial dose 2.5 mg twice daily with further dose up-titration to 5 mg two times a day. The effect of ivabradine was unsatisfactory, in the follow up, the blood pressure was 95/60 mmHg and heart rate was 60 bpm. Therefore, the alpha-agonist - midodrine was introduced as the third line of pharmacological treatment, which finally improved the orthostatic symptoms of the patient. The symptoms relieved at dose of 5 mg three times daily. The blood pressure increased to 110/70 mmHg and resting heart rate was 70 bpm.
Discussion
In this report we describe a patient with POTS having a documented autoimmune origin. The clinical symptoms of the patient started earlier before COVID-19 pandemic and were not triggered by any known viral infection, vaccination or stress situation. Evaluation of serum antibodies demonstrated their presence against multiple autonomic receptors, i.e. angiotensin II type 1 receptor, alpha-1 adrenergic receptor and both beta-1 and beta-2 adrenergic receptor. Moreover, the high titers of autoantibodies against muscarinic acetylcholine receptor type 3 were detected. These results clearly demonstrate an autoimmune POTS in probably genetically predisposed patient, without known trigger. No evidence on the presence of other autoimmune disease was detected.
Sympathetic nerve terminals in the heart showed normal density on MIBG scan, thus any structural pathologies of autonomic nervous system were excluded. Taken into account the presence of autoantibodies, it could be speculated that certain types of autonomic antibodies could exert stimulating effect on cardiac beta adrenergic receptors.
Another interesting finding is the occurrence of POTS simultaneously with reflex vasodepressor syncope. After three years the symptoms of POTS resolved in our patient and the clinical manifestation of reflex syncope prevailed with tendency to hypotension and bradycardia. However, approximately 25-30% of POTS patients have an “overlap“ syndrome with vasovagal syncope, it is not clear, if this association is mediated by autoimmune mechanisms [16]. Reflex syncope typically represents the situation of a sympathetic inhibition and a parasympathetic activation. How a complex interplay between the actions of our patient´s spectrum of stimulating and blocking anti-receptor antibodies may play a role in this specific situation remains to be answered. No studies about any possible autoimmune mechanisms in the pathogenesis of vasovagal syncope have been published to date.
Within two decades a few studies on relatively small groups of patients with POTS demonstrated the presence of autoantibodies against various receptors implicated in blood pressure regulation – such as angiotensin II type 1, alpha- and beta- adrenergic and muscarinic cholinergic receptors, indicating that autoimmune etiology may play a significant role in the etiopathogenesis of this syndrome [11-14]. After former reports on the presence of antibodies against ganglionic acetylcholine receptors, and concomitant experimental studies showing that autoantibodies against beta-1 adrenergic receptors have lead to a hyperadrenergic state due to overstimulation of cardiac beta-1-adreneric receptors, investigators started to measure these autoantibodies in the sera of POTS patients [17]. First, Wang XL, et al. [11] in a proteomic study demonstrated presence of autoantibodies cross-reacting with various myocardial proteins. Li H, et al. [12,18] documented that patients with POTS have elevated alpha-1 adrenergic receptor autoantibodies and exerting a partial peripheral antagonistic effect resulting in beta – adrenergic receptor mediated tachycardia. Coexisting beta-1 and beta-2 agonistic autoantibodies facilitate this state. Autoimmune etiology of POTS was also considered due to studies showing a higher prevalence of autoimmune disorders in patients with orthostatic tachycardia [19]. About 16 to 20% of POTS patients have coexisting autoimmune disease [20]. In a large cross-sectional online community based survey, the most common coexisting autoimmune conditions were Hashimoto thyroiditis (present in 6%), celiac disease (3%), Sjögren syndrome (3%), rheumatoid arthritis (2%), and systemic lupus erythematosus (2%) [21]. These findings regarding autoimmune etiology of POTS were supported by other studies, although on relatively small groups of patients. Several authors confirmed presence of autoantibodies against alpha-1 and beta-1 and beta-2 adrenergic receptor autoantibodies. Gunnning WT, et al. [22] documented almost 90% positivity for at least one antiadrenergic antibody among POTS patients, with the highest prevalence the highest prevalence against alpha-1 receptor. Furthermore, up to 50% of all POTS patients exhibited positivity for any muscarinic acetylcholine receptor antibodies, out of them anti-M4 was most prevalent. Authors also identified a positive correlation between the POTS symptoms and levels of alpha-1, alpha-2 and beta-1 adrenergic autoantibodies.
Our patient was positive for alpha-1 and both beta-1 and beta-2 receptor antibodies, which seem to be most tightly associates with POTS. Out of them, anti-beta-1 receptor antibodies were in the highest concentration (50 IU/ml). In the study group of Gunning WT, et al. [22] only one patient had higher level of beta-1 adrenergic antibodies then 40 IU/ml. Anti-angiotensin II receptor have been also previously described in the literature in POTS patients. Studies on antimuscarinic receptor antibodies are most inconsistent, probably the high levels of certain anti-M receptor antibodies reflect the general autoimmune affection of autonomic nervous system, however with lower specificity for POTS.
To the date, there have been few studies documenting higher prevalence of autonomic antibodies in patients with POTS published. However, the activity of these antibodies remained long time unknown. First, Fedorowski A, et al. [17] documented, that IgGs from sera of POTS patients activated alpha-1, beta-1 and beta-2 receptors in vitro. Similarly, Yu, et al. [23] identified in vitro activation of alpha-1 receptor (in 76%) and angiotensin II receptor (70%) after administration serum IgGs from POTS patients. Moreover, Li H, et al. [13] published, that POTS IgGs activated M2 acetylcholin receptors in dose-dependent manner. Additionally, authors suggest a possible inhibitory allosteric effect. Moreover, some studies also documented abnormal plasma cytokine concentrations in these patients, especially IL1-beta, IL21, TNF-alpha etc. [24]. Recently a few case reports of POTS triggered by COVID-19 infection or vaccination have been published [25,26].
However, it should be mentioned that some studies doubt the role of autoimmunity in POTS. Serum autoantibodies concentrations were not different in POTS patients and healthy controls in an international study from Canada and Sweden [27]. It seems that further studies are required to elucidate this question.
POTS has an excellent prognosis, though the quality of life might be significantly impaired. Therapeutical options include anti-adrenergic acting medication or modalities increasing the blood pressure or volume. There are no strict guidelines, however for patients without significant hypotension, most common first line medication are betablockers. Previously, propranolol has been considered as the most effective one in POTS, though recent study of Moon J, et al. [28] revealed comparable effects of propranolol, metoprolol and bisoprolol. In case of symptomatic hypotension, such as our case, another option is ivabradine, which selectively decreases sinus activity, though the effects in monotherapy are often insufficient [29]. Patients with low blood pressure usually respond well to alpha-1 agonist – midodrine, which increases blood pressure and consequently through the baroreflex leads to decrease in heart rate in POTS. According to Deng W, et al. [30] the efficacy of midodrine hydrochloride is significantly higher in patients with no increase in systolic blood pressure during orthostatic phase of HUTT, which usually excludes hyperadrenergic type of POTS, such as in our case.
Some studies have proven a positive effect of desmopressin on POTS, predominantly in patients with hypovolemic POTS with a tendency to hypotension. The role of autoimmunity may perspectively bring a novel therapeutical approach to patients with proven autoimmune cause of POTS [31]. Kesterson K, et al. [32] described a significant improvement of orthostatic symptoms in patients with severe POTS after administration of intravenous immunoglobulins. To the date, there have been only few case series published about the immunotherapy in POTS, therefore further research is definitely needed.
Conclusion
POTS is a syndrome with heterogenous and often unclear etiopathogenesis, however in significant number of patients, the autoimmunity plays a role. This case report highlights the autoimmune dysautonomia as a cause of POTS. The presence of autoantibodies against adrenergic and muscarinic receptors, as well as antibodies targeted to angiotensin and endothelin receptors should be evaluated in patients with POTS. Moreover, presented patient had an overlap syndrome including POTS and vasodepressor syncope, which generates further questions about the possible role of anti-receptor antibodies in vasovagal reflex syncope.
Conflict of Interest
All authors declare they have no conflict of interest.
Informed Consent
Informed consent was obtained from the patient.
References
- Schondorf R, Low PA. Idiopathic postural orthostatic tachycardia syndrome: an attenuated form of acute pandysautonomia? Neurology. 1993 Jan;43(1):132-7. doi: 10.1212/wnl.43.1_part_1.132. PMID: 8423877.
- Sheldon RS, Grubb BP 2nd, Olshansky B, Shen WK, Calkins H, Brignole M, Raj SR, Krahn AD, Morillo CA, Stewart JM, Sutton R, Sandroni P, Friday KJ, Hachul DT, Cohen MI, Lau DH, Mayuga KA, Moak JP, Sandhu RK, Kanjwal K. 2015 heart rhythm society expert consensus statement on the diagnosis and treatment of postural tachycardia syndrome, inappropriate sinus tachycardia, and vasovagal syncope. Heart Rhythm. 2015 Jun;12(6):e41-63. doi: 10.1016/j.hrthm.2015.03.029. Epub 2015 May 14. PMID: 25980576; PMCID: PMC5267948.
- Olshansky B, Cannom D, Fedorowski A, Stewart J, Gibbons C, Sutton R, Shen WK, Muldowney J, Chung TH, Feigofsky S, Nayak H, Calkins H, Benditt DG. Postural Orthostatic Tachycardia Syndrome (POTS): A critical assessment. Prog Cardiovasc Dis. 2020 May-Jun;63(3):263-270. doi: 10.1016/j.pcad.2020.03.010. Epub 2020 Mar 25. PMID: 32222376; PMCID: PMC9012474.
- Fedorowski A. Postural orthostatic tachycardia syndrome: clinical presentation, aetiology and management. J Intern Med. 2019 Apr;285(4):352-366. doi: 10.1111/joim.12852. Epub 2018 Nov 23. PMID: 30372565.
- Malkova AM, Shoenfeld Y. Autoimmune autonomic nervous system imbalance and conditions: Chronic fatigue syndrome, fibromyalgia, silicone breast implants, COVID and post-COVID syndrome, sick building syndrome, post-orthostatic tachycardia syndrome, autoimmune diseases and autoimmune/inflammatory syndrome induced by adjuvants. Autoimmun Rev. 2023 Jan;22(1):103230. doi: 10.1016/j.autrev.2022.103230. Epub 2022 Nov 5. PMID: 36347462.
- Aboseif A, Bireley JD, Li Y, Polston D, Abbatemarco JR. Autoimmunity and postural orthostatic tachycardia syndrome: Implications in diagnosis and management. Cleve Clin J Med. 2023 Jul 3;90(7):439-447. doi: 10.3949/ccjm.90a.22093. PMID: 37400156.
- Tomljenovic L, Colafrancesco S, Perricone C, Shoenfeld Y. Postural Orthostatic Tachycardia With Chronic Fatigue After HPV Vaccination as Part of the "Autoimmune/Auto-inflammatory Syndrome Induced by Adjuvants": Case Report and Literature Review. J Investig Med High Impact Case Rep. 2014 Mar 18;2(1):2324709614527812. doi: 10.1177/2324709614527812. PMID: 26425598; PMCID: PMC4528866.
- Vernino S, Bourne KM, Stiles LE, Grubb BP, Fedorowski A, Stewart JM, Arnold AC, Pace LA, Axelsson J, Boris JR, Moak JP, Goodman BP, Chémali KR, Chung TH, Goldstein DS, Diedrich A, Miglis MG, Cortez MM, Miller AJ, Freeman R, Biaggioni I, Rowe PC, Sheldon RS, Shibao CA, Systrom DM, Cook GA, Doherty TA, Abdallah HI, Darbari A, Raj SR. Postural orthostatic tachycardia syndrome (POTS): State of the science and clinical care from a 2019 National Institutes of Health Expert Consensus Meeting - Part 1. Auton Neurosci. 2021 Nov;235:102828. doi: 10.1016/j.autneu.2021.102828. Epub 2021 Jun 5. PMID: 34144933; PMCID: PMC8455420.
- Basantsova NY, Starshinova AA, Dori A, Zinchenko YS, Yablonskiy PK, Shoenfeld Y. Small-fiber neuropathy definition, diagnosis, and treatment. Neurol Sci. 2019 Jul;40(7):1343-1350. doi: 10.1007/s10072-019-03871-x. Epub 2019 Apr 9. PMID: 30968230.
- Sandroni P, Low PA. Other autonomic neuropathies associated with ganglionic antibody. Auton Neurosci. 2009 Mar 12;146(1-2):13-7. doi: 10.1016/j.autneu.2008.10.022. Epub 2008 Dec 4. PMID: 19058765; PMCID: PMC2671239.
- Wang XL, Chai Q, Charlesworth MC, Figueroa JJ, Low P, Shen WK, Lee HC. Autoimmunoreactive IgGs from patients with postural orthostatic tachycardia syndrome. Proteomics Clin Appl. 2012 Dec;6(11-12):615-25. doi: 10.1002/prca.201200049. Epub 2012 Nov 8. PMID: 23002038; PMCID: PMC3786406.
- Li H, Yu X, Liles C, Khan M, Vanderlinde-Wood M, Galloway A, Zillner C, Benbrook A, Reim S, Collier D, Hill MA, Raj SR, Okamoto LE, Cunningham MW, Aston CE, Kem DC. Autoimmune basis for postural tachycardia syndrome. J Am Heart Assoc. 2014 Feb 26;3(1):e000755. doi: 10.1161/JAHA.113.000755. PMID: 24572257; PMCID: PMC3959717.
- Li H, Zhang G, Forsythe E, Okamoto LE, Yu X. Implications of Antimuscarinic Autoantibodies in Postural Tachycardia Syndrome. J Cardiovasc Transl Res. 2022 Apr;15(2):438-440. doi: 10.1007/s12265-021-10167-z. Epub 2021 Aug 18. PMID: 34409582; PMCID: PMC8854442.
- Shoenfeld Y, Ryabkova VA, Scheibenbogen C, Brinth L, Martinez-Lavin M, Ikeda S, Heidecke H, Watad A, Bragazzi NL, Chapman J, Churilov LP, Amital H. Complex syndromes of chronic pain, fatigue and cognitive impairment linked to autoimmune dysautonomia and small fiber neuropathy. Clin Immunol. 2020 May;214:108384. doi: 10.1016/j.clim.2020.108384. Epub 2020 Mar 17. PMID: 32171889.
- Brignole M, Menozzi C, Del Rosso A, Costa S, Gaggioli G, Bottoni N, Bartoli P, Sutton R. New classification of haemodynamics of vasovagal syncope: beyond the VASIS classification. Analysis of the pre-syncopal phase of the tilt test without and with nitroglycerin challenge. Vasovagal Syncope International Study. Europace. 2000 Jan;2(1):66-76. doi: 10.1053/eupc.1999.0064. PMID: 11225598.
- Garland EM, Celedonio JE, Raj SR. Postural Tachycardia Syndrome: Beyond Orthostatic Intolerance. Curr Neurol Neurosci Rep. 2015 Sep;15(9):60. doi: 10.1007/s11910-015-0583-8. PMID: 26198889; PMCID: PMC4664448.
- Fedorowski A, Li H, Yu X, Koelsch KA, Harris VM, Liles C, Murphy TA, Quadri SMS, Scofield RH, Sutton R, Melander O, Kem DC. Antiadrenergic autoimmunity in postural tachycardia syndrome. Europace. 2017 Jul 1;19(7):1211-1219. doi: 10.1093/europace/euw154. PMID: 27702852; PMCID: PMC5834103.
- Li H, Zhang G, Zhou L, Nuss Z, Beel M, Hines B, Murphy T, Liles J, Zhang L, Kem DC, Yu X. Adrenergic Autoantibody-Induced Postural Tachycardia Syndrome in Rabbits. J Am Heart Assoc. 2019 Oct;8(19):e013006. doi: 10.1161/JAHA.119.013006. Epub 2019 Sep 24. PMID: 31547749; PMCID: PMC6806023.
- Blitshteyn S. Autoimmune markers and autoimmune disorders in patients with postural tachycardia syndrome (POTS). Lupus. 2015 Nov;24(13):1364-9. doi: 10.1177/0961203315587566. Epub 2015 Jun 1. PMID: 26038344.
- Dahan S, Tomljenovic L, Shoenfeld Y. Postural Orthostatic Tachycardia Syndrome (POTS)--A novel member of the autoimmune family. Lupus. 2016 Apr;25(4):339-42. doi: 10.1177/0961203316629558. Epub 2016 Feb 3. PMID: 26846691.
- Shaw BH, Stiles LE, Bourne K, Green EA, Shibao CA, Okamoto LE, Garland EM, Gamboa A, Diedrich A, Raj V, Sheldon RS, Biaggioni I, Robertson D, Raj SR. The face of postural tachycardia syndrome - insights from a large cross-sectional online community-based survey. J Intern Med. 2019 Oct;286(4):438-448. doi: 10.1111/joim.12895. Epub 2019 Apr 16. PMID: 30861229; PMCID: PMC6790699.
- Gunning WT 3rd, Kvale H, Kramer PM, Karabin BL, Grubb BP. Postural Orthostatic Tachycardia Syndrome Is Associated With Elevated G-Protein Coupled Receptor Autoantibodies. J Am Heart Assoc. 2019 Sep 17;8(18):e013602. doi: 10.1161/JAHA.119.013602. Epub 2019 Sep 9. PMID: 31495251; PMCID: PMC6818019.
- Yu X, Li H, Murphy TA, Nuss Z, Liles J, Liles C, Aston CE, Raj SR, Fedorowski A, Kem DC. Angiotensin II Type 1 Receptor Autoantibodies in Postural Tachycardia Syndrome. J Am Heart Assoc. 2018 Apr 4;7(8):e008351. doi: 10.1161/JAHA.117.008351. PMID: 29618472; PMCID: PMC6015435.
- Gunning WT 3rd, Stepkowski SM, Kramer PM, Karabin BL, Grubb BP. Inflammatory Biomarkers in Postural Orthostatic Tachycardia Syndrome with Elevated G-Protein-Coupled Receptor Autoantibodies. J Clin Med. 2021 Feb 6;10(4):623. doi: 10.3390/jcm10040623. PMID: 33562074; PMCID: PMC7914580.
- Minhas R, Bharadwaj AS. COVID-19-Induced Postural Orthostatic Tachycardia Syndrome and Dysautonomia. Cureus. 2023 Jun 10;15(6):e40235. doi: 10.7759/cureus.40235. PMID: 37435242; PMCID: PMC10332885.
- Mallick D, Goyal L, Chourasia P, Zapata MR, Yashi K, Surani S. COVID-19 Induced Postural Orthostatic Tachycardia Syndrome (POTS): A Review. Cureus. 2023 Mar 31;15(3):e36955. doi: 10.7759/cureus.36955. PMID: 37009342; PMCID: PMC10065129.
- Hall J, Bourne KM, Vernino S, Hamrefors V, Kharraziha I, Nilsson J, Sheldon RS, Fedorowski A, Raj SR. Detection of G Protein-Coupled Receptor Autoantibodies in Postural Orthostatic Tachycardia Syndrome Using Standard Methodology. Circulation. 2022 Aug 23;146(8):613-622. doi: 10.1161/CIRCULATIONAHA.122.059971. Epub 2022 Jun 29. PMID: 35766055; PMCID: PMC9390234.
- Moon J, Kim DY, Lee WJ, Lee HS, Lim JA, Kim TJ, Jun JS, Park B, Byun JI, Sunwoo JS, Lee ST, Jung KH, Park KI, Jung KY, Kim M, Lee SK, Chu K. Efficacy of Propranolol, Bisoprolol, and Pyridostigmine for Postural Tachycardia Syndrome: a Randomized Clinical Trial. Neurotherapeutics. 2018 Jul;15(3):785-795. doi: 10.1007/s13311-018-0612-9. PMID: 29500811; PMCID: PMC6095784.
- Taub PR, Zadourian A, Lo HC, Ormiston CK, Golshan S, Hsu JC. Randomized Trial of Ivabradine in Patients With Hyperadrenergic Postural Orthostatic Tachycardia Syndrome. J Am Coll Cardiol. 2021 Feb 23;77(7):861-871. doi: 10.1016/j.jacc.2020.12.029. PMID: 33602468.
- Deng W, Liu Y, Liu AD, Holmberg L, Ochs T, Li X, Yang J, Tang C, Du J, Jin H. Difference between supine and upright blood pressure associates to the efficacy of midodrine on postural orthostatic tachycardia syndrome (POTS) in children. Pediatr Cardiol. 2014 Apr;35(4):719-25. doi: 10.1007/s00246-013-0843-9. Epub 2013 Nov 20. PMID: 24253613.
- Coffin ST, Black BK, Biaggioni I, Paranjape SY, Orozco C, Black PW, Dupont WD, Robertson D, Raj SR. Desmopressin acutely decreases tachycardia and improves symptoms in the postural tachycardia syndrome. Heart Rhythm. 2012 Sep;9(9):1484-90. doi: 10.1016/j.hrthm.2012.05.002. Epub 2012 May 3. PMID: 22561596; PMCID: PMC3419341.
- Kesterson K, Schofield J, Blitshteyn S. Immunotherapy with subcutaneous immunoglobulin or plasmapheresis in patients with postural orthostatic tachycardia syndrome (POTS). J Neurol. 2023 Jan;270(1):233-239. doi: 10.1007/s00415-022-11344-z. Epub 2022 Aug 26. PMID: 36008726.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.