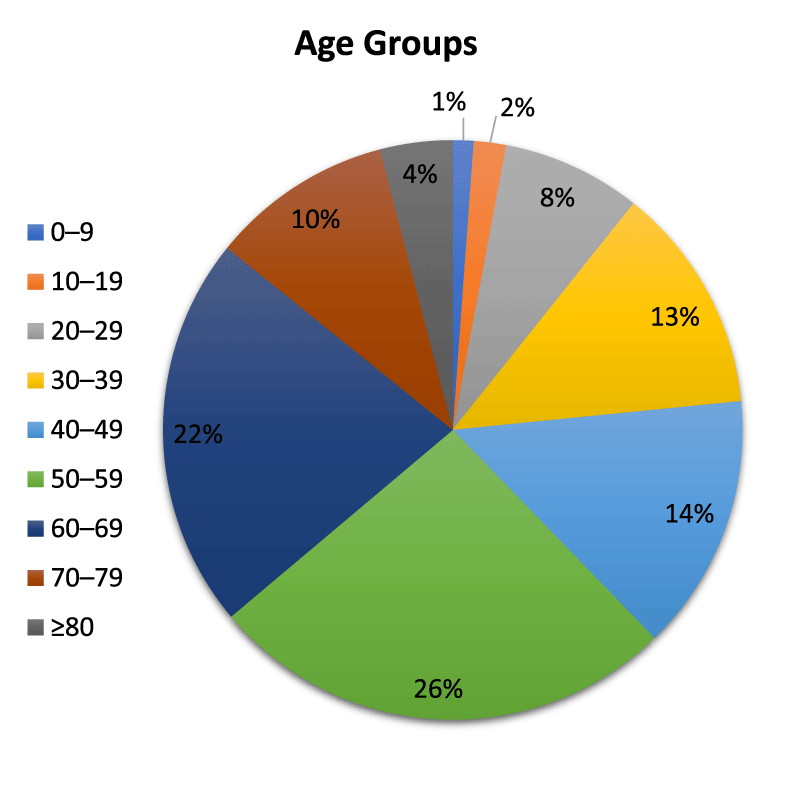> Biology. 2020 November 12;1(7):303-310. doi: 10.37871/jbres1159.
-
Subject area(s):
- Infectious Diseases
- Virology
- Genetics
Sex and Age Differences in Telomere Length and Susceptibility to COVID-19
Manar Ahmed Kamal1*, Kareem Reda Alamiry2 and Mahmoud Zaki2
2Faculty of Pharmacy, Suez Canal University, Ismailia, Egypt
- Sex variations
- Age variations
- Shortened telomere length
- Respiratory viral infections
- COVID-19
Background: Telomeres are the ends of a chromosome and play a fundamental role as vanguards contra the chromosomal decay. Due to the inability of DNA polymerase to replicate chromosomal ends, a reduction in telomeres length happens after each cell division. The existence of shorter telomeres in older people is related to diminish immune functions. Viral infections able to stimulate remodeling of cells, stress responses, and telomere shortening. Moreover, telomere shortening can be caused by extrinsic environmental variables which induce oxidative stress under conditions of inflammation.
Aim: To identify the correlation between telomere shortening and susceptibility to Novel Coronavirus Disease 2019 (COVID-19). In addition to clarifying changes in telomere length according to the viral infection, the effect of sex and age differences in telomere length in confirmed positive COVID-19 cases are also reviewed.
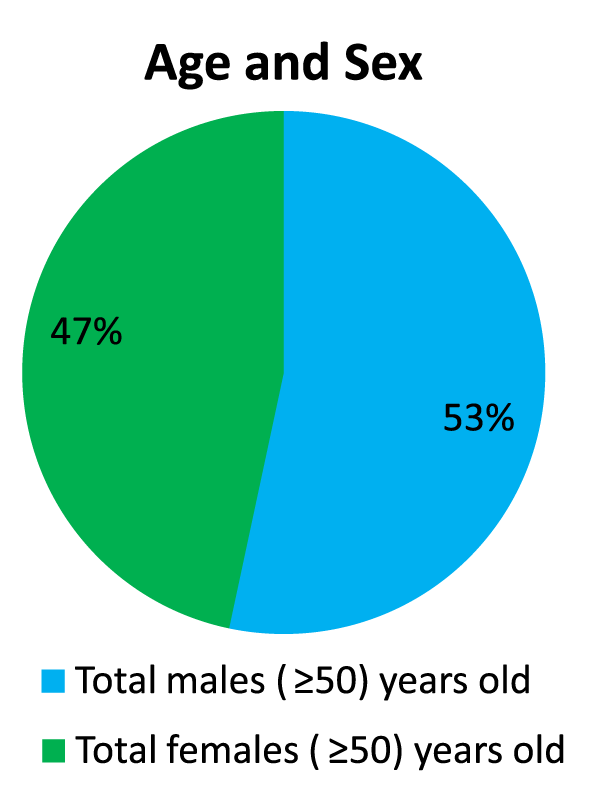
Conclusion: There is a correlation between telomere length and COVID-19 infection with higher susceptibly of elderly patients and males due to shortening in their telomere length. Approximately 53% of (111,428) infected cases (≥ 50) years old are males, and 47% of (111,428) infected cases (≥ 50) years old are females.
Akt: Protein Kinase B; BatCov: Beta Coronavirus; CD8CD28: Cluster of Differentiation 8, Cluster of Differentiation 28; COVID-19: Novel Coronavirus Disease 2019; CRP: C-Reactive Protein; DNA: Deoxyribonucleic Acid; DDR: DNA Damage Response; IL: Interleukins; IL-1β: Interleukin1 Beta; IL-6: Interleukin-6; NF-Κb: Nuclear Factor-Κb; NF-B: Family of Transcription Factors; PI3k: Phosphoinositide 3-Kinase; RNA: Ribonucleic Acid; ROS: Receptive Oxygen Species; SARS-Cov: Severe Acute Respiratory Syndrome Coronavirus; STAT1/STAT3: Signal Transducer and Activator of Transcription 1/3; TERRA: Telomere Repeat-Containing RNA Termed; Tert: Telomerase Reverse Transcriptase; Terc: Telomerase RNA Component; TNF-a: Tumor Necrosis Factor a; TRF: Telomere Restriction Fragment; WHO: World Health Organization
Telomere is a non-coding part of DNA sequences at the end of each chromosome. Mammalian telomeres contain 5-8 nucleotides with reduplicated sequences of TTAAGGG. Their functions include recognizing the end of chromosomes, avoiding the end of the chromosome from being adhesive, protecting the chromosomal ends from inaccurate connection and corruption, appropriate chromosome site in a nucleus, and synthesizing the end of chromosomes in DNA replication [1]. Telomeres are a simple DNA sequenc composed of a large number of repeats called (TTAGGG) in vertebrates mainly humans. Telomeres are surrounded by a protein component called shelterin or telosome. Shelterin or telosome has an important function in the regulation of telomere length and chromosomes’ protection [2,3]. Telomeres and their associated proteins can protect every chromosome end against terminal DNA degeneration, chromosomal recombination, and fusion [4]. Moreover, accelerating telomere shortening is induced by inflammation, exposure to infectious pathogens, and oxidative stress, which damage telomeres and diminish their repair mechanisms. Thus, people who were exposed to viral infections have shorter telomeres. Infectious diseases, for example, decrease telomere length and, hence, increasing the susceptibility to infection [5].
Coronaviruses are a large family of viruses that affect the respiratory tract of animals and humans [6]. They can be zoonotic, which means they can be transmitted from animals to humans. Common signs and symptoms of the disease include respiratory indications, fever, cough, weakness of breath, and difficulty breathing. In other severe cases, the disease can lead to pneumonia, Severe Acute Respiratory Syndrome (SARS), kidney failure, and even death [7]. Furthermore, coronaviruses are enveloped positive-stranded RNA viruses and characterized by club-like spikes that extend from their surface, an abnormally huge RNA genome, and a unique replication technique, which belongs to the family “Coronaviridae” and the order “Nidovirales” [6]. According to WHO, COVID-19 first appeared in December 2019 in Wuhan, China. Bioinformatic analyses revealed that COVID-19 has characteristics that are typical to the coronavirus family and belong to the ß-coronavirus 2B lineage. On the other hand, the genome sequence of the COVID-19 virus and other available genomes of ß-coronavirus showing the closest relationship with the bat SARS-like coronavirus strain BatCov ratg13, identified 96% [8].
The primary purpose of this review is to identify the correlat5on between telomere shortening and susceptibility to COVID-19 infection, by investigating the changes in telomere length according to variations in age, sex in confirmed cases with COVID-19 infection, and the relation between the inflammatory markers and telomere length during respiratory viral infection.
Telomere length changes according to age and sex
Many studies have shown that age, gender, current health status, and mortality may affect telomere length that varies within the same group [9]. Telomere shortening is considered as an important biomarker (biological thermometer) [10]. Since it might be related to the replication problem phenomena, which says that DNA polymerases are not able to replicate in the linear chromosome that leads the telomeres to shorten after each cell division, consequently, this may be followed by cell death [11].
Telomere and sex
There is an association between sex and telomere length [12]. Previous studies clarified the association between sex and telomere length especially in leukocyte telomeres which are longer in women than men [13]. Several hypotheses have been postulated to explain this association, for example, due to the action of estrogen [14]. An estrogen-responsive element is present in Telomerase Reverse Transcriptase (hTERT), subsequently, estrogen might stimulate telomerase to add telomere repeats to the ends of chromosomes [15]. Telomeres are particularly sensitive to oxidative stress [16]. Women produce fewer reactive oxygen species than men due to high level of estrogen in female than males [15]. So, it has been suggested that women might also metabolize reactive oxygen species better because of the antioxidant properties of estrogen [15]. At birth, one study found that there was a difference in telomere length between the sexes with female newborns have longer telomeres than males [17]. An animal study on mice (males and females) indicated that the repeated experimental inoculation resulted in systemic infection and disease with higher susceptibility in males than females [5]. Moreover, they examined in the previous study the changes in telomeres in WBCs over nine months and five consecutive infections and found that the experimentally infected males showed significantly greater telomere attrition compared to female infected controls; unlike males, the infection did not affect the telomeres of the females, this sex-difference in telomere dynamics could be due to the higher susceptibility of males leading to greater infections than females [5]. There are concerns about the robustness of telomere length as a biomarker of aging [18,19]. In conclusion, telomere length is longer on average in females than males and the strength of these associations varies by the measurement methods but not by age group [13].
Telomere and age
A steady decline in telomere length at a relatively constant rate with advancing age has been demonstrated in various cross-sectional studies. This decrease in length is further accelerated with the onset of several diseases that develop with aging [20]. Various studies on human models have attempted to correlate between telomere length with age; a study accentuated that individuals with long telomeres lived longer than their counterparts with the same age and had shorter telomeres [21]. However, the presence of short telomeres among young people [22,23] might result from many factors as genetics modifications, [21,22] chronic psychological stress [24,25], older paternal age at conception [26], poor health behaviors, and oxidative stress. The reason behind these discrepancies is still unclear and may have several causes. A conclusion from previous various studies is that telomere length correlates with somatic cell growth till puberty and with cellular senescence after puberty [27].
Characteristics of COVID-19 infection
At the end of December 2019 in Wuhan, China, Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) appeared with confirmed human cases [28,29]. It spreads rapidly worldwide causing a pandemic of Coronavirus Disease (COVID-19). Symptoms range from fever and breathing difficulty to pneumonia and death [30]. Patients have COVID-19 show increased leukocyte numbers, and elevated levels of plasma proinflammatory cytokines in blood. Cytokines and chemokines which noted in patients with COVID-19 infection include [ IL1-β, IL1RA, IL7, IL8, IL9, IL10, basic FGF2, GCSF, GMCSF, IFNΓ, IP10, MCP1, MIP1α, MIP1β, PDGFB, TNFa, and VEGFA ]. Some severe cases show high levels of pro-inflammatory cytokines as [IL2, IL7, IL10, GCSF, IP10, MCP1, MIP1α, and TNFα] that are reasoned to increase the severity of infection.
COVID-19 variations according to age and sex
rom the beginning of the outbreak of SARS-CoV-2 in December 2019 in Wuhan, China, many press articles discussed the correlation between age and gender and susceptibility to COVID-19. So, to prepare this review, we use a published data about all confirmed positive cases included the age and sex as showed in (Table 1). The data is from nine countries and cities Mainland China [31], South Korea [32], Japan [33], Philippines [34], Finland [35,36], California, USA [37], Italy [38], Czechia [39], and Estonia [40].
Table 1: Show all confirmed positive cases in Mainland China, South Korea, Japan, Philippines, Finland, California, Italy, Czechia and Estonia. |
|||||||||
| Characteristics | Country/City | ||||||||
| Mainland China | South Korea | Japan | Philippines | Finland | California | Italy | Czechia | Estonia | |
| Till date | February 11, 2020 | April 1, 2020 | March 22, 2020 |
April 2, 2020 |
April 2, 2020 | March 25, 2020 |
April 1, 2020 | July 13, 2020 | July 13, 2020 |
| Ages (year) | |||||||||
| 0 - 9 | 416 | 116 | 12 | 6 | 21 | 37 | 1,437 | 840 | 30 |
| 10 - 19 | 549 | 519 | 4 | 15 | 62 | 1159 | 78 | ||
| 20 - 29 | 3,619 | 2,682 | 84 | 206 | 228 | 1,505 | 26,489 | 1852 | 193 |
| 30 - 39 | 7,600 | 1,027 | 92 | 325 | 272 | 2219 | 263 | ||
| 40 - 49 | 8,571 | 1,323 | 142 | 329 | 299 | 2631 | 348 | ||
| 50 - 59 | 10,008 | 1,865 | 187 | 496 | 324 | 1,442 | 74,433 | 1552 | 426 |
| 60 - 69 | 8,583 | 1,245 | 171 | 543 | 160 | 1026 | 253 |
||
| 70 - 79 | 3,918 | 658 | 138 | 290 | 102 | 671 | 252 |
||
| ≥80 | 1,408 | 452 | 93 | 102 | 50 | 409 | 171 |
||
| Unknown | - | - | - | 323 | - | 22 | - | 815 | - |
| Sex | |||||||||
| Male | 22,981 | 3,946 | 520 | 1425 | 778 | 1,665 | 56,673 | 6497 | 685 |
| Female | 21,691 | 5,941 | 403 | 885 | 740 | 1,309 | 45,996 | 5862 | 812 |
| Unknown | - | - | - | 323 | - | 32 | - | 815 | 517 |
| Total | 44,672 | 9,887 | 923 | 2,633 | 1,518 | 3,006 | 102,669 | 13,174 | 2,014 |
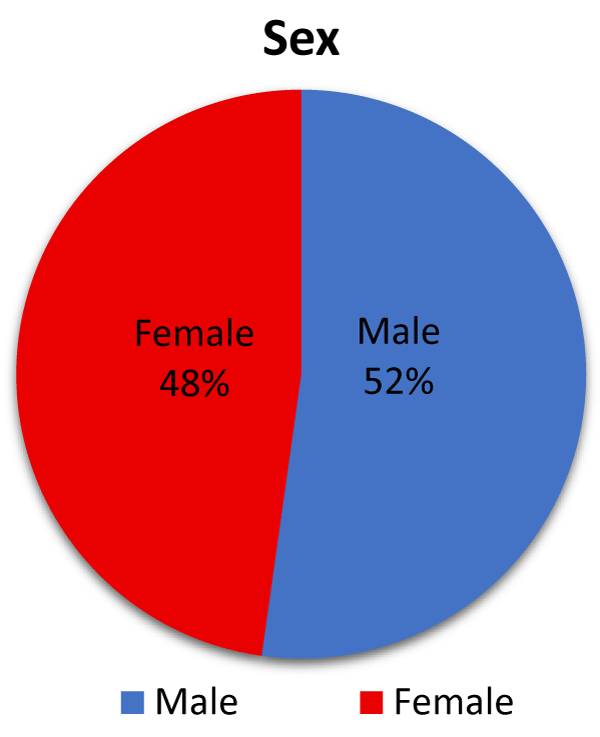
The age groups are classified by using 10-years intervals: 0-9, 10-19, 20-29…., 70-79, and ≥ 80 years old. In addition to unknown cases, so the total of 10 age groups are designated. The sex groups are classified into male, female, and unknown.
We analyzed the known data according to the age groups as in figure 1, according to sex groups as in figure 2 and according to the age and sex groups together as in figure 3.
Telomere length in response to viral infection
Telomeres are repetitive elements at the ends of linear chromosomes that are essential for maintaining genomic stability [41,42]. Telomere repeats can be transcribed to make a non-coding RNA, Telomere Repeat-Containing RNA Termed (TERRA), that has been identified in numerous organisms, and contributes structurally and functionally to telomere regulation [43,44]. TERRA can be induced in response to various types of stress [45,46], including DNA damage and viral infection.
Telomere length changes during DNA viral infection
DNA viral infections can activate a DNA Damage Response (DDR) signaling pathway similar to the chromosome double-strand break or an uncapped telomere [47]. In the same way as the telomere, viruses have many mechanisms to avoid the DDR, including the assembly of the protective complexes of viral DNA ends that can actively inhibit the cellular DDR [48]. Several acute nuclear DNA viruses and one RNA virus were found to increase TERRA expression [49]. Telomeres are responsive to various stress response pathways, including viral infection, reactive oxygen species [46], and DNA damage signaling [50]. Consequently, TERRA transcription can also be induced by p53 activation, thus, binding sites for p53 have been identified in human sub-telomeres by Chip-Seq [51]. While TERRA can be induced to high levels in response to various stresses including DNA damage and viral infection, its potential function during viral infection remains unknown. In an experimental study, an association between shorter telomeres and upper respiratory viral infection was found [52]. CD8CD28 was the only cell population in which shorter telomeres were related to a higher risk of clinical illness like a viral infection, where the association between CD8CD28-telomere length and infection increased with age [53].
Telomere length changes during RNA viral infection
Viruses with single-stranded RNA, enveloped as (Togaviridae, Flaviviridae, and Coronaviridae) or non-enveloped as (Astroviridae, Caliciviridae, and Picornaviridae) [54] can attack human cells at any time. Enveloped viruses (like coronavirus) the envelope fuses with the endosomal membrane, releasing viral genome into the host cytosol [55]. Although viruses can replicate in multiple types of cells, the pathological outcome manifests in only one or a few specific cell/tissue types [56]. Coronavirus infections usually start benign causing self-limiting mild flu-like symptoms. Severe acute respiratory syndrome Coronavirus (SARS-CoV), which jumped the species barrier through gaining slight genome mutations, are severe human pathogens [57]. SARS-CoV mainly infects lung cells stimulating an often-fatal inflammatory response, which is clinically called Acute Respiratory Distress Syndrome (ARDS) that begins with severe hypoxia, pulmonary edema progressing to systemic inflammation, and failure of multiple organs, culminating in a high rate of mortality [58-61]. Shorter Peripheral Blood Leukocyte (PBL) Telomere Length (TL) is associated with higher mortality among patients with ARDS and more severe lung injury. Highly significant positive associations between telomere length and lung function, shorter telomeres were seen in patients with lung function diseases compared with healthy patients [62].
The relation between RNA virus and TLR signaling
Pattern Recognition Receptors (PRRs) are the proteins, communicated by an assortment of cells, which are dependable for detecting the presence of microbial attack. The individuals of these receptor families can be recognized by ligand specificity, cellular localization, and actuation of unique, but meeting, downstream signaling pathways [63]. PRRs are known to be activated by invasions of RNA viral infection. Toll-Like Receptors (TLRs) are the foremost broadly studied family of PRRs so far, and they are of considerable significance within the initiation of an antiviral reaction upon disease. The human TLR multigene family comprises 10 individuals, of which TLR 2, 3, 4, 7, and 8 are thought to be of importance within the recognition of basic components of RNA infections, counting viral double-stranded RNA (dsRNA), single-stranded RNA (ssRNA), and surface glycoproteins [64].
Toll-Like Receptors (TLR) 7 and 8 are intracellular sensors found in endosomes that recognize single-stranded RNA. Both sorts of receptors induce the expression of pro-inflammatory cytokines and sort I IFN reaction upon RNA viral infection sensing [65,66]. TLR7 and TLR8 activated differential signaling cascades that contributed to the particular phenotypes observed. It has been found that FOSL1 restrained sort 1 cytokines after TLR7 signaling and revealed the part of TLR7-dependent Ca2+ flux in modulating sort I IFN reactions. It is illustrated that although both TLR7 and TLR8 recognize single-stranded RNA, they activated diverse signaling pathways in human monocytes that contribute to particular phenotypes during RNA infection disease. In addition, it is characterized by individual targets inside these pathways that advanced particular T helper and antiviral reactions [67]. TLR7 activation induces a Th17-polarizing phenotype whereas TLR8 incitement actuates a Th1-polarizing phenotype on human CD14+ monocytes. The diverse effects of TLR7 and TLR8 inhibition on pro-inflammatory cytokine expression and sort I IFN responses led to hypothesize that TLR-7 and TLR-8 incitement initiate distinctive functional phenotypes on CD14+ monocytes. To test this speculation, Imiquimod (IMQ) has been chosen as a human TLR7-specific ligand and ssRNA40-LyoVec (ssRNA40) as a human TLR8-specific ligand and stimulated ex vivo confined CD14+ monocytes with them to examine the expression of pro-inflammatory cytokines [68].
Telomere length and telomerase activity
An important type of proteins related to telomere is called Telomerase Reverse Transcriptase (TERT), which is encoded by the “Tert” gene that specifically recognizes the 3'-OH group at the end of G-rich strand overhang. This arrangement is decided by the action of telomerase, which lengthens terminal regions of eukaryotic telomeric DNA by RNA-templated addition of the repeated DNA arrangement. Complete replication of telomeric DNA requires telomerase [41]. This polymerization activity was then appeared to happen on natural telomeres in vivo which initially called ‘telomere terminal transferase [69]. Telomerase could be a specialized cellular RT. It is a Ribonucleoprotein (RNP) complex, it synthesizes one strand of the telomeric DNA - namely, the strand running 5 to 3 towards the distal end of the chromosome—by replicating a short format arrangement inside its natural RNA moiety. This activity extends the 3 terminal, single-stranded overhang found at the closes of telomeric DNA [70]. Several Studies reported the link between oxidative stress and telomere shortening. Telomerase neutralizes telomere shortening and cellular senescence in germ, stem, and cancer cells by including repetitive DNA arrangements to the ends of chromosomes [71].
C-reactive protein
C-Reactive Protein (CRP) is a pentameric protein circulating in blood plasma which is a marker of inflammation [72], besides, being a member of the pentraxin family of proteins [73]. CRP is synthesized by the liver in reaction to components discharged by macrophages and fat cells (adipocytes) [74]. There are various causes of a raised CRP. These include acute and chronic conditions, and these can be infectious or non-infectious in etiology [75]. However, markedly elevated levels of CRP are most often associated with infectious causes like respiratory viral Infection. CRP reaches an abnormal level in patients tested positive to respiratory viral infection. An obvious increase in CRP level was noted during specific respiratory viral infections with more prominent increases observed in elderly patients [75]. CRP inversely correlates with leukocyte telomere length [71]. Hence, during respiratory viral infections, leukocyte telomere length becomes shorter.
Pro-inflammatory cytokines
Inflammation is one of the complex biological responses by the immune system to neutralize the damages caused either by injury or microbial infection. Pro-inflammatory cytokines are produced mostly by the activated macrophages and are involved in the up-regulation of inflammatory reactions such as interleukins (IL-1β, IL-6) and TNF-α [76].
Interleukins (ILs) are a type of cytokines that play essential roles in the activation and differentiation of immune cells. They also have pro-inflammatory and anti-inflammatory properties [77]. Interleukins consist of a large group of proteins that can get many reactions in cells and tissues by binding to high-affinity receptors on cell surfaces [78]. One of ILs is called Interleukin-6 (IL-6) that is synthesized by T and B lymphocytes, fibroblasts, and macrophages [79]. IL-6 is a pleiotropic cytokine produced in response to tissue damage and infections. IL-6 is increasing at the site of inflammation and plays a key role in the acute phase response [80]. Shorter telomeres are associated with higher IL-6 [81]. Increasing the production of cytokines has been shown to adversely affect telomerase activity and telomere length [82]. An experimental study showed that STAT3 was synergistically activated by IL-6 and TNF-α. STAT3, STAT1, and NF-ΚB formed triplet complexes with IL-6 and TNF-α stimulation, thereby increasing telomerase activity by binding to hTERT promoter more tightly [83]. Cell-level invasion assay revealed that cytokine treatments contributed to the cell invasiveness. Combined treatment of IL-6 and TNF-α synergistically phosphorylated transcription factors STAT3 [84]. STAT3, STAT1, and NF-κb physically interacted upon the cytokine stimulation. STAT3 was bound to the promoter region of hTERT. IL-6 and TNF-α stimulation further enhanced STAT3 binding affinity and increase the activity of telomerase [85]. The immune system dysfunction/accelerated maturing observed in chronic conditions are associated with telomeres and telomerase activity. Numerous analysts documented relationships between lower telomerase activity and/or shorter telomeres in immune system cells and raised cytokines in blood serum from patients with a chronic disorder.
Reactive oxygen species
Several Studies reported the link between oxidative stress and telomere shortening. Telomerase neutralizes telomere shortening and cellular senescence in germ, stem, and cancer cells by including repetitive DNA arrangements to the ends of chromosomes. Telomeres are susceptible to damage by Reactive Oxygen Species (ROS) [86]. Based on in vitro studies, ROS have been proposed to inhibit telomerase activity [87,88].
During the last years, many studies have continually provided evidence that links shortened telomeres with common respiratory viral diseases, infection risk, and longevity. Telomeres were considered as a potential biomarker that could evaluate the susceptibility to a specific pathogen as SARS-COV-2. The previous studies also provided an association between sex and telomere length, especially in leukocyte. Many studies on adults have found that the female telomere length is longer than the male ones, and one study explained that female newborns have longer telomeres than males. Males were somewhat more susceptible to infection than females this may due to greater telomere attrition in males than females.
According to the reviewed data, we found that: (1) 52% of total infected cases are males, and 48% of total infected cases are females; (2) 62% of total infected cases are (≥ 50) years old; (3) approximately 53% of (111,428) infected cases (≥ 50) years old are males, and 47% of (111,428) infected cases (≥ 50) years old are females. The previous results approve that elder males are the most susceptible to COVID-19 infection.
Patients have COVID-19 show an increase in leukocyte numbers, and increased levels of plasma pro-inflammatory cytokines as IL-1β, IL-6, and TNF-α in blood and an elevation in the production of cytokines has been shown to adversely affect telomerase activity and telomere length. For example, shorter telomeres are associated with higher IL-6. CRP rises to an abnormal level in patients tested positive to a respiratory viral infection and CRP inversely correlated with leukocyte telomere length. Unfortunately, there is a lack of data that discussed the relationship between telomerase activity and CRP. All markers of inflammation provide the relation between telomere length and COVID-19 infection.
We are very pleased to direct special thanks to people who kept checking our progress and directed the review process, Dr. Mennatallah Abdelkader, Department of Microbiology and Immunology Faculty of Pharmacy Suez Canal University; Ahmed Ibrahim Sorour, Pharmacy graduate, Damanhour University; Dr. Marwa El-Araby, Medical Microbiology & Immunology Department, Faculty of Medicine, Benha University; Dr. Ahmed Ibrahim, Pharmacognosy Department, Faculty of Pharmacy Suez Canal University.
- Mandeh M, Omidi M, Maali Amiri R. Telomere: Characteristics and Functions. Genet 3rd Millenn. 2009;7(1):1589-1596 https://bit.ly/3pgMzN5.
- Artandi SE. Telomeres, telomerase, and human disease. N Engl J Med. 2006 Sep 21;355(12):1195-7. doi: 10.1056/NEJMp068187. PMID: 16990382.
- Blasco MA. Telomere length, stem cells and aging. Nat Chem Biol. 2007 Oct;3(10):640-9. doi: 10.1038/nchembio.2007.38. PMID: 17876321.
- Hiyama E, Hiyama K. Telomere and telomerase in stem cells. Br J Cancer. 2007 Apr 10;96(7):1020-4. doi: 10.1038/sj.bjc.6603671. Epub 2007 Mar 13. PMID: 17353922; PMCID: PMC2360127.
- Ilmonen P, Kotrschal A, Penn DJ. Telomere attrition due to infection. PLoS One. 2008 May 14;3(5):e2143. doi: 10.1371/journal.pone.0002143. PMID: 18478110; PMCID: PMC2366059.
- Douglas D. Richman, Richard J. Whitley FGH. Clinicalvirology - Fourth Edition [Internet]. ASM Press. Elsevier; 2017. 474-478 p. https://bit.ly/32AKP7x
- Ye ZW, Yuan S, Yuen KS, Fung SY, Chan CP, Jin DY. Zoonotic origins of human coronaviruses. Int J Biol Sci. 2020 Mar 15;16(10):1686-1697. doi: 10.7150/ijbs.45472. PMID: 32226286; PMCID: PMC7098031.
- WHO. Report of the WHO-China Joint Mission on Coronavirus Disease 2019 (COVID-19). Who [Internet]. 2020 Feb:16–24. https://bit.ly/3loqAkM
- Das B, Saini D, Seshadri M. Telomere length in human adults and high level natural background radiation. PLoS One. 2009 Dec 23;4(12):e8440. doi: 10.1371/journal.pone.0008440. PMID: 20037654; PMCID: PMC2793520.
- Kudryashova KS, Burka K, Kulaga AY, Vorobyeva NS, Kennedy BK. Aging Biomarkers: From Functional Tests to Multi-Omics Approaches. Proteomics. 2020 Mar;20(5-6):e1900408. doi: 10.1002/pmic.201900408. Epub 2020 Mar 11. PMID: 32084299.
- Cesare AJ, Reddel RR. Alternative lengthening of telomeres: models, mechanisms and implications. Nat Rev Genet. 2010 May;11(5):319-30. doi: 10.1038/nrg2763. Epub 2010 Mar 30. PMID: 20351727.
- Okuda K, Bardeguez A, Gardner JP, Rodriguez P, Ganesh V, Kimura M, Skurnick J, Awad G, Aviv A. Telomere length in the newborn. Pediatr Res. 2002 Sep;52(3):377-81. doi: 10.1203/00006450-200209000-00012. PMID: 12193671.
- Gardner M, Bann D, Wiley L, Cooper R, Hardy R, Nitsch D, Martin-Ruiz C, Shiels P, Sayer AA, Barbieri M, Bekaert S, Bischoff C, Brooks-Wilson A, Chen W, Cooper C, Christensen K, De Meyer T, Deary I, Der G, Diez Roux A, Fitzpatrick A, Hajat A, Halaschek-Wiener J, Harris S, Hunt SC, Jagger C, Jeon HS, Kaplan R, Kimura M, Lansdorp P, Li C, Maeda T, Mangino M, Nawrot TS, Nilsson P, Nordfjall K, Paolisso G, Ren F, Riabowol K, Robertson T, Roos G, Staessen JA, Spector T, Tang N, Unryn B, van der Harst P, Woo J, Xing C, Yadegarfar ME, Park JY, Young N, Kuh D, von Zglinicki T, Ben-Shlomo Y; Halcyon study team. Gender and telomere length: systematic review and meta-analysis. Exp Gerontol. 2014 Mar;51:15-27. doi: 10.1016/j.exger.2013.12.004. Epub 2013 Dec 21. PMID: 24365661; PMCID: PMC4523138.
- Mayer S, Brüderlein S, Perner S, Waibel I, Holdenried A, Ciloglu N, Hasel C, Mattfeldt T, Nielsen KV, Möller P. Sex-specific telomere length profiles and age-dependent erosion dynamics of individual chromosome arms in humans. Cytogenet Genome Res. 2006;112(3-4):194-201. doi: 10.1159/000089870. PMID: 16484772.
- Nawrot TS, Staessen JA, Gardner JP, Aviv A. Telomere length and possible link to X chromosome. Lancet. 2004 Feb 14;363(9408):507-10. doi: 10.1016/S0140-6736(04)15535-9. PMID: 14975611.
- von Zglinicki T. Oxidative stress shortens telomeres. Trends Biochem Sci. 2002 Jul;27(7):339-44. doi: 10.1016/s0968-0004(02)02110-2. PMID: 12114022.
- Aubert G, Baerlocher GM, Vulto I, Poon SS, Lansdorp PM. Collapse of telomere homeostasis in hematopoietic cells caused by heterozygous mutations in telomerase genes. PLoS Genet. 2012;8(5):e1002696. doi: 10.1371/journal.pgen.1002696. Epub 2012 May 17. PMID: 22661914; PMCID: PMC3355073.
- Shiels PG. Improving precision in investigating aging: why telomeres can cause problems. J Gerontol A Biol Sci Med Sci. 2010 Aug;65(8):789-91. doi: 10.1093/gerona/glq095. Epub 2010 Jun 10. PMID: 20538902.
- Shiels PG, McGlynn LM, MacIntyre A, Johnson PC, Batty GD, Burns H, Cavanagh J, Deans KA, Ford I, McConnachie A, McGinty A, McLean JS, Millar K, Sattar N, Tannahill C, Velupillai YN, Packard CJ. Accelerated telomere attrition is associated with relative household income, diet and inflammation in the pSoBid cohort. PLoS One. 2011;6(7):e22521. doi: 10.1371/journal.pone.0022521. Epub 2011 Jul 27. PMID: 21818333; PMCID: PMC3144896.
- Steffens JP, Masi S, D’Aiuto F, Spolidorio LC. Telomere length and its relationship with chronic diseases - new perspectives for periodontal research. Arch Oral Biol. 2013 Feb;58(2):111-7. doi: 10.1016/j.archoralbio.2012.09.009. Epub 2012 Nov 30. PMID: 23201158.
- Cawthon RM, Smith KR, O’Brien E, Sivatchenko A, Kerber RA. Association between telomere length in blood and mortality in people aged 60 years or older. Lancet. 2003 Feb 1;361(9355):393-5. doi: 10.1016/S0140-6736(03)12384-7. PMID: 12573379.
- Unryn BM, Cook LS, Riabowol KT. Paternal age is positively linked to telomere length of children. Aging Cell. 2005 Apr;4(2):97-101. doi: 10.1111/j.1474-9728.2005.00144.x. PMID: 15771613.
- Soerensen M, Thinggaard M, Nygaard M, Dato S, Tan Q, Hjelmborg J, Andersen-Ranberg K, Stevnsner T, Bohr VA, Kimura M, Aviv A, Christensen K, Christiansen L. Genetic variation in TERT and TERC and human leukocyte telomere length and longevity: a cross-sectional and longitudinal analysis. Aging Cell. 2012 Apr;11(2):223-7. doi: 10.1111/j.1474-9726.2011.00775.x. Epub 2011 Dec 28. PMID: 22136229; PMCID: PMC3303949.
- Damjanovic AK, Yang Y, Glaser R, Kiecolt-Glaser JK, Nguyen H, Laskowski B, Zou Y, Beversdorf DQ, Weng NP. Accelerated telomere erosion is associated with a declining immune function of caregivers of Alzheimer’s disease patients. J Immunol. 2007 Sep 15;179(6):4249-54. doi: 10.4049/jimmunol.179.6.4249. PMID: 17785865; PMCID: PMC2262924.
- Epel ES, Blackburn EH, Lin J, Dhabhar FS, Adler NE, Morrow JD, Cawthon RM. Accelerated telomere shortening in response to life stress. Proc Natl Acad Sci U S A. 2004 Dec 7;101(49):17312-5. doi: 10.1073/pnas.0407162101. Epub 2004 Dec 1. PMID: 15574496; PMCID: PMC534658.
- Lin J, Epel E, Cheon J, Kroenke C, Sinclair E, Bigos M, Wolkowitz O, Mellon S, Blackburn E. Analyses and comparisons of telomerase activity and telomere length in human T and B cells: insights for epidemiology of telomere maintenance. J Immunol Methods. 2010 Jan 31;352(1-2):71-80. doi: 10.1016/j.jim.2009.09.012. Epub 2009 Oct 21. PMID: 19837074; PMCID: PMC3280689.
- Sanders JL, Newman AB. Telomere length in epidemiology: a biomarker of aging, age-related disease, both, or neither? Epidemiol Rev. 2013;35(1):112-31. doi: 10.1093/epirev/mxs008. Epub 2013 Jan 9. PMID: 23302541; PMCID: PMC4707879.
- Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, Si HR, Zhu Y, Li B, Huang CL, Chen HD, Chen J, Luo Y, Guo H, Jiang RD, Liu MQ, Chen Y, Shen XR, Wang X, Zheng XS, Zhao K, Chen QJ, Deng F, Liu LL, Yan B, Zhan FX, Wang YY, Xiao GF, Shi ZL. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020 Mar;579(7798):270-273. doi: 10.1038/s41586-020-2012-7. Epub 2020 Feb 3. PMID: 32015507; PMCID: PMC7095418.
- Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, Zhao X, Huang B, Shi W, Lu R, Niu P, Zhan F, Ma X, Wang D, Xu W, Wu G, Gao GF, Tan W; China Novel Coronavirus Investigating and Research Team. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N Engl J Med. 2020 Feb 20;382(8):727-733. doi: 10.1056/NEJMoa2001017. Epub 2020 Jan 24. PMID: 31978945; PMCID: PMC7092803.
- Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z, Yu T, Xia J, Wei Y, Wu W, Xie X, Yin W, Li H, Liu M, Xiao Y, Gao H, Guo L, Xie J, Wang G, Jiang R, Gao Z, Jin Q, Wang J, Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020 Feb 15;395(10223):497-506. doi: 10.1016/S0140-6736(20)30183-5. Epub 2020 Jan 24. Erratum in: Lancet. 2020 Jan 30;: PMID: 31986264; PMCID: PMC7159299.
- Epidemiology Working Group for NCIP Epidemic Response, Chinese Center for Disease Control and Prevention. [The epidemiological characteristics of an outbreak of 2019 novel coronavirus diseases (COVID-19) in China]. Zhonghua Liu Xing Bing Xue Za Zhi. 2020 Feb 10;41(2):145-151. Chinese. doi: 10.3760/cma.j.issn.0254-6450.2020.02.003. PMID: 32064853.
- Korean Centers for Disease Control and Prevention. The updates on COVID-19 in Korea as of 9 March. 2020;4–11.
- Toyo Keizai. Patients profile of coronavirus disease (COVID-19) cases in Japan as of March 22, 2020, by age group and gender. 2020;(March). https://bit.ly/35hLhJy
- Department of Health website. Confirmed cases by age group: COVID-19 Cases: Philippines, as of April 2, 2020 Department of Health website. 2020; https://bit.ly/2Uk6ImU
- National Institute for Health and Welfare. Number of coronavirus (COVID-19) cases in Finland as of April 2, 2020, by gender. 2020;(April):1107748. https://bit.ly/38yt0tE
- Welfare NI for H and. Number of COVID-19 cases in Finland April 2020, by age group Number of coronavirus (COVID-19) cases in Finland as of April 2, 2020, by age group. 2020;(April):1103926. https://bit.ly/38yMvlY
- Needs E, March PDF. California is issuing daily updates on COVID-19-March 19, 2020. 2020;1–10. https://bit.ly/3kl5dzx
- Istituto Superiore di Sanità. Sorveglianza Integrata COVID-19 in Italia. 2020;11857:1–1. https://bit.ly/2UfPNlx
- Sas A. Coronavirus (COVID-19) new cases in Czechia 2020, by age and gender Number of new coronavirus (COVID - 19) cases confirmed in Czechia in 2020, by age and gender. 2020;1-2. https://bit.ly/3kle9Fl
- Sas A. Coronavirus (COVID-19) new cases in Estonia 2020, by age and gender Number of new coronavirus (COVID-19) cases confirmed in Estonia in 2020, by age and gender. 2020;1-2. https://bit.ly/36ojB5k
- Blackburn EH. Telomeres and telomerase: their mechanisms of action and the effects of altering their functions. FEBS Lett. 2005 Feb 7;579(4):859-62. doi: 10.1016/j.febslet.2004.11.036. PMID: 15680963.
- Cech TR. Beginning to understand the end of the chromosome. Cell. 2004 Jan 23;116(2):273-9. doi: 10.1016/s0092-8674(04)00038-8. PMID: 14744437.
- Cusanelli E, Chartrand P. Telomeric repeat-containing RNA TERRA: a noncoding RNA connecting telomere biology to genome integrity. Front Genet. 2015 Apr 14;6:143. doi: 10.3389/fgene.2015.00143. PMID: 25926849; PMCID: PMC4396414.
- Azzalin CM, Lingner J. Telomere functions grounding on TERRA firma. Trends Cell Biol. 2015 Jan;25(1):29-36. doi: 10.1016/j.tcb.2014.08.007. Epub 2014 Sep 23. PMID: 25257515.
- Luke B, Lingner J. TERRA: telomeric repeat-containing RNA. EMBO J. 2009 Sep 2;28(17):2503-10. doi: 10.1038/emboj.2009.166. Epub 2009 Jul 23. PMID: 19629047; PMCID: PMC2722245.
- Diman A, Boros J, Poulain F, Rodriguez J, Purnelle M, Episkopou H, Bertrand L, Francaux M, Deldicque L, Decottignies A. Nuclear respiratory factor 1 and endurance exercise promote human telomere transcription. Sci Adv. 2016 Jul 27;2(7):e1600031. doi: 10.1126/sciadv.1600031. PMID: 27819056; PMCID: PMC5087959.
- Weitzman MD, Weitzman JB. What’s the damage? The impact of pathogens on pathways that maintain host genome integrity. Cell Host Microbe. 2014 Mar 12;15(3):283-94. doi: 10.1016/j.chom.2014.02.010. PMID: 24629335; PMCID: PMC4501477.
- de Jong RN, van der Vliet PC, Brenkman AB. Adenovirus DNA replication: protein priming, jumping back and the role of the DNA binding protein DBP. Curr Top Microbiol Immunol. 2003;272:187-211. doi: 10.1007/978-3-662-05597-7_7. PMID: 12747551.
- Deng Z, Kim ET, Vladimirova O, Dheekollu J, Wang Z, Newhart A, Liu D, Myers JL, Hensley SE, Moffat J, Janicki SM, Fraser NW, Knipe DM, Weitzman MD, Lieberman PM. HSV-1 remodels host telomeres to facilitate viral replication. Cell Rep. 2014 Dec 24;9(6):2263-78. doi: 10.1016/j.celrep.2014.11.019. Epub 2014 Dec 11. PMID: 25497088; PMCID: PMC4356630.
- Caslini C, Connelly JA, Serna A, Broccoli D, Hess JL. MLL associates with telomeres and regulates telomeric repeat-containing RNA transcription. Mol Cell Biol. 2009 Aug;29(16):4519-26. doi: 10.1128/MCB.00195-09. Epub 2009 Jun 15. PMID: 19528237; PMCID: PMC2725733.
- Su D, Wang X, Campbell MR, Song L, Safi A, Crawford GE, Bell DA. Interactions of chromatin context, binding site sequence content, and sequence evolution in stress-induced p53 occupancy and transactivation. PLoS Genet. 2015 Jan 8;11(1):e1004885. doi: 10.1371/journal.pgen.1004885. PMID: 25569532; PMCID: PMC4287438.
- Wang Z, Lieberman PM. The crosstalk of telomere dysfunction and inflammation through cell-free TERRA containing exosomes. RNA Biol. 2016 Aug 2;13(8):690-5. doi: 10.1080/15476286.2016.1203503. Epub 2016 Jun 28. PMID: 27351774; PMCID: PMC4993293.
- Cohen S, Janicki-Deverts D, Turner RB, Casselbrant ML, Li-Korotky HS, Epel ES, Doyle WJ. Association between telomere length and experimentally induced upper respiratory viral infection in healthy adults. JAMA. 2013 Feb 20;309(7):699-705. doi: 10.1001/jama.2013.613. PMID: 23423415; PMCID: PMC3786437.
- Liu L. Fields Virology, 6th Edition. Clin Infect Dis [Internet]. 2014 May 7;59(4):613. https://bit.ly/3ncl7hr
- Kumar CS, Dey D, Ghosh S, Banerjee M. Breach: Host Membrane Penetration and Entry by Nonenveloped Viruses. Trends Microbiol. 2018 Jun;26(6):525-537. doi: 10.1016/j.tim.2017.09.010. Epub 2017 Oct 25. PMID: 29079499.
- During I, Infection V. Inflammation During Virus Infection: Swings and Roundabouts. 43–59. https://bit.ly/38wdRZO
- Guan Y, Peiris JS, Zheng B, Poon LL, Chan KH, Zeng FY, Chan CW, Chan MN, Chen JD, Chow KY, Hon CC, Hui KH, Li J, Li VY, Wang Y, Leung SW, Yuen KY, Leung FC. Molecular epidemiology of the novel coronavirus that causes severe acute respiratory syndrome. Lancet. 2004 Jan 10;363(9403):99-104. doi: 10.1016/s0140-6736(03)15259-2. PMID: 14726162; PMCID: PMC7112497.
- Peiris JS, Yuen KY, Osterhaus AD, Stöhr K. The severe acute respiratory syndrome. N Engl J Med. 2003 Dec 18;349(25):2431-41. doi: 10.1056/NEJMra032498. PMID: 14681510.
- Lew TW, Kwek TK, Tai D, Earnest A, Loo S, Singh K, Kwan KM, Chan Y, Yim CF, Bek SL, Kor AC, Yap WS, Chelliah YR, Lai YC, Goh SK. Acute respiratory distress syndrome in critically ill patients with severe acute respiratory syndrome. JAMA. 2003 Jul 16;290(3):374-80. doi: 10.1001/jama.290.3.374. PMID: 12865379.
- Farcas GA, Poutanen SM, Mazzulli T, Willey BM, Butany J, Asa SL, Faure P, Akhavan P, Low DE, Kain KC. Fatal severe acute respiratory syndrome is associated with multiorgan involvement by coronavirus. J Infect Dis. 2005 Jan 15;191(2):193-7. doi: 10.1086/426870. Epub 2004 Dec 10. PMID: 15609228; PMCID: PMC7109982.
- Liu S, Wang C, Green G, Zhuo H, Liu KD, Kangelaris KN, Gomez A, Jauregui A, Vessel K, Ke S, Hendrickson C, Matthay MA, Calfee CS, Ware LB, Wolters PJ. Peripheral blood leukocyte telomere length is associated with survival of sepsis patients. Eur Respir J. 2020 Jan 16;55(1):1901044. doi: 10.1183/13993003.01044-2019. PMID: 31619475; PMCID: PMC7359873.
- Albrecht E, Sillanpää E, Karrasch S, Alves AC, Codd V, Hovatta I, Buxton JL, Nelson CP, Broer L, Hägg S, Mangino M, Willemsen G, Surakka I, Ferreira MA, Amin N, Oostra BA, Bäckmand HM, Peltonen M, Sarna S, Rantanen T, Sipilä S, Korhonen T, Madden PA, Gieger C, Jörres RA, Heinrich J, Behr J, Huber RM, Peters A, Strauch K, Wichmann HE, Waldenberger M, Blakemore AI, de Geus EJ, Nyholt DR, Henders AK, Piirilä PL, Rissanen A, Magnusson PK, Viñuela A, Pietiläinen KH, Martin NG, Pedersen NL, Boomsma DI, Spector TD, van Duijn CM, Kaprio J, Samani NJ, Jarvelin MR, Schulz H. Telomere length in circulating leukocytes is associated with lung function and disease. Eur Respir J. 2014 Apr;43(4):983-92. doi: 10.1183/09031936.00046213. Epub 2013 Dec 5. PMID: 24311771.
- Akira S, Uematsu S, Takeuchi O. Pathogen recognition and innate immunity. Cell. 2006 Feb 24;124(4):783-801. doi: 10.1016/j.cell.2006.02.015. PMID: 16497588.
- Jensen S, Thomsen AR. Sensing of RNA viruses: a review of innate immune receptors involved in recognizing RNA virus invasion. J Virol. 2012 Mar;86(6):2900-10. doi: 10.1128/JVI.05738-11. Epub 2012 Jan 18. PMID: 22258243; PMCID: PMC3302314.
- Heil F, Hemmi H, Hochrein H, Ampenberger F, Kirschning C, Akira S, Lipford G, Wagner H, Bauer S. Species-specific recognition of single-stranded RNA via toll-like receptor 7 and 8. Science. 2004 Mar 5;303(5663):1526-9. doi: 10.1126/science.1093620. Epub 2004 Feb 19. PMID: 14976262.
- Loo YM, Gale M Jr. Immune signaling by RIG-I-like receptors. Immunity. 2011 May 27;34(5):680-92. doi: 10.1016/j.immuni.2011.05.003. PMID: 21616437; PMCID: PMC3177755.
- Amaral G, Bushee J, Cordani UG, KAWASHITA K, Reynolds JH, ALMEIDA FFMDE, et al. FosL1 determines the differential signaling by TLR7 and TLR8 during RNA virus infection. J Petrol [Internet]. 2013;369(1):1689–99. https://bit.ly/2Imuhce
- Cros J, Cagnard N, Woollard K, Patey N, Zhang SY, Senechal B, Puel A, Biswas SK, Moshous D, Picard C, Jais JP, D’Cruz D, Casanova JL, Trouillet C, Geissmann F. Human CD14dim monocytes patrol and sense nucleic acids and viruses via TLR7 and TLR8 receptors. Immunity. 2010 Sep 24;33(3):375-86. doi: 10.1016/j.immuni.2010.08.012. Epub 2010 Sep 9. PMID: 20832340; PMCID: PMC3063338.
- Chan SRWL, Blackburn EH. Telomeres and telomerase. Philos Trans R Soc B Biol Sci. 2004;359(1441):109–21. https://bit.ly/2Un1C9v
- Prasad KN, Wu M, Bondy SC. Telomere shortening during aging: Attenuation by antioxidants and anti-inflammatory agents. Mech Ageing Dev. 2017 Jun;164:61-66. doi: 10.1016/j.mad.2017.04.004. Epub 2017 Apr 18. PMID: 28431907.
- Aviv A, Valdes A, Gardner JP, Swaminathan R, Kimura M, Spector TD. Menopause modifies the association of leukocyte telomere length with insulin resistance and inflammation. J Clin Endocrinol Metab. 2006 Feb;91(2):635-40. doi: 10.1210/jc.2005-1814. Epub 2005 Nov 22. PMID: 16303830.
- Thompson D, Pepys MB, Wood SP. The physiological structure of human C-reactive protein and its complex with phosphocholine. Structure. 1999 Feb 15;7(2):169-77. doi: 10.1016/S0969-2126(99)80023-9. PMID: 10368284.
- C-reactive protein (CRP). 2020;5224:9635. https://bit.ly/3pfnWAi
- Sara M Nehring, Amandeep Goyal, Pankaj Bansal, Bhupendra C Patel. Reactive Protein (CRP). Sem des Hop [Internet]. 1989;65(5):237–44. https://bit.ly/3n5WgMq
- Jeon J-S, Rheem I, Kim JK. C - reactive protein and Respiratory Viral Infection. Korean J Clin Lab Sci. 2017;49(1):15–21. https://bit.ly/38yWkQW
- Kany S, Vollrath JT, Relja B. Cytokines in inflammatory disease. Int J Mol Sci. 2019;20(23):1–31. https://bit.ly/3eULNjZ
- Justiz Vaillant AA QA. Interleukin. 2020;1:1–8. https://bit.ly/2GQFx0b
- Chang AM, Oakland M. Biomarkers in Shortness of Breath. In: Biomarkers in Cardiovascular Disease [Internet]. Elsevier; 2019. p. 129–37. https://bit.ly/2IojvSX
- Tanaka T, Narazaki M, Kishimoto T. IL-6 in inflammation, immunity, and disease. Cold Spring Harb Perspect Biol. 2014 Sep 4;6(10):a016295. doi: 10.1101/cshperspect.a016295. PMID: 25190079; PMCID: PMC4176007.
- Gabay C. Interleukin-6 and chronic inflammation. Arthritis Res Ther. 2006;8 Suppl 2(Suppl 2):S3. doi: 10.1186/ar1917. Epub 2006 Jul 28. PMID: 16899107; PMCID: PMC3226076.
- Carrero JJ, Stenvinkel P, Fellström B, Qureshi AR, Lamb K, Heimbürger O, Bárány P, Radhakrishnan K, Lindholm B, Soveri I, Nordfors L, Shiels PG. Telomere attrition is associated with inflammation, low fetuin-A levels and high mortality in prevalent haemodialysis patients. J Intern Med. 2008 Mar;263(3):302-12. doi: 10.1111/j.1365-2796.2007.01890.x. Epub 2007 Dec 7. PMID: 18070000.
- Xu D, Erickson S, Szeps M, Gruber A, Sangfelt O, Einhorn S, Pisa P, Grandér D. Interferon alpha down-regulates telomerase reverse transcriptase and telomerase activity in human malignant and nonmalignant hematopoietic cells. Blood. 2000 Dec 15;96(13):4313-8. PMID: 11110707.
- Chung SS, Wu Y, Okobi Q, Adekoya D, Atefi M, Clarke O, Dutta P, Vadgama JV. Proinflammatory Cytokines IL-6 and TNF-α Increased Telomerase Activity through NF-κB/STAT1/STAT3 Activation, and Withaferin A Inhibited the Signaling in Colorectal Cancer Cells. Mediators Inflamm. 2017;5958429. doi: 10.1155/2017/5958429. Epub 2017 Jun 6. PMID: 28676732; PMCID: PMC5476880.
- Akiyama M, Hideshima T, Hayashi T, Tai YT, Mitsiades CS, Mitsiades N, Chauhan D, Richardson P, Munshi NC, Anderson KC. Cytokines modulate telomerase activity in a human multiple myeloma cell line. Cancer Res. 2002 Jul 1;62(13):3876-82. PMID: 12097303.
- Kordinas V, Ioannidis A, Chatzipanagiotou S. The Telomere/Telomerase System in Chronic Inflammatory Diseases. Cause or Effect? Genes (Basel). 2016 Sep 3;7(9):60. doi: 10.3390/genes7090060. PMID: 27598205; PMCID: PMC5042391.
- Ahmed W, Lingner J. PRDX1 and MTH1 cooperate to prevent ROS-mediated inhibition of telomerase. Genes Dev. 2018 May 1;32(9-10):658-669. doi: 10.1101/gad.313460.118. Epub 2018 May 17. PMID: 29773556; PMCID: PMC6004070.
- Aeby E, Ahmed W, Redon S, Simanis V, Lingner J. Peroxiredoxin 1 Protects Telomeres from Oxidative Damage and Preserves Telomeric DNA for Extension by Telomerase. Cell Rep. 2016 Dec 20;17(12):3107-3114. doi: 10.1016/j.celrep.2016.11.071. PMID: 28009281.
- Fouquerel E, Lormand J, Bose A, Lee HT, Kim GS, Li J, Sobol RW, Freudenthal BD, Myong S, Opresko PL. Oxidative guanine base damage regulates human telomerase activity. Nat Struct Mol Biol. 2016 Dec;23(12):1092-1100. doi: 10.1038/nsmb.3319. Epub 2016 Nov 7. PMID: 27820808; PMCID: PMC5140714.
Content Alerts
SignUp to our
Content alerts.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.