Biology Group. 2024 August 31;5(8):1073-1080. doi: 10.37871/jbres1992.
Effects of Microplastics on the Intestinal Barrier, Gut Microbiota and Antibiotic Resistance Genes: A Mini-Review
Faezeh Jahedi1,2, Behnam Ahmadi1, Neamatollah Jaafarzadeh Haghighi Fard2,3*, Pouria Abolfazli4 and Mahmood Maniati5
2Department of Environmental Health Engineering, School of Health, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran
3Environmental Technologies Research Center, Medical Basic Sciences Research Institute, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran
4College of Medicine, Xi’an Jiaotong University, Xi’an, 710061, China
5School of Medicine, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran
- Microplastics
- Nanoplastics
- Antibiotic-Resistant Genes (ARG)
- Microbiota
Abstract
Microplastics (MPs) have aroused a global health concern and their coexistence with antibiotics is inevitable. However, how MPs would affect the bioaccumulation and risks of antibiotics in humans remains poorly understood. In this study, we reviewed several articles that examined the abundance and expression changes of ARGs in various environments. These changes were observed upon exposure to antibiotics such as tetracycline, sulfamethoxazole, and macrolides, as well as MPs, or a combination of both. To assess the impact of MPs on ARG abundance, we compiled data from various studies using tables of ARG abundances in tissues or environmental samples. By synthesizing this information, we aimed to identify which ARGs were upregulated or downregulated in response to micronanoplastics and antibiotics. Our findings provide insights into the potential risks posed by environmental pollutants and underscore the importance of understanding the dynamics of ARGs in response to MPs. We identified that shifts in gut microbiota contributed to the changes in ARG profiles. Combined, our results demonstrate that MPs reduced the bioaccumulation of tetracycline, sulfamethoxazole, and macrolides, but they enhanced its effects on gut microbiota and the antibiotic resistome indicating they might have high risks to humans.
Introduction
Over the past few decades, plastic production has increased significantly, leading to widespread environmental contamination [1,2]. Due to their low cost, lightweight, and durability, plastics are used in a wide range of products [3]. In 2021 alone, more than 390 million tons of plastic were produced worldwide, and this amount is rapidly increasing [4-6]. Large plastic wastes break down into smaller fragments known as Microplastics (MPs), which are less than 5 mm in size [7,8]. These MPs have been found in various environments and foods, and humans can be exposed to them through eating, drinking, breathing, and skin contact. Studies have even found MPs in human stool samples, indicating their presence in the human body [9]. MPs can harm organisms by reducing their feeding, survival, and reproduction rates [10]. They can also disrupt gut microbiota, which is essential for health, potentially leading to diseases and behavioral issues [11]. While these effects have been studied in soil worms, mussels, and fish, more research is needed on mammals. MPs can interact with other pollutants, such as antibiotics, affecting their accumulation and impact on organisms [12]. Combining MPs and antibiotics is unavoidable, and MPs can alter the gut microbiome and antibiotic-resistance genes. Most studies use commercially produced MPs, which differ from real-life MPs in various properties. Antibiotic Resistance Genes (ARGs) are present in bacterial chromosomes, phages, and plasmids, which serve as biological vectors for spreading antibiotic resistance to humans [13]. A total of 469 articles were initially retrieved from Scopus, Web of Science, and PubMed. After the removal of duplicates, 256 unique articles were retained. The authors conducted an initial screening of all abstracts and titles based on predetermined inclusion and exclusion criteria, eliminating studies that were entirely unrelated. The exclusion criteria encompassed errata, reviews, notes, conference papers, books, book sections, responses, comments, opinions, discussions, and articles that were not relevant. The inclusion criteria, guided by the study's objectives and keywords, targeted research focused on the impact of Microplastics (MPs) on Antibiotic Resistance Genes (ARG) in organisms. While previous studies have explored the effects of MPs on ARG and their environmental implications, this study presents the first systematic review examining the influence of MPs on ARG and gut microbiota in animals.
Overview of effects of MPs and antibiotic resistance in the environment
MPs are small plastic particles that can transport Antibiotic Resistance Genes (ARGs), aiding in the spread of antibiotic-resistant bacteria. In sewage treatment plants, MPs frequently carry bacteria with ARGs, especially on materials like PVC [14]. High concentrations of ARGs are found on MPs in marine and freshwater environments, posing threats to aquatic organisms and human health [15,16]. In agricultural soils, MPs and ARGs interact with various pollutants and can be inhaled from the air, highlighting the necessity for further research on their health effects [17-21].
Impact of MPs on intestinal health and antibiotic resistance
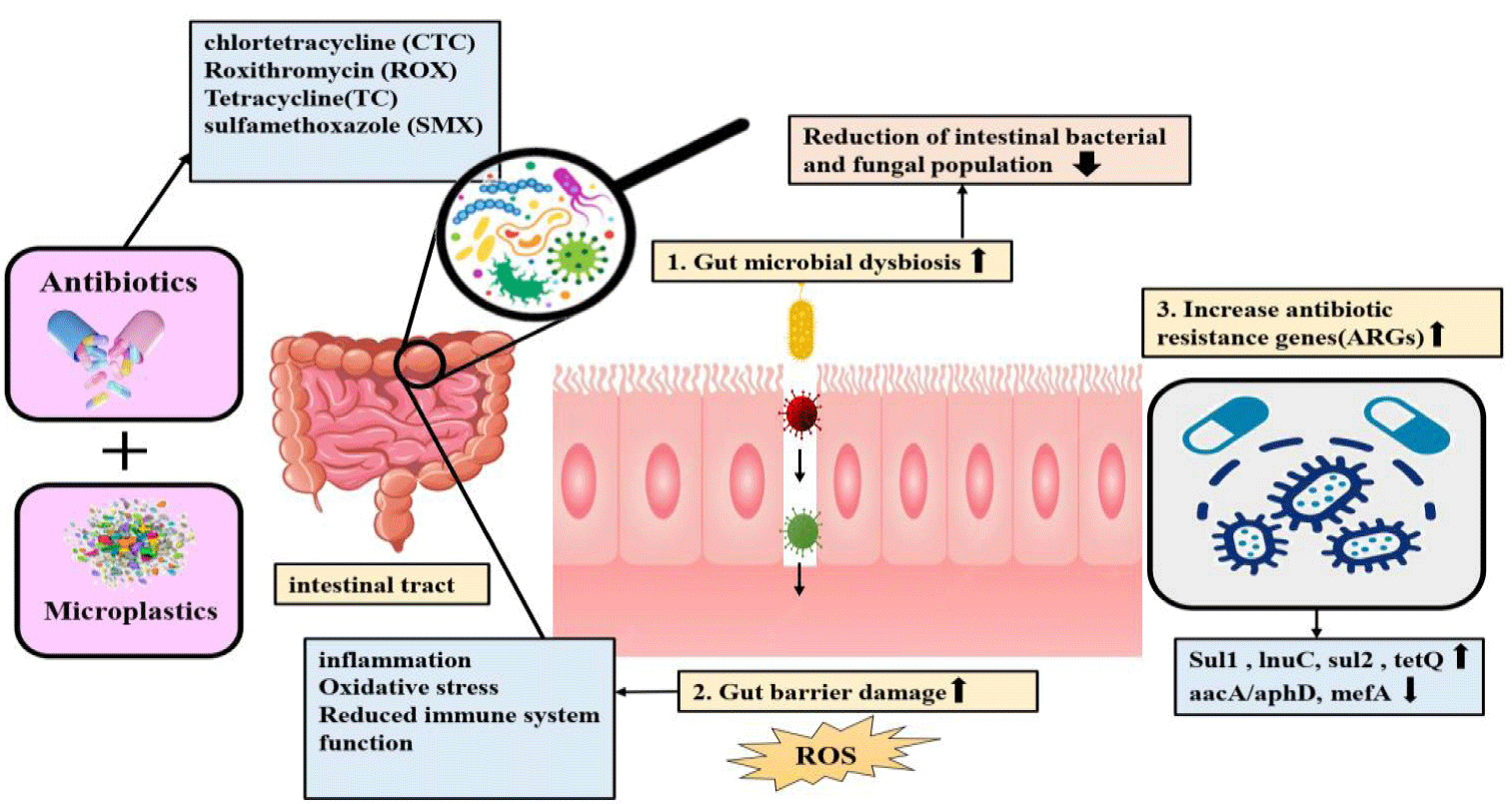
MPs significantly harm the gut, weakening its barrier against pollutants. A study on mice revealed that MPs, combined with sulfamethoxazole, cause intestinal disorders, barrier destruction, reduced drug efficacy, and increased antibiotic-resistant genes [22]. MPs disrupt the intestinal microbiota, affecting microbial and fungal populations, metabolic pathways, and genes responsible for antibiotic resistance. They also damage epithelial cells, leading to the release of reactive oxygen species and the activation of antioxidant enzymes. This damage allows pollutants and pathogens to enter, increasing inflammation and cytokines like interleukin 10 and 6. These findings align with studies conducted on soil organisms [23,24].
Antibiotic resistance mechanisms include the chemical modification of target enzymes, enzymatic inactivation of antibiotics, and efflux pumps. Bacteria can alter the enzymes that antibiotics target, making the antibiotics less effective. Some bacteria produce enzymes that break down and inactivate antibiotics. Additionally, bacteria can pump antibiotics out of their cells, reducing the concentration of the antibiotic inside the cell and thus its effectiveness. In natural environments, MPs particles can act as carriers for environmental microorganisms, including antibiotic-resistant bacteria, due to their hydrophobic surfaces. These particles can support the formation of microbial biofilms, which provide a suitable environment for the exchange of antibiotic resistance genes between bacteria. MPs can also aid in the accumulation and transfer of antibiotic resistance genes among bacteria, contributing to the spread of antibiotic resistance in the environment. These two topics are interconnected and demonstrate how environmental factors like MPs can contribute to the spread of antibiotic resistance.
In the reviewed studies, various organisms were exposed to different drugs and MPs of varying sizes through diverse methods and under specific conditions (Table 1). This exposure resulted in changes in gene expression and the functioning of various organs. By extracting DNA, researchers can precisely analyze these changes and determine the specific effects of MPs on altered genes. Liu J, et al. [22] discovered that when mice were exposed to Polystyrene (MP), the bioaccumulation of Sulfamethoxazole (SMX) decreased in their liver, lungs, spleen, heart, and kidneys. This exposure also led to increased antibiotic resistance in the mice. Using high-throughput quantitative PCR (HT-qPCR) with the Wafergen SmartChip Real-time PCR system, they found an increase in the genes sul1, lnuC, acrA-03, mefA, and ermB, while the gene aacA/aphD decreased. The impact of MPs on gut microbiota and antibiotic-resistance genes is illustrated in figure 1. Gao B, et al. [23] showed that MPs can hurt the intestinal microbiota (microorganisms that live in the intestines of animals). Gut microbiota is involved in nutrition, growth, and immune response [31]. The animal gut can act as a reservoir of ARGs for various reasons [32]. Previous studies have shown that changes in gut microbiota can affect the amount of ARGs in the gut of animals [33]. Research suggests that fungi in our gut are more vulnerable to PS than bacteria. These gut fungi can influence our immune system by triggering T helper cells 17, which can help control inflammation [34]. Disruptions in gut fungi balance can negatively impact the host organism’s health. In this study, male mice were fed Polystyrene (MPs) of various sizes. The results showed that MPs influence antibiotic resistance in mice, with an increase in genes like Sul2 and VanSD, and a decrease in MepR-macrolide-efflux protein-reverse transcriptase. Zhimin Xu’s research also indicates that MPs (PVC) can trigger the release of inflammatory cytokines, leading to intestinal inflammation, and cause stool hardening due to mucus from Black Soldier Flies (BSFs). This prevents organic mineralization and antibiotic degradation. Additionally, there was an increase in genes such as tetX2, tetQ, tetG, tetM, tetA(G), tetA(P), sul1, and sul2, particularly those coding for tetracyclines and sulfonamides.
| Table 1: Effects of MPs on antibiotic resistance in living creatures. | |||||||
| Host | Antibiotics/MPs Exposure | Genes (ARGs) | Enrichment | Type MPs | Method for ARG extraction | Size | Ref |
| Mice | MPs + SMX | sul1- lnuC- acrA-03- mefA- ermB | Up-regulated | polyethylene terephthalate | High Throughput Quantitative PCR (HT-qPCR) | 2 μm to 631 μm | [22] |
| aacA/aphD | Down-regulated | ||||||
| SMX | sul1 | No change | |||||
| lnuC- acrA-03- mefA- ermB- aacA/aphD | Up-regulated | ||||||
| MPs | sul1- lnuC- mefA- ermB- | Down-regulated | |||||
| acrA-03- aacA/aphD | Up-regulated | ||||||
| D. Magna | MPs | EcR- sul1 | Down-regulated | Polystyrene | Polymerase Chain amplification (PCR) | 20-30 μm | [25] |
| AK- TRxR- Vtg1- CAT- GSTs | Up-regulated | ||||||
| MPs +S. flexneri | AK- Vtg1 | No change | |||||
| EcR- sul1 | Down-regulated | ||||||
| GSTs- CAT- TRxR | Up regulated | ||||||
| S. flexneri | Vtg1- GSTs | No change | |||||
| TRxR- EcR | Down-regulated | ||||||
| AK- CAT- sul1 | Up-regulated | ||||||
| oyster farm-Biofilms | MPs | blaTEM- intI1- qnrS- sul1 | Up-regulated | Polypropylene (PP), Polyethylene (PE), Polyethylene terephthalate (PET), Polyhydroxybutyrate (PHB) and Polylactic acid (PLA) | qPCR | 3-4 mm | [26] |
| oyster farm-Water | blaTEM- intI1- qnrS | Down-regulated | |||||
| sul1 | Up-regulated | ||||||
| Muscovy ducks (Cairina moschata) | MPs | macB- bcrA- tetA (58)- msbA- tetQ | Up-regulated | polystyrene | PCR | 10-100 μm | [27] |
| tetA(46)- tetB (60)- tet (43) | Down-regulated | ||||||
| CTC + MPs | tet(35)- tet36- tet37- tet38- tetA (60)- tetB (46)- tetW- tetS- otr (B) | Up-regulated | |||||
| CTC | tetQ- tet37 | Up-regulated | |||||
| Carassius auratus | polystyrene microplastics alone or in combination with the 100 μg/L Roxithromycin (ROX) | Baca- tetpb - sul1 - tetpa - cml e3- tetg- ant2ia- teta- tetm- mefa- tetz- cml e8- Inub- aph6id- aph33ib- bl2a_nps- ermg- sul2- tetc- cml el- catb3 | Up-regulated | Polystyrene | To extract metagenomic DNA, the intestinal contents were gently squeezed out by a sterile instrument and collected into a sterile container | 5 µm | [28] |
| mice | PS0.1 | VanSD- Sul2- tetracycline resistance protein- dihydropteroate synthase- sulfonamide resistance protein | Up-regulated | polystyrene | ShortBRED was used to profile antibiotic resistance genes | PS0.1 (diameter 0.05-0.1 µm) or PS10 (diameter 9-10 µm) | [23] |
| MepR- macrolide-efflux protein- reverse transcriptase | Down-regulated | ||||||
| PS10 | ANT(6)-Ib-APH(3’)-IIIa-TetX-TnpA-aminoglycoside phosphotransferase-integrase-(sigE)RNA polymerase sigma-70 factor-microcompartment protein | Up regulated | |||||
| MepR | Down-regulated | ||||||
| black soldier fly | antibiotics | tetX2- tetQ- tetG- tetM- tetA(G)- tetA(P)- sul1- sul2 | Up-regulated | PVC | HT-qPCR | 200 nm | [24] |
| earthworms | M4* | tetX-sul1-QnrS2-TriC-Klebsiella pneumoniae acrA | Down-regulated | Polystyrene | polymerase chain reaction | (100 nm, 10 and 100 µm) | [29] |
| FosB3-TRU-1-OXA-12-cphA2 | Up-regulated | ||||||
| soil fauna (Enchytraus crypticus) | TC* | blaSHV-nisB | Down-regulated | polystyrene | high-throughput quantitative PCR(HT-qPCR) | 50-100 nm | [30] |
| ermX-aadA1-cfxA-blaGES-ttgB- tetG-aadA2-qacEdelta1-qacH-tetB-02-vanXD | Up-regulated | ||||||
| TC and NP | aac(6")-II- aac(6")-lb(akaaac A4)- fox5 - fosX- cmlA1- blaZ | Up-regulated | |||||
| TC: Tetracycline; PS: Polystyrene; CTC: Chlortetracycline; M4: 100 mg/kg of 10 µm MPs. | |||||||
Similar studies also confirm the accuracy of this article [25]. It is necessary to mention that MP pollution is creating new breeding grounds for bacteria in water [35]. These bacteria can form layers on the MPs, which can make them more resistant to antibiotics and harbor genes that spread antibiotic resistance [36,37]. As MPs become more widespread, this could lead to the increased transfer of ARGs among bacteria, harming both the environment and living things [38-40]. For example, in D. magna as an important part of the aquatic food chain, similar effects on the intestine and the increase of the ARG sul1 have been observed. This study deals with the effect of MP particle size, types, and concentration, which are important factors in the emergence of effects. Since nanoplastics have a smaller size and a larger specific surface area than MPs, the possibility of combining them with antibiotics is higher [41]. Also, studies have shown that MPs can cause more damage when they are older [28]. In this study, exposure to 20-30 micrometer polystyrene MPs led to the increase of AK-TRxR-Vtg1-CAT-GSTs genes, which shows that micro PS may cause disturbance in the balance of vitellogenesis, and endocrine disorders. These MPs also led to the enrichment of ARGs (sul1, qnrS and blaTEM) in oysters [42] In addition to Daphnia, the effect of MPs with Roxithromycin (ROX) on the intestine of Carassius auratus was investigated by Peng Zhang. The result showed that MPs caused the accumulation of ROX in the intestine of fish. ROX is an antibiotic that inhibits protein synthesis in cells. [43] In this study, ROX altered microbial diversity in the gut and reduced beneficial bacteria [44]. It also increased bacteria resistant genes Baca- tetpb- sul1- tetpa- cml e3- tetg- ant2ia- teta- tetm- mefa- tetz- cml e8- Inub- aph6id- aph33ib- bl2a_nps- ermg- sul2- tetc- cml el- catb3.
In addition to the studies conducted on aquatic animals, studies on terrestrial animals and earthworms [45]. Have also been conducted by Ma J, et al. [30] and Xu G, et al. [29] who exposed soil invertebrates Enchytraeus crypticus to MPs and tetracycline and investigated antibiotic resistance genes using quantitative PCR method. Their results showed an increase in aac(6")-II-aac(6")-lb (akaaacA4) - fox5- fosX- cmlA1- blaZ. Also, higher concentrations of MPs were found to have a direct effect on the increase of ARGs.
Research has also focused on the presence of ARGs and MPs in birds. For example, Bing Liu studied the effects of MPs and tetracycline on Muscovy ducks. The study found that exposure to MPs increased the levels of macB, bcrA, tetA, msbA, and tetQ genes. Liu concluded that MPs not only disrupt the balance of reactive oxygen species (ROS) and antioxidant capacity but also alter antibiotic resistance and facilitate the transfer of ARGs to the ecosystem [46].
Conclusion
There is a connection between human antibiotic resistance and the presence of MPs in the human body. Antibiotic resistance in 2019 led to the death of about 1 million people. The effects of this resistance in the human body can increase the chance of contracting the disease as well as increasing the cost and length of treatment and recovery. Although the development of resistance is commonly associated with the overuse and prescription of antibiotics for many diseases, recent studies show that MPs play a very influential role in Antibiotic resistance and Gut microbiota. Gut microbiota has a great impact on host homeostasis and disease. Changes in the composition of gut bacteria may have additional health implications. In the present study, studies were reviewed that investigated the frequency and expression changes of antibiotic resistance genes (ARGs) in the body of different organisms. In these studies, living organisms are exposed to MPs and antibiotics such as tetracycline, sulfamethoxazole, and macrolides. The results showed that ingested MPs increase resistance to macrolides, lincosamides and aminoglycosides, tetracycline, and sulfamethoxazole, all of which are drugs that are used to treat infections such as pneumonia and upper and lower respiratory tract infections. Antibiotic resistance mechanisms include altering target enzymes, inactivating antibiotics, and using efflux pumps to expel antibiotics from bacterial cells. In natural environments, MPs particles can carry microorganisms, including antibiotic-resistant bacteria, and support biofilm formation. This facilitates the exchange and accumulation of antibiotic resistance genes, contributing to the spread of antibiotic resistance. It is suggested that future research should go towards human research investigating the effect of MPs on the increase of resistant genes.
Innovations and Limitations of this Study
Innovations
Focus on Gut Microbiota and ARGs: This study uniquely examines the impact of microplastics on gut microbiota and antibiotic resistance genes in animals, addressing a relatively unexplored area of research.
Comprehensive Approach: Unlike previous research that may have examined specific aspects in isolation, this study provides a thorough review of the effects of MPs on ARGs across various environments and organisms, offering a more holistic understanding of the issue.
Limitations
Scope and Generalizability: This review predominantly focuses on research involving non-human organisms such as mice, soil worms, and aquatic species. Consequently, the findings may not be directly transferable to human health impacts. Additionally, the Microplastics (MPs) used in the reviewed studies are typically commercially manufactured, differing in size, shape, and chemical composition from those encountered in natural environments. This discrepancy could affect the generalizability of the results to real-world scenarios.
Consideration of pollutant interactions
While the interaction between MPs and antibiotics was analyzed, the review did not account for other environmental pollutants. This omission may limit the comprehensive understanding of how multiple pollutants interact and their cumulative effects on Antibiotic Resistance Genes (ARGs).
Methodological variability
The studies included in this review utilized diverse methodologies for the extraction and analysis of ARGs, potentially introducing variability and inconsistencies in the results. The adoption of standardized methodologies is necessary for achieving more consistent and comparable findings across studies.
References
- Zhao J, Lan R, Wang Z, Su W, Song D, Xue R, Liu Z, Liu X, Dai Y, Yue T, Xing B.Microplastic fragmentation by rotifers in aquatic ecosystems contributes to global nanoplastic pollution. Nature nanotechnology. 2024;19(3):406-414. doi: 10.1038/s41565-023-01534-9
- Ben Stride, Abolfathi S, Bending GD, Pearson J. Quantifying microplastic dispersion due to density effects. J Hazard Mater. 2024 Mar 15;466:133440. doi: 10.1016/j.jhazmat.2024.133440. Epub 2024 Jan 6. PMID: 38246058.
- Lee Y, Cho J, Sohn J, Kim C. Health Effects of Microplastic Exposures: Current Issues and Perspectives in South Korea. Yonsei Med J. 2023 May;64(5):301-308. doi: 10.3349/ymj.2023.0048. PMID: 37114632; PMCID: PMC10151227.
- Sabatino R, Zullo R, Di Cesare A, Piscia R, Musazzi S, Corno G, Volta P, Galafassi S. Traditional and biodegradable plastics host distinct and potentially more hazardous microbes when compared to both natural materials and planktonic community. J Hazard Mater. 2024 Mar 5;465:133166. doi: 10.1016/j.jhazmat.2023.133166. Epub 2023 Dec 5. PMID: 38101010.
- Han Z, Osman R, Liu Y, Wei Z, Wang L, Xu M. Analyzing the impacts of cadmium alone and in co-existence with polypropylene microplastics on wheat growth. Front Plant Sci. 2023 Aug 10;14:1240472. doi: 10.3389/fpls.2023.1240472. PMID: 37636097; PMCID: PMC10449543.
- Zhang Z, Kang Y, Wang W, Xu L, Liu J, Zhang Z, Wu H. Low-density polyethylene microplastics and biochar interactively affect greenhouse gas emissions and microbial community structure and function in paddy soil. Chemosphere. 2023 Nov;340:139860. doi: 10.1016/j.chemosphere.2023.139860. Epub 2023 Aug 21. PMID: 37611773.
- Tian, W, Song P, Zhang H, Duan X, Wei Y, Wang H, Wang S. Microplastic materials in the environment: Problem and strategical solutions. Progress in Materials Science. 2023;132:101035. doi: 10.1016/j.pmatsci.2022.101035.
- Bostan N, Ilyas N, Akhtar N, Mehmood S, Saman RU, Sayyed RZ, Shatid AA, Alfaifi MY, Elbehairi SEI, Pandiaraj S. Toxicity assessment of microplastic (MPs); a threat to the ecosystem. Environ Res. 2023 Oct 1;234:116523. doi: 10.1016/j.envres.2023.116523. Epub 2023 Jul 7. PMID: 37422115.
- Talukdar A, Kundu P, Bhattacharya S, Dutta N. Microplastic contamination in wastewater: Sources, distribution, detection and remediation through physical and chemical-biological methods. Sci Total Environ. 2024 Mar 15;916:170254. doi: 10.1016/j.scitotenv.2024.170254. Epub 2024 Jan 20. PMID: 38253100.
- Jaafarzadeh Haghighi Fard N, Mohammadi MJ, Jahedi F. Effects of nano and microplastics on the reproduction system: In vitro and in vivo studies review. Food Chem Toxicol. 2023 Aug;178:113938. doi: 10.1016/j.fct.2023.113938. Epub 2023 Jul 8. PMID: 37429406.
- Xu R, Cao JW, Lv HL, Geng Y, Guo MY. Polyethylene microplastics induced gut microbiota dysbiosis leading to liver injury via the TLR2/NF-κB/NLRP3 pathway in mice. Sci Total Environ. 2024 Mar 20;917:170518. doi: 10.1016/j.scitotenv.2024.170518. Epub 2024 Jan 28. PMID: 38286276.
- Fard JH, Jahedi F, Dehdarirad H. The possibility of microplastic removal by earthworms and comparing with conventional chemical removal methods (A Global and Deeply Systematic Review). Journal of Polymers and the Environment. 2023;31(12):5050-5064.
- Zhang W, Geng J, Sun M, Jiang C, Lin H, Chen H, Yang Y. Distinct species turnover patterns shaped the richness of antibiotic resistance genes on eight different microplastic polymers. Environmental Research. 2024;259:119562. doi: 10.1016/j.envres.2024.119562.
- Perveen S, Pablos C, Reynolds K, Stanley S, Marugán J. Growth and prevalence of antibiotic-resistant bacteria in microplastic biofilm from wastewater treatment plant effluents. Science of the Total Environment. 2023;856:159024. doi: 10.1016/j.scitotenv.2022.159024.
- Zheng Z, Huang Y, Liu L, Wang L, Tang J. Interaction between microplastic biofilm formation and antibiotics: Effect of microplastic biofilm and its driving mechanisms on antibiotic resistance gene. J Hazard Mater. 2023 Oct 5;459:132099. doi: 10.1016/j.jhazmat.2023.132099. Epub 2023 Jul 21. PMID: 37517232.
- Li N, Zheng N, Pan J, An Q, Li X, Sun S, Chen C, Zhu H, Li Z, Ji Y. Distribution and major driving elements of antibiotic resistance genes in the soil-vegetable system under microplastic stress. Sci Total Environ. 2024 Jan 1;906:167619. doi: 10.1016/j.scitotenv.2023.167619. Epub 2023 Oct 6. PMID: 37806594.
- de Souza Machado AA, Lau CW, Till J, Kloas W, Lehmann A, Becker R, Rillig MC. Impacts of Microplastics on the Soil Biophysical Environment. Environ Sci Technol. 2018 Sep 4;52(17):9656-9665. doi: 10.1021/acs.est.8b02212. Epub 2018 Aug 20. PMID: 30053368; PMCID: PMC6128618.
- He D, Luo Y, Lu S, Liu M, Song Y, Lei L. Microplastics in soils: Analytical methods, pollution characteristics and ecological risks. TrAC Trends in Analytical Chemistry, 2018. 109: p. 163-172.
- Chen H, Wang Y, Sun X, Peng Y, Xiao L. Mixing effect of polylactic acid microplastic and straw residue on soil property and ecological function. Chemosphere. 2020 Mar;243:125271. doi: 10.1016/j.chemosphere.2019.125271. Epub 2019 Nov 2. PMID: 31760289.
- Huang Y, Zhao Y, Wang J, Zhang M, Jia W, Qin X. LDPE microplastic films alter microbial community composition and enzymatic activities in soil. Environ Pollut. 2019 Nov;254(Pt A):112983. doi: 10.1016/j.envpol.2019.112983. Epub 2019 Jul 30. PMID: 31394342.
- Sun M, Ye M, Jiao W, Feng Y, Yu P, Liu M, Jiao J, He X, Liu K, Zhao Y, Wu J, Jiang X, Hu F. Changes in tetracycline partitioning and bacteria/phage-comediated ARGs in microplastic-contaminated greenhouse soil facilitated by sophorolipid. Journal of Hazardous Materials, 2018. 345: p. 131-139.
- Liu J, Lv M, Sun A, Ding J, Wang Y, Chang X, Chen L. Exposure to microplastics reduces the bioaccumulation of sulfamethoxazole but enhances its effects on gut microbiota and the antibiotic resistome of mice. Chemosphere. 2022 May;294:133810. doi: 10.1016/j.chemosphere.2022.133810. Epub 2022 Jan 29. PMID: 35104539.
- Gao B, Shi X, Li S, Xu W, Gao N, Shan J, Shen W. Size-dependent effects of polystyrene microplastics on gut metagenome and antibiotic resistance in C57BL/6 mice. Ecotoxicol Environ Saf. 2023 Apr 1;254:114737. doi: 10.1016/j.ecoenv.2023.114737. Epub 2023 Mar 10. PMID: 36950986.
- Xu Z, Wu X, Zhang J, Cheng P, Xu Z, Sun W, Zhong Y, Wang Y, Yu G, Liu H. Microplastics existence intensified bloom of antibiotic resistance in livestock feces transformed by black soldier fly. Environ Pollut. 2023 Jan 15;317:120845. doi: 10.1016/j.envpol.2022.120845. Epub 2022 Dec 7. PMID: 36496063.
- Chen X, Wang X, Huang Y, Zhu Z, Li T, Cai Z, Li M, Gong H, Yan M. Combined effects of microplastics and antibiotic-resistant bacteria on Daphnia magna growth and expression of functional genes. Science of the Total Environment. 2023;905:166880. doi: 10.1016/j.scitotenv.2023.166880.
- Chan SY, Liu SY, Wu R, Wei W, Fang JK, Chua SL. Simultaneous Dissemination of Nanoplastics and Antibiotic Resistance by Nematode Couriers. Environ Sci Technol. 2023 Jun 13;57(23):8719-8727. doi: 10.1021/acs.est.2c07129. Epub 2023 Jun 2. PMID: 37267481.
- Liu B, Yu D, Ge C, Luo X, Du L, Zhang X, Hui C. Combined effects of microplastics and chlortetracycline on the intestinal barrier, gut microbiota, and antibiotic resistome of Muscovy ducks (Cairina moschata). Sci Total Environ. 2023 Aug 20;887:164050. doi: 10.1016/j.scitotenv.2023.164050. Epub 2023 May 11. PMID: 37178843.
- Zhang P, Lu G, Sun Y, Yan Z, Dang T, Liu J. Metagenomic analysis explores the interaction of aged microplastics and roxithromycin on gut microbiota and antibiotic resistance genes of Carassius auratus. J Hazard Mater. 2022 Mar 5;425:127773. doi: 10.1016/j.jhazmat.2021.127773. Epub 2021 Nov 15. PMID: 34802820.
- Xu G, Yu Y. Polystyrene microplastics impact the occurrence of antibiotic resistance genes in earthworms by size-dependent toxic effects. J Hazard Mater. 2021 Aug 15;416:125847. doi: 10.1016/j.jhazmat.2021.125847. Epub 2021 Apr 20. PMID: 34492800.
- Ma J, Sheng GD, Chen QL, O'Connor P. Do combined nanoscale polystyrene and tetracycline impact on the incidence of resistance genes and microbial community disturbance in Enchytraeus crypticus? J Hazard Mater. 2020 Apr 5;387:122012. doi: 10.1016/j.jhazmat.2019.122012. Epub 2020 Jan 3. PMID: 31927355.
- McCallum G, Tropini C. The gut microbiota and its biogeography. Nat Rev Microbiol. 2024 Feb;22(2):105-118. doi: 10.1038/s41579-023-00969-0. Epub 2023 Sep 22. PMID: 37740073.
- Xue YX, Huang LJ, Wang HY, Peng JJ, Jin MK, Hu SL, Li HB, Xue XM, Zhu YG. Interaction of tetracycline and copper co-intake in inducing antibiotic resistance genes and potential pathogens in mouse gut. Environ Int. 2024 Apr;186:108594. doi: 10.1016/j.envint.2024.108594. Epub 2024 Mar 21. PMID: 38527398.
- Fu Y, Jia F, Su J, Xu X, Zhang Y, Li X, Jiang X, Schäffer A, Virta M, Tiedje JM, Wang F. Co-occurrence patterns of gut microbiome, antibiotic resistome and the perturbation of dietary uptake in captive giant pandas. J Hazard Mater. 2024 Jun 5;471:134252. doi: 10.1016/j.jhazmat.2024.134252. Epub 2024 Apr 12. PMID: 38657507.
- Jensen O, Trujillo E, Hanson L, Ost KS. Controlling Candida: immune regulation of commensal fungi in the gut. Infect Immun. 2024 Apr 22:e0051623. doi: 10.1128/iai.00516-23. Epub ahead of print. PMID: 38647290.
- Zhang J, Ma W, Li Y, Zhong D, Zhou Z, Ma J. The resistance change and stress response mechanisms of chlorine-resistant bacteria under microplastic stress in drinking water distribution system. Environ Pollut. 2024 Sep 1;356:124331. doi: 10.1016/j.envpol.2024.124331. Epub 2024 Jun 5. PMID: 38848962.
- Li H, Lian Y, Li S, Yang M, Xie Q, Qiu L, Liu H, Long Y, Hu L, Fang C. The stress response of tetracycline resistance genes and bacterial communities under the existence of microplastics in typical leachate biological treatment system. J Environ Manage. 2024 Aug;366:121865. doi: 10.1016/j.jenvman.2024.121865. Epub 2024 Jul 16. PMID: 39018858.
- Khare T, Mathur V, Kumar V. Agro-ecological Microplastics enriching the antibiotic resistance in aquatic environment. Current Opinion in Environmental Science & Health, 2024: p. 100534. doi: 10.1016/j.coesh.2024.100534.
- Okaiyeto SA, Sutar PP, Chen C, Ni JB, Wang J, Mujumdar AS, Zhang JS, Xu MQ, Fang XM, Zhang C, Xiao HW. Antibiotic resistant bacteria in food systems: Current status, resistance mechanisms, and mitigation strategies. Agriculture Communications. 2024;100027. doi: 10.1016/j.agrcom.2024.100027.
- Wei L, Su Z, Yue Q, Huang X, Wei M, Wang J. Microplastics, heavy metals, antibiotics, and antibiotic resistance genes in recirculating aquaculture systems. TrAC Trends in Analytical Chemistry. 2024;117564. doi: 10.1016/j.trac.2024.117564.
- Gao B, Shi X, Li S, Xu W, Gao N, Shan J, Shen W. Size-dependent effects of polystyrene microplastics on gut metagenome and antibiotic resistance in C57BL/6 mice. Ecotoxicol Environ Saf. 2023 Apr 1;254:114737. doi: 10.1016/j.ecoenv.2023.114737. Epub 2023 Mar 10. PMID: 36950986.
- Zhang Z, Chen W, Chan H, Peng J, Zhu P, Li J, Jiang X, Zhang Z, Wang Y, Tan Z, Peng Y, Zhang S, Lin K, Yung KK. Polystyrene microplastics induce size-dependent multi-organ damage in mice: Insights into gut microbiota and fecal metabolites. J Hazard Mater. 2024 Jan 5;461:132503. doi: 10.1016/j.jhazmat.2023.132503. Epub 2023 Sep 6. PMID: 37717443.
- Xiao S, Zhang Y, Wu Y, Li J, Dai W, Pang K, Liu Y, Wu R. Bacterial community succession and the enrichment of antibiotic resistance genes on microplastics in an oyster farm. Mar Pollut Bull. 2023 Sep;194(Pt A):115402. doi: 10.1016/j.marpolbul.2023.115402. Epub 2023 Aug 21. PMID: 37611336.
- Csongradi C, du Plessis J, Aucamp ME, Gerber M. Topical delivery of roxithromycin solid-state forms entrapped in vesicles. Eur J Pharm Biopharm. 2017 May;114:96-107. doi: 10.1016/j.ejpb.2017.01.006. Epub 2017 Jan 22. PMID: 28119103.
- Zhang C, Li X, Liu L, Gao L, Ou S, Luo J, Peng X. Roxithromycin regulates intestinal microbiota and alters colonic epithelial gene expression. Appl Microbiol Biotechnol. 2018 Nov;102(21):9303-9316. doi: 10.1007/s00253-018-9257-1. Epub 2018 Aug 2. PMID: 30073395.
- Jiang X, Wang X, Kah M, Li M. Environmentally relevant concentrations of microplastics from agricultural mulch and cadmium negatively impact earthworms by triggering neurotoxicity and disrupting homeostasis. J Hazard Mater. 2024 Sep 5;476:135158. doi: 10.1016/j.jhazmat.2024.135158. Epub 2024 Jul 10. PMID: 39002475.
- Liu B, Yu D, Ge C, Luo X, Du L, Zhang X, Hui C. Combined effects of microplastics and chlortetracycline on the intestinal barrier, gut microbiota, and antibiotic resistome of Muscovy ducks (Cairina moschata). Sci Total Environ. 2023 Aug 20;887:164050. doi: 10.1016/j.scitotenv.2023.164050. Epub 2023 May 11. PMID: 37178843.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.